Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-650G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-FAK (Tyr407) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Chicken/Avian
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Endothelial neuropilin-2 influences angiogenesis by regulating actin pattern development and α5-integrin-p-FAK complex recruitment to assembling adhesion sites.
Perfluorooctanesulfonate (PFOS)-induced Sertoli cell injury through a disruption of F-actin and microtubule organization is mediated by Akt1/2.
Simultaneous deactivation of FAK and Src improves the pathology of hypertrophic scar.
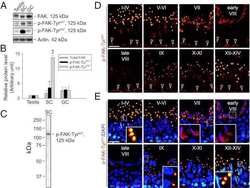
Connexin 43 reboots meiosis and reseals blood-testis barrier following toxicant-mediated aspermatogenesis and barrier disruption.
Coordination of Actin- and Microtubule-Based Cytoskeletons Supports Transport of Spermatids and Residual Bodies/Phagosomes During Spermatogenesis in the Rat Testis.
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
N-wasp is required for structural integrity of the blood-testis barrier.
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
Tumor necrosis factor α-mediated restructuring of the Sertoli cell barrier in vitro involves matrix metalloprotease 9 (MMP9), membrane-bound intercellular adhesion molecule-1 (ICAM-1) and the actin cytoskeleton.
LCB 03-0110, a novel pan-discoidin domain receptor/c-Src family tyrosine kinase inhibitor, suppresses scar formation by inhibiting fibroblast and macrophage activation.
Focal adhesion kinase-Tyr407 and -Tyr397 exhibit antagonistic effects on blood-testis barrier dynamics in the rat.
VEGF binding to NRP1 is essential for VEGF stimulation of endothelial cell migration, complex formation between NRP1 and VEGFR2, and signaling via FAK Tyr407 phosphorylation.
Cholesterol depletion induces anoikis-like apoptosis via FAK down-regulation and caveolae internalization.
Analyzing FAK and Pyk2 in early integrin signaling events.
FAK integrates growth-factor and integrin signals to promote cell migration.
Benwell CJ, Taylor JAGE, Robinson SD
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2021 Aug;35(8):e21679
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2021 Aug;35(8):e21679
Perfluorooctanesulfonate (PFOS)-induced Sertoli cell injury through a disruption of F-actin and microtubule organization is mediated by Akt1/2.
Gao Y, Chen H, Xiao X, Lui WY, Lee WM, Mruk DD, Cheng CY
Scientific reports 2017 Apr 24;7(1):1110
Scientific reports 2017 Apr 24;7(1):1110
Simultaneous deactivation of FAK and Src improves the pathology of hypertrophic scar.
Su L, Li X, Wu X, Hui B, Han S, Gao J, Li Y, Shi J, Zhu H, Zhao B, Hu D
Scientific reports 2016 May 16;6:26023
Scientific reports 2016 May 16;6:26023
Connexin 43 reboots meiosis and reseals blood-testis barrier following toxicant-mediated aspermatogenesis and barrier disruption.
Li N, Mruk DD, Mok KW, Li MW, Wong CK, Lee WM, Han D, Silvestrini B, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Apr;30(4):1436-52
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Apr;30(4):1436-52
Coordination of Actin- and Microtubule-Based Cytoskeletons Supports Transport of Spermatids and Residual Bodies/Phagosomes During Spermatogenesis in the Rat Testis.
Tang EI, Lee WM, Cheng CY
Endocrinology 2016 Apr;157(4):1644-59
Endocrinology 2016 Apr;157(4):1644-59
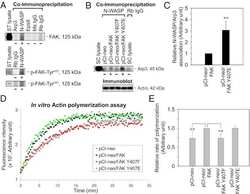
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
Robertson J, Jacquemet G, Byron A, Jones MC, Warwood S, Selley JN, Knight D, Humphries JD, Humphries MJ
Nature communications 2015 Feb 13;6:6265
Nature communications 2015 Feb 13;6:6265
N-wasp is required for structural integrity of the blood-testis barrier.
Xiao X, Mruk DD, Tang EI, Massarwa R, Mok KW, Li N, Wong CK, Lee WM, Snapper SB, Shilo BZ, Schejter ED, Cheng CY
PLoS genetics 2014 Jun;10(6):e1004447
PLoS genetics 2014 Jun;10(6):e1004447
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Hoffmann JE, Fermin Y, Stricker RL, Ickstadt K, Zamir E
eLife 2014 Jun 3;3:e02257
eLife 2014 Jun 3;3:e02257
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Hoffmann JE, Fermin Y, Stricker RL, Ickstadt K, Zamir E
eLife 2014 Jun 3;3:e02257
eLife 2014 Jun 3;3:e02257
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Wan HT, Mruk DD, Wong CK, Cheng CY
Endocrinology 2014 Jan;155(1):249-62
Endocrinology 2014 Jan;155(1):249-62
Palladin is a regulator of actin filament bundles at the ectoplasmic specialization in adult rat testes.
Qian X, Mruk DD, Wong EW, Lie PP, Cheng CY
Endocrinology 2013 May;154(5):1907-20
Endocrinology 2013 May;154(5):1907-20
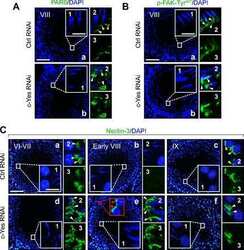
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
Xiao X, Mruk DD, Cheng CY
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
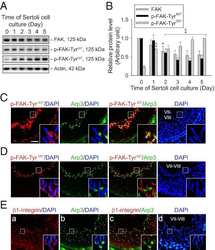
Tumor necrosis factor α-mediated restructuring of the Sertoli cell barrier in vitro involves matrix metalloprotease 9 (MMP9), membrane-bound intercellular adhesion molecule-1 (ICAM-1) and the actin cytoskeleton.
Lydka M, Bilinska B, Cheng CY, Mruk DD
Spermatogenesis 2012 Oct 1;2(4):294-303
Spermatogenesis 2012 Oct 1;2(4):294-303
LCB 03-0110, a novel pan-discoidin domain receptor/c-Src family tyrosine kinase inhibitor, suppresses scar formation by inhibiting fibroblast and macrophage activation.
Sun X, Phan TN, Jung SH, Kim SY, Cho JU, Lee H, Woo SH, Park TK, Yang BS
The Journal of pharmacology and experimental therapeutics 2012 Mar;340(3):510-9
The Journal of pharmacology and experimental therapeutics 2012 Mar;340(3):510-9
Focal adhesion kinase-Tyr407 and -Tyr397 exhibit antagonistic effects on blood-testis barrier dynamics in the rat.
Lie PP, Mruk DD, Mok KW, Su L, Lee WM, Cheng CY
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 31;109(31):12562-7
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 31;109(31):12562-7
VEGF binding to NRP1 is essential for VEGF stimulation of endothelial cell migration, complex formation between NRP1 and VEGFR2, and signaling via FAK Tyr407 phosphorylation.
Herzog B, Pellet-Many C, Britton G, Hartzoulakis B, Zachary IC
Molecular biology of the cell 2011 Aug 1;22(15):2766-76
Molecular biology of the cell 2011 Aug 1;22(15):2766-76
Cholesterol depletion induces anoikis-like apoptosis via FAK down-regulation and caveolae internalization.
Park EK, Park MJ, Lee SH, Li YC, Kim J, Lee JS, Lee JW, Ye SK, Park JW, Kim CW, Park BK, Kim YN
The Journal of pathology 2009 Jul;218(3):337-49
The Journal of pathology 2009 Jul;218(3):337-49
Analyzing FAK and Pyk2 in early integrin signaling events.
Bernard-Trifilo JA, Lim ST, Hou S, Schlaepfer DD, Ilic D
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
FAK integrates growth-factor and integrin signals to promote cell migration.
Sieg DJ, Hauck CR, Ilic D, Klingbeil CK, Schaefer E, Damsky CH, Schlaepfer DD
Nature cell biology 2000 May;2(5):249-56
Nature cell biology 2000 May;2(5):249-56
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
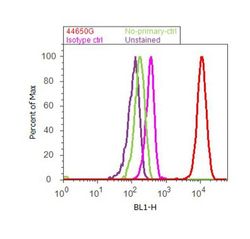
- Experimental details
- Flow cytometry analysis of FAK [pY407] was done on A549 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with FAK [pY407] Rabbit Polyclonal Antibody (44650G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor¨ 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
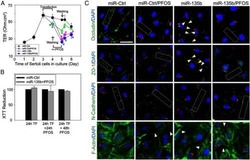
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
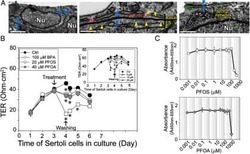
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
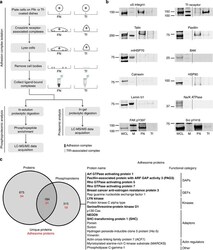
- Figure 1 Combined proteomic and phosphoproteomic analysis of isolated adhesion complexes. ( a ) Schematic workflow for the isolation and proteomic/phosphoproteomic analysis of adhesion complexes. Cells were allowed to spread on FN or, as a control, Tf and complexes were isolated by a combination of crosslinking, cell lysis and a high-pressure wash to remove cell bodies. Collected complexes were analysed using either a proteomic or phosphoproteomic workflow, after which the FN-specific proteins and phosphoproteins were identified by performing a subtractive comparison with controls. ( b ) Immunoblot analysis of complexes isolated from cells spread on FN and Tf, as well as the WCLs of cells spread on FN. M, MW markers (kDa; values displayed to the left of each blot). Dashed lines indicate where images have been cropped for display purposes. ( c ) A Venn diagram showing the overlap between the FN-specific proteins (left circle) and phosphoproteins (right circle) identified by proteomic and phosphoproteomic analyses of isolated complexes, respectively. In addition to the total number of proteins (black text), the number of adhesome proteins identified in each data set is also displayed (red text). To the right of the panel, all 19 adhesome components identified exclusively by the phosphoproteomic analysis are displayed. Proteins in bold text were not identified by any other proteomic analyses of isolated FN-induced adhesion complexes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
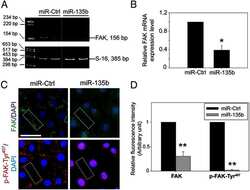
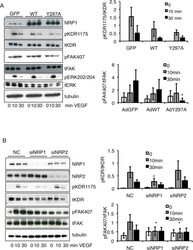
- FIGURE 6: VEGF binding to NRP1 is important for FAK Tyr-407 phosphorylation in endothelial cells. (A) HUVECs were infected with GFP, WT NRP1, or Y297A NRP1. Growth medium was replaced by serum-free medium 72 h after infection, and the following day quiescent infected cells were treated with VEGF-A 165 for the times indicated. Whole-cell extracts were then analyzed by using Western blotting with the indicated antibodies. Blots from panel A (n = 4) were quantified by scanning densitometry to determine the extent of phosphorylation KDR Y1175 and FAK Y407 (right). The bar graph shows the mean ratio of phosphorylated to total protein from four independent experiments +- SD; *p < 0.05 vs. WT 30 min VEGF treatment. (B) HUVECs were transfected with negative control siRNA (NC) or siRNAs targeted to NRP1 or NRP2, and 48 h later were treated with 25 ng/ml VEGF-A 165 for 10 or 30 min. Cells were then lysed and immunoblotted as indicated. Blots were quantified as described earlier; *p < 0.05 vs. control siRNA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
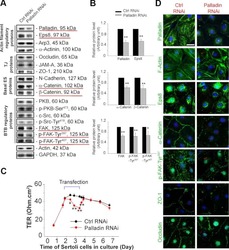
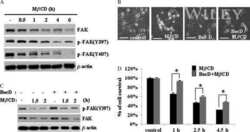
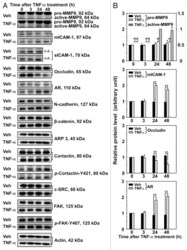
- Figure 3. Effects of TNFalpha on the steady-state levels of integral membrane, scaffolding and regulatory proteins in Sertoli cells in vitro. Sertoli cells were cultured at high density on Matrigel(tm)-coated 12-well dishes as described in Materials and Methods. On day 4 (designated as 0 h in this figure), TNFalpha (25 ng/ml) was added into Sertoli cell cultures, and cells were terminated at specific time points thereafter for lysate preparation. TNFalpha was dissolved in 10 mM NaH 2 PO 4 pH 7.4 at 22degC containing 0.15 M NaCl and 0.1% BSA (wt/vol). The control consisted of culturing Sertoli cells in media containing an equivalent amount of BSA. ( A ) Immunoblots of selected proteins involved in the regulation and in the maintenance of Sertoli cell barrier function. Actin served as an indicator of equal protein loading. ( B ) Histograms summarizing results shown in ( A ) from at least three independent experiments. Histograms are not shown for proteins whose levels did not change significantly. Each data point was normalized against its corresponding actin data point and then against the protein level at 0 h, which was arbitrarily set as 1. Each bar represents the mean +- SD of n = 3-4 experiments. **p < 0.01 (ANOVA followed by Dunnett's post-hoc test). n.d., not determined.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
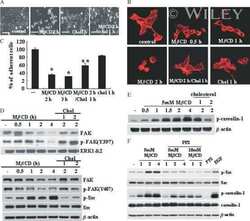
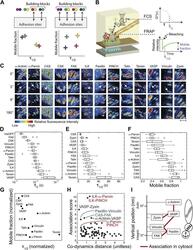
- Figure 2. The cytosolic building blocks of cell-matrix adhesion sites are combinatorially diversified. ( A ) Not all cytosolic integrin-adhesome complexes are necessarily building blocks for adhesion sites. If each protein is in only one type of building blocks then physically associated proteins should exhibit the same dwell times ( tau 1/2 ) and mobile fractions in focal adhesions. ( B ) REF52 cells expressing the analyzed proteins tagged with meGFP were measured using FRAP and FCS to quantify their tau 1/2 and mobile fractions in focal adhesions and their dwell times in a confocal volume in the cytosol ( tau D ). ( C ) Example FRAP images before (0'') and after bleaching a focal adhesion (arrows). Scale bar, 5 mum. ( D - F ) Box plots of the tau D (26 >= n >= 14 cells), tau 1/2 and mobile fractions (31 >= n >= 7 cells) of each protein. ( G ) Median tau 1/2 vs median mobile fraction of each protein normalized to zero-mean and unit-variance. Thus, in this plot the Euclidean distance (co-dynamics distance) between proteins quantifies the difference in their dynamics. ( H ) The co-dynamics distance vs median association score of all possible 78 heteromeric protein pairs. ( I ) The reported vertical distance from substrate of 6 of the analyzed components across focal adhesions ( Kanchanawong et al., 2010 ) and the cytosolic associations between them as measured here ( Figure 1E ). Anchor, spring, and actin symbols indicate vertical layers of integrin signalling, mecha
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
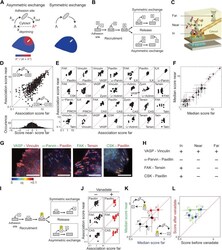
- Figure 3. Symmetric material exchange between stationary focal adhesions and cytosol. ( A ) The symmetric and asymmetric models of material exchange between adhesion sites and cytosol. In symmetric exchange a component, A , exits from adhesion site in the same state it had upon entering to it. In asymmetric exchange A exits in a different, primed state A* and relaxes back to state A in the cytosol, thereby generating a spatial gradient of the primed state emanating from adhesion sites. ( B ) Formulation of the two models for the case in which priming (of A ) is based on interaction (with protein B ). Here, asymmetric exchange would generate a spatial gradient of AB complex concentration emanating from adhesion sites. ( C - F ) Discriminating between the two models by measuring the physical associations near (= 9) ( E ) or for the median score of each pair +- MAD ( F ). Data-points far from the equality diagonals (dashed red lines) would correspond to asymmetric exchanges, as illustrated in ( D ), while data-points along the diagonal indicate symmetric exchange. Histogram in ( D ) shows the distribution of the difference in association scores near and far from focal adhesions. ( G ) FLIM images color-coding the fraction, alpha, of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
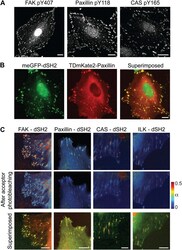
- Experimental details
- Figure 3--figure supplement 1. FAK, paxillin, and CAS are tyrosine-phosphorylated and interact with SH2 domain in focal adhesions. ( A ) Immunofluorescence images of REF52 stained for FAK pY407, paxillin pY118, and CAS pY165 using phospho-specific antibodies. Scale bars, 15 mum. ( B ) A REF52 cell co-expressing meGFP-dSH2 and TD-mKate2-paxillin. Note the localization of dSH2 in cell-matrix adhesion sites, as previously reported ( Kirchner et al., 2003 ). Scale bar, 10 mum. ( C ) Top and middle rows, FLIM images color-coding the fraction (alpha) of the donor- (mCitrine) tagged protein (FAK, paxillin, CAS, or ILK) that FRET to the acceptor- (mCherry) tagged dSH2 before and after acceptor photobleaching. ILK is used here as a negative control for tyrosine phosphorylation. Note that low levels of alpha are observed also for ILK in focal adhesions, plausibly due to density effects. However, FAK, paxillin, and CAS exhibit higher alpha values, suggesting FRET due to direct interaction. Scale bars, 10 mum. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
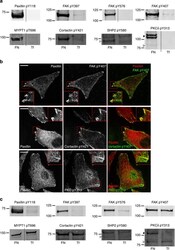
- Experimental details
- Figure 4 Validation of adhesion complex-localized phosphorylation sites. ( a ) Immunoblot analyses of the protein complexes isolated from cells spread on FN or Tf for 120 min. Samples were probed using phosphospecific antibodies targeting: paxillin pTyr118; FAK pTyr397, pTyr576 and pTyr407; MYPT1 pThr696; cortactin pTyr421; SHP2 pTyr580; and PKCdelta pTyr313. ( b ) Immunofluorescence images of cells spread on FN for 120 min. Cells were stained using the phosphospecific antibodies FAK pTyr407, cortactin pTyr421 and PKCdelta pTyr313 (all green). In each case, cells were also stained using an antibody targeting the focal adhesion marker paxillin (red). Scale bar, 20 mum. ( c ) Immunoblot analyses of the WCLs of cells spread on FN or Tf for 120 min. Samples were probed using the same antibodies as used to probe isolated complexes in a . Dashed lines in a , c indicate where the images have been cropped for display purposes. White arrows to left of blots highlight the expected MWs of the corresponding proteins. Black arrows to the left of blots highlight a putative high MW isoform of PKCdelta.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry