MA5-12394
antibody from Invitrogen Antibodies
Targeting: CD44
CD44R, CSPG8, HCELL, IN, MC56, MDU2, MDU3, MIC4, Pgp1
Antibody data
- Antibody Data
- Antigen structure
- References [24]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12394 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD44 Monoclonal Antibody (5F12)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA5-12394 has been successfully used in immunofluorescence analysis of CD44 in human bone marrow derived mesenchymal stem cells.
- Antibody clone number
- 5F12
- Concentration
- 0.2 mg/mL
Submitted references Regulation of amyloid-β levels by matrix metalloproteinase-2/9 (MMP2/9) in the media of lung cancer cells.
IGFBP-3 Blocks Hyaluronan-CD44 Signaling, Leading to Increased Acetylcholinesterase Levels in A549 Cell Media and Apoptosis in a p53-Dependent Manner.
Biochemical determinants of the IGFBP-3-hyaluronan interaction.
Neighboring cells override 3D hydrogel matrix cues to drive human MSC quiescence.
Bi-directional cell-pericellular matrix interactions direct stem cell fate.
Soluble CD83 Inhibits T Cell Activation by Binding to the TLR4/MD-2 Complex on CD14(+) Monocytes.
Serglycin in tumor microenvironment promotes non-small cell lung cancer aggressiveness in a CD44-dependent manner.
Breast cancer cells respond differentially to modulation of TGFβ2 signaling after exposure to chemotherapy or hypoxia.
CD44 mediated hyaluronan adhesion of Toxoplasma gondii-infected leukocytes.
Hyaluronan initiates chondrogenesis mainly via CD44 in human adipose-derived stem cells.
Differentiation of human mesenchymal stem cells: the potential mechanism for estrogen-induced preferential osteoblast versus adipocyte differentiation.
Inhibition of CD44 N- and O-linked glycosylation decreases endometrial cell lines attachment to peritoneal mesothelial cells.
Role of hyaluronan and hyaluronan-binding proteins in human asthma.
Adhesion-mediated self-renewal abilities of Ph+ blastoma cells.
An evidence for adhesion-mediated acquisition of acute myeloid leukemic stem cell-like immaturities.
High molecular weight hyaluronic acid inhibits IL-6-induced MMP production from human chondrocytes by up-regulating the ERK inhibitor, MKP-1.
Hyaluronan receptors involved in cytokine induction in monocytes.
Inhibition of human pancreatic cell line MIA PaCa2 proliferation by HA-But, a hyaluronic butyric ester: a preliminary report.
Suppressive effects of hyaluronan on MMP-1 and RANTES production from chondrocytes.
High molecular weight hyaluronic acid down-regulates the gene expression of osteoarthritis-associated cytokines and enzymes in fibroblast-like synoviocytes from patients with early osteoarthritis.
Mesenchymal stem cells transduced by PLEGFP-N1 retroviral vector maintain their biological features and differentiation.
Inhibition of hepatocellular carcinomas in vitro and hepatic metastases in vivo in mice by the histone deacetylase inhibitor HA-But.
Hyaluronic-acid butyric esters as promising antineoplastic agents in human lung carcinoma: a preclinical study.
Hyaluronic acid or TNF-alpha plus fibronectin triggers granulocyte macrophage-colony-stimulating factor mRNA stabilization in eosinophils yet engages differential intracellular pathways and mRNA binding proteins.
Dorandish S, Williams A, Atali S, Sendo S, Price D, Thompson C, Guthrie J, Heyl D, Evans HG
Scientific reports 2021 May 6;11(1):9708
Scientific reports 2021 May 6;11(1):9708
IGFBP-3 Blocks Hyaluronan-CD44 Signaling, Leading to Increased Acetylcholinesterase Levels in A549 Cell Media and Apoptosis in a p53-Dependent Manner.
Price D, Muterspaugh R, Clegg B, Williams A, Stephens A, Guthrie J, Heyl D, Evans HG
Scientific reports 2020 Mar 19;10(1):5083
Scientific reports 2020 Mar 19;10(1):5083
Biochemical determinants of the IGFBP-3-hyaluronan interaction.
Dorandish S, Devos J, Clegg B, Price D, Muterspaugh R, Guthrie J, Heyl DL, Evans HG
FEBS open bio 2020 Aug;10(8):1668-1684
FEBS open bio 2020 Aug;10(8):1668-1684
Neighboring cells override 3D hydrogel matrix cues to drive human MSC quiescence.
Ferreira SA, Faull PA, Seymour AJ, Yu TTL, Loaiza S, Auner HW, Snijders AP, Gentleman E
Biomaterials 2018 Sep;176:13-23
Biomaterials 2018 Sep;176:13-23
Bi-directional cell-pericellular matrix interactions direct stem cell fate.
Ferreira SA, Motwani MS, Faull PA, Seymour AJ, Yu TTL, Enayati M, Taheem DK, Salzlechner C, Haghighi T, Kania EM, Oommen OP, Ahmed T, Loaiza S, Parzych K, Dazzi F, Varghese OP, Festy F, Grigoriadis AE, Auner HW, Snijders AP, Bozec L, Gentleman E
Nature communications 2018 Oct 3;9(1):4049
Nature communications 2018 Oct 3;9(1):4049
Soluble CD83 Inhibits T Cell Activation by Binding to the TLR4/MD-2 Complex on CD14(+) Monocytes.
Horvatinovich JM, Grogan EW, Norris M, Steinkasserer A, Lemos H, Mellor AL, Tcherepanova IY, Nicolette CA, DeBenedette MA
Journal of immunology (Baltimore, Md. : 1950) 2017 Mar 15;198(6):2286-2301
Journal of immunology (Baltimore, Md. : 1950) 2017 Mar 15;198(6):2286-2301
Serglycin in tumor microenvironment promotes non-small cell lung cancer aggressiveness in a CD44-dependent manner.
Guo JY, Hsu HS, Tyan SW, Li FY, Shew JY, Lee WH, Chen JY
Oncogene 2017 Apr 27;36(17):2457-2471
Oncogene 2017 Apr 27;36(17):2457-2471
Breast cancer cells respond differentially to modulation of TGFβ2 signaling after exposure to chemotherapy or hypoxia.
O'Brien SK, Chen L, Zhong W, Armellino D, Yu J, Loreth C, Follettie M, Damelin M
Cancer research 2015 Nov 1;75(21):4605-16
Cancer research 2015 Nov 1;75(21):4605-16
CD44 mediated hyaluronan adhesion of Toxoplasma gondii-infected leukocytes.
Hayashi T, Unno A, Baba M, Ohno T, Kitoh K, Takashima Y
Parasitology international 2014 Apr;63(2):479-84
Parasitology international 2014 Apr;63(2):479-84
Hyaluronan initiates chondrogenesis mainly via CD44 in human adipose-derived stem cells.
Wu SC, Chen CH, Chang JK, Fu YC, Wang CK, Eswaramoorthy R, Lin YS, Wang YH, Lin SY, Wang GJ, Ho ML
Journal of applied physiology (Bethesda, Md. : 1985) 2013 Jun;114(11):1610-8
Journal of applied physiology (Bethesda, Md. : 1985) 2013 Jun;114(11):1610-8
Differentiation of human mesenchymal stem cells: the potential mechanism for estrogen-induced preferential osteoblast versus adipocyte differentiation.
Zhao JW, Gao ZL, Mei H, Li YL, Wang Y
The American journal of the medical sciences 2011 Jun;341(6):460-8
The American journal of the medical sciences 2011 Jun;341(6):460-8
Inhibition of CD44 N- and O-linked glycosylation decreases endometrial cell lines attachment to peritoneal mesothelial cells.
Rodgers AK, Nair A, Binkley PA, Tekmal R, Schenken RS
Fertility and sterility 2011 Feb;95(2):823-5
Fertility and sterility 2011 Feb;95(2):823-5
Role of hyaluronan and hyaluronan-binding proteins in human asthma.
Liang J, Jiang D, Jung Y, Xie T, Ingram J, Church T, Degan S, Leonard M, Kraft M, Noble PW
The Journal of allergy and clinical immunology 2011 Aug;128(2):403-411.e3
The Journal of allergy and clinical immunology 2011 Aug;128(2):403-411.e3
Adhesion-mediated self-renewal abilities of Ph+ blastoma cells.
Funayama K, Saito-Kurimoto Y, Ebihara Y, Shimane M, Nomura H, Tsuji K, Asano S
Biochemical and biophysical research communications 2010 May 28;396(2):193-8
Biochemical and biophysical research communications 2010 May 28;396(2):193-8
An evidence for adhesion-mediated acquisition of acute myeloid leukemic stem cell-like immaturities.
Funayama K, Shimane M, Nomura H, Asano S
Biochemical and biophysical research communications 2010 Feb 12;392(3):271-6
Biochemical and biophysical research communications 2010 Feb 12;392(3):271-6
High molecular weight hyaluronic acid inhibits IL-6-induced MMP production from human chondrocytes by up-regulating the ERK inhibitor, MKP-1.
Hashizume M, Mihara M
Biochemical and biophysical research communications 2010 Dec 10;403(2):184-9
Biochemical and biophysical research communications 2010 Dec 10;403(2):184-9
Hyaluronan receptors involved in cytokine induction in monocytes.
Yamawaki H, Hirohata S, Miyoshi T, Takahashi K, Ogawa H, Shinohata R, Demircan K, Kusachi S, Yamamoto K, Ninomiya Y
Glycobiology 2009 Jan;19(1):83-92
Glycobiology 2009 Jan;19(1):83-92
Inhibition of human pancreatic cell line MIA PaCa2 proliferation by HA-But, a hyaluronic butyric ester: a preliminary report.
Pellizzaro C, Speranza A, Zorzet S, Crucil I, Sava G, Scarlata I, Cantoni S, Fedeli M, Coradini D
Pancreas 2008 May;36(4):e15-23
Pancreas 2008 May;36(4):e15-23
Suppressive effects of hyaluronan on MMP-1 and RANTES production from chondrocytes.
Tanaka M, Masuko-Hongo K, Kato T, Nishioka K, Nakamura H
Rheumatology international 2006 Jan;26(3):185-90
Rheumatology international 2006 Jan;26(3):185-90
High molecular weight hyaluronic acid down-regulates the gene expression of osteoarthritis-associated cytokines and enzymes in fibroblast-like synoviocytes from patients with early osteoarthritis.
Wang CT, Lin YT, Chiang BL, Lin YH, Hou SM
Osteoarthritis and cartilage 2006 Dec;14(12):1237-47
Osteoarthritis and cartilage 2006 Dec;14(12):1237-47
Mesenchymal stem cells transduced by PLEGFP-N1 retroviral vector maintain their biological features and differentiation.
He X, Li YL, Wang XR, Guo X, Niu Y
Chinese medical journal 2005 Oct 20;118(20):1728-34
Chinese medical journal 2005 Oct 20;118(20):1728-34
Inhibition of hepatocellular carcinomas in vitro and hepatic metastases in vivo in mice by the histone deacetylase inhibitor HA-But.
Coradini D, Zorzet S, Rossin R, Scarlata I, Pellizzaro C, Turrin C, Bello M, Cantoni S, Speranza A, Sava G, Mazzi U, Perbellini A
Clinical cancer research : an official journal of the American Association for Cancer Research 2004 Jul 15;10(14):4822-30
Clinical cancer research : an official journal of the American Association for Cancer Research 2004 Jul 15;10(14):4822-30
Hyaluronic-acid butyric esters as promising antineoplastic agents in human lung carcinoma: a preclinical study.
Coradini D, Pellizzaro C, Abolafio G, Bosco M, Scarlata I, Cantoni S, Stucchi L, Zorzet S, Turrin C, Sava G, Perbellini A, Daidone MG
Investigational new drugs 2004 Aug;22(3):207-17
Investigational new drugs 2004 Aug;22(3):207-17
Hyaluronic acid or TNF-alpha plus fibronectin triggers granulocyte macrophage-colony-stimulating factor mRNA stabilization in eosinophils yet engages differential intracellular pathways and mRNA binding proteins.
Esnault S, Malter JS
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 15;171(12):6780-7
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 15;171(12):6780-7
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
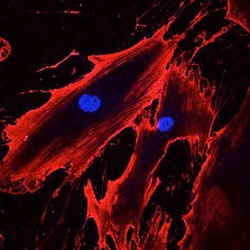
- Experimental details
- Immunofluorescent analysis of CD44 (red) in human bone marrow derived mesenchymal stem cells. The cells were fixed with 2% paraformaldehyde, and blocked with 2% normal donkey serum diluted in PBS for 30 minutes at room temperature. Cells were stained with a CD44 monoclonal antibody (Product # MA5-12394) at a dilution of 10 µg/mL in blocking buffer for 1 hour at room temperature, and then incubated with a Goat anti-mouse IgG (H+L) Secondary Antibody, Alexa Fluor® 555 conjugate (Product # A-21422) at a dilution of 1:800 for 1 hour at room temperature (red). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Note: Data courtesy of Innovators Program.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
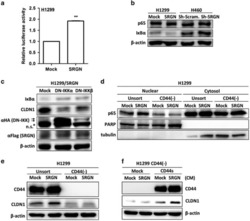
- Experimental details
- Figure 6 SRGN/CD44 axis induces CLDN1 expression via NF-kappaB activation. ( a ) NF-kappaB reporter assay was performed in H1299/Mock and H1299/SRGN cells. ( b ) Cells were cultured in serum-free medium for 48 h, and subjected to western blot analysis using anti-IkappaBalpha (#4812, Cell Signaling Technology) and anti-p65 (sc-372, Santa Cruz Biotech). ( c ) H1299/SRGN cells were transiently transfected with vectors encoding dominant-negative IkappaB kinase alpha (DN-IKKalpha) or DN-IKKbeta and cultured in serum-free medium for 48 h, followed by western blot analysis of IkappaB and CLDN1. HA-fused DN-IKKs were detected by anti-hemagglutinin (sc-805, Santa Cruz Biotech). ( d ) Unsorted H1299 and CD44(-) cells stably harboring the Mock-control or SRGN-expressing vectors were cultured in serum-free medium for 48 h. Nuclear and cytosolic fractions were prepared for western blot analysis using anti-p65, anti-PARP (sc-7150, Santa Cruz Biotech) and anti-tubulin (GTX112141, GeneTex). ( e ) Cells described in d were cultured in serum-free medium for 48 h, and subjected to western blotting of CD44 and CLDN1. ( f ) H1299 CD44(-) cells stably harboring Mock-control or CD44s-expressing vectors were incubated in medium supplemented with CM collected from H1299/Mock or H1299/SRGN cells for 24 h, and subjected to western blot analysis of CLDN1. Data are presented as the mean+-s.d. of three independent experiments. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
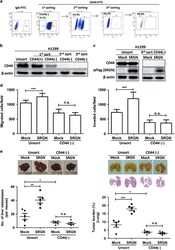
- Experimental details
- Figure 3 CD44 is critical for SRGN-instigated malignant phenotypes. ( a ) CD44-negative H1299 cells were enriched by three rounds of FACS procedure. ( b and c ) Western blot analysis was performed to show the expression of CD44 (using Hermes-3, ATCC) and SRGN (using anti-Flag M2, Sigma-Aldrich) in unsorted and CD44(-) cells ( b ), and in cells stably harboring the Mock-control and SRGN-expressing vectors ( c ). ( d ) Migration and invasion assay. ( e ) 2 x 10 6 cells were injected to NOD-SCID mice through tail-vein, and tumors developed in the liver (left panel) and lung (right panel) were assessed. ( f ) H1299 CD44(-) cells stably harboring Mock-control vector as well as vector encoding CD44s were subjected to Boyden chamber migration assay in the presence of CM collected from H1299/Mock and H1299/SRGN cells. Western blot analysis shows CD44 expression in H1299 CD44(-)/Mock and H1299 CD44(-)/CD44s cells. ( g , h ) Migration assays of HTB-40/Mock and HTB-40/CD44 cells incubated with SRGN-containing CM ( g ) or with serum-free medium supplemented with or without recombinant human SRGN (5 mug/ml) ( h ). Western blot analysis shows CD44 expression. Data are presented as the mean+-s.d. of three independent experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
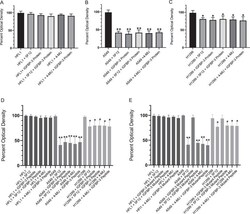
- Experimental details
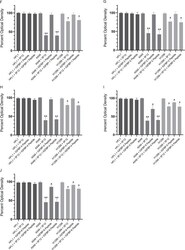
- Figure 3(A-E) The IGFBP-3 protein and WT peptide, but not the mutant, block HA-CD44 signaling and more effectively inhibit cell viability of A549 cells than either the p53-negative H1299 or CD44-negative HFL1 cell lines. IGFBP-3 protein or peptides were added to cells in the absence or presence of the CD44 antibody, 5F12, known to block HA-CD44 interactions, or 4-MU. Cell viability was assessed by the MTT assay. Cells were seeded in 96-well plates at 0.2 x 10 5 cells per well in 10% FBS-supplemented media. The following day, the cell monolayers were incubated in serum-free medium for 12h, then treated as indicated for 48h with the media containing the specific components in the different treatments replaced every 12h. The concentration of IGFBP-3 protein or peptides added was 50 nM and that of 4-MU was 600 muM. The CD44 antibody (5 mug/mL) was added either separately or 2h prior to addition of IGFBP-3 and/or peptides. Optical density measurements (570 nm) were normalized by expressing each point in relation to the untreated control of each cell line (set to 100%). Each column represents the mean +- S.D. of three independent experiments, each run in triplicate. Asterisks (*) indicate a statistically significant difference from the corresponding untreated cell line control, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3(F-J) The IGFBP-5 and IGFBP-6 peptides are able to block HA-CD44 signaling and cell viability of A549 cells, albeit less effectively than the IGFBP-3 peptide. Cell viability was measured as described in ( A-E ) legend.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
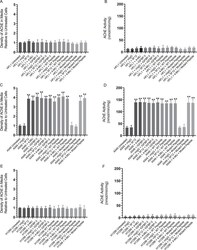
- Figure 5 Both AChE concentration and activity are increased to a comparable extent in the media of A549 cells upon treatment with the IGFBP-3 protein or peptide, 5F12, 4-MU, or in combination. Cells (0.2 x 10 5 ) were grown in 10% FBS-supplemented media overnight then in serum-free medium for 12 h prior to treatment with mIgG (5 mug/mL), 5F12 (5 mug/mL), 50 nM IGFBP-3 protein or peptides, 600 uM 4-MU, or in combination. The cells were then incubated for 48 h and the media collected. Samples (3 uL of 600 ug/mL total protein) were spotted onto a nitrocellulose membrane and AChE was visualized using anti-AChE antibodies (Methods). The dots were quantitated, averaged, normalized and expressed as fold change relative to untreated cells ( A,C,E ). AChE activity using the same amount of protein was measured as described in Methods ( B,D,F ). The graphs prepared using the GraphPad 8.3.1 software, summarize the results expressed as means +- S.D. of five independent experiments, each performed in triplicate. Asterisks (*) indicate a statistically significant difference from the corresponding untreated cell line control, *p < 0.05, **p < 0.0 l of each cell line. Absence of asterisks indicates no significance, Mann-Whitney test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 WT IGFBP-3 l -peptide is more effective than the d -peptide in both binding HA and in blocking viability of A549 cells that express CD44 as compared to the CD44-negative cell line, HFL1. (A) IGFBP-3 peptides (50 n m each) were bound to the ELISA plate wells, and then, 200 n m biotin-HA was added and processed as described in the Materials and methods section. The data were normalized to the control incubated with BSA (control 1), and fold change relative to the control was calculated. Control 2 is a negative control that included bound WT IGFBP-3 l -peptide and added streptavidin-HRP and TMB without addition of biotin-HA. Each column represents the mean +- SD of three independent experiments, each run in triplicate. The asterisks (** P < 0.01) indicate a statistically significant difference from control 1 and of the IGFBP-3 l -peptide compared to the d -counterpart. The absence of asterisks indicates no significance, Mann-Whitney test. (B, C) IGFBP-3 peptides were added to cells in the absence or presence of the mIgG (5 mug*mL -1 ) antibody control or the CD44, 5F12 antibody (5 mug*mL -1 ), known to block HA-CD44 interactions. Cell viability was assessed by the MTT assay. Cells were seeded in 96-well plates at 0.2 x 10 5 cells per well in 10% FBS-supplemented media. The following day, the cell monolayers were incubated in serum-free medium for 12 h and then treated as indicated for 48 h with the media containing the specific components in the different treatments repla
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
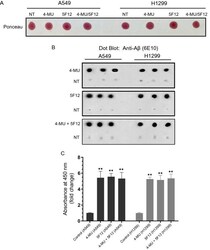
- Figure 1 Treatment of cells with either 4-MU or 5F12 resulted in increased Abeta in the media of A549 and H1299 cells. Cells (0.2 x 10 5 ) were grown in 10% FBS-supplemented media for 24 h. The following day, the cell monolayers were incubated in serum-free media for 24 h, then treated as indicated for 72 h with 600 uM 4-MU and/or the CD44 antibody (5F12, 5 mug/mL). The media was collected then the same amount of protein (3 uL of 600 ug/mL total protein) of each sample not-treated (NT) or treated as indicated, was spotted onto a nitrocellulose membrane. The blots were stained with Ponceau ( A ). ( B ) Blots were incubated with anti-Abeta (6E10) antibodies, and the signal on the membrane was detected using super signal west pico luminol (chemiluminescence) reagent. The membranes were imaged with a Bio-Rad molecular imager, and quantitated with Image J. The dots from five independent assays, each carried out in triplicate, were quantitated, averaged, normalized, and expressed as fold change relative to untreated control cells after subtraction of the values for the blank consisting of dot blots probed with mouse IgG isotype control with no relevant specificity to a target antigen (mIgG, 5 mug/mL) or anti-Abeta (6E10) antibodies incubated with blots dotted with media not incubated with cells ( C ) using the GraphPad 8.4.3 software. The graphs summarize the results expressed as means +- SD (n = 5). Asterisks (*) indicate a statistically significant difference from the correspondi
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry