Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-3179-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD317 (BST2, PDCA-1) Monoclonal Antibody (26F8), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This 26F8 monoclonal antibody reacts with human CD317 (also known as BST2 and tetherin). CD317 is a 30-36-kDa type II transmembrane protein expressed on B cells and bone marrow stromal cells. Although reports have indicated that CD317 mRNA is detectable in activated T cells, protein expression in freshly isolated primary T cells and macrophages is undetectable. Moreover, certain T cell lines, such as Jurkat, do not express detectable levels of CD317 protein. However, CD317 expression can be induced in many cell types (e.g., T and B cells, 293T, and HeLa) by IFNalpha treatment. CD317 has been associated with pre-B cell growth and the terminal differentiation of plasma B cells. More recently, this molecule has been reported to prevent HIV-1 virion release from the surface of infected cells, leading to reuptake and degradation of the virus. This activity is inhibited by the HIV-1 accessory protein Vpu. CD317 has been identified as the ligand for the ILT7 receptor.
- Conjugate
- Yellow dye
- Antibody clone number
- 26F8
- Concentration
- 5 µL/Test
Submitted references Japanese encephalitis virus counteracts BST2 restriction via its envelope protein E.
CD317 is over-expressed in B-cell chronic lymphocytic leukemia, but not B-cell acute lymphoblastic leukemia.
Nef proteins of epidemic HIV-1 group O strains antagonize human tetherin.
Ig-like transcript 7, but not bone marrow stromal cell antigen 2 (also known as HM1.24, tetherin, or CD317), modulates plasmacytoid dendritic cell function in primary human blood leukocytes.
Vpu enhances HIV-1 virus release in the absence of Bst-2 cell surface down-modulation and intracellular depletion.
Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu.
The interferon-induced protein BST-2 restricts HIV-1 release and is downregulated from the cell surface by the viral Vpu protein.
Molecular cloning and chromosomal mapping of a bone marrow stromal cell surface gene, BST2, that may be involved in pre-B-cell growth.
Li M, Wang P, Zheng Z, Hu K, Zhang M, Guan X, Fu M, Zhang D, Wang W, Xiao G, Hu Q, Liu Y
Virology 2017 Oct;510:67-75
Virology 2017 Oct;510:67-75
CD317 is over-expressed in B-cell chronic lymphocytic leukemia, but not B-cell acute lymphoblastic leukemia.
Gong S, Osei ES, Kaplan D, Chen YH, Meyerson H
International journal of clinical and experimental pathology 2015;8(2):1613-21
International journal of clinical and experimental pathology 2015;8(2):1613-21
Nef proteins of epidemic HIV-1 group O strains antagonize human tetherin.
Kluge SF, Mack K, Iyer SS, Pujol FM, Heigele A, Learn GH, Usmani SM, Sauter D, Joas S, Hotter D, Bibollet-Ruche F, Plenderleith LJ, Peeters M, Geyer M, Sharp PM, Fackler OT, Hahn BH, Kirchhoff F
Cell host & microbe 2014 Nov 12;16(5):639-50
Cell host & microbe 2014 Nov 12;16(5):639-50
Ig-like transcript 7, but not bone marrow stromal cell antigen 2 (also known as HM1.24, tetherin, or CD317), modulates plasmacytoid dendritic cell function in primary human blood leukocytes.
Tavano B, Galao RP, Graham DR, Neil SJ, Aquino VN, Fuchs D, Boasso A
Journal of immunology (Baltimore, Md. : 1950) 2013 Mar 15;190(6):2622-30
Journal of immunology (Baltimore, Md. : 1950) 2013 Mar 15;190(6):2622-30
Vpu enhances HIV-1 virus release in the absence of Bst-2 cell surface down-modulation and intracellular depletion.
Miyagi E, Andrew AJ, Kao S, Strebel K
Proceedings of the National Academy of Sciences of the United States of America 2009 Feb 24;106(8):2868-73
Proceedings of the National Academy of Sciences of the United States of America 2009 Feb 24;106(8):2868-73
Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu.
Neil SJ, Zang T, Bieniasz PD
Nature 2008 Jan 24;451(7177):425-30
Nature 2008 Jan 24;451(7177):425-30
The interferon-induced protein BST-2 restricts HIV-1 release and is downregulated from the cell surface by the viral Vpu protein.
Van Damme N, Goff D, Katsura C, Jorgenson RL, Mitchell R, Johnson MC, Stephens EB, Guatelli J
Cell host & microbe 2008 Apr 17;3(4):245-52
Cell host & microbe 2008 Apr 17;3(4):245-52
Molecular cloning and chromosomal mapping of a bone marrow stromal cell surface gene, BST2, that may be involved in pre-B-cell growth.
Ishikawa J, Kaisho T, Tomizawa H, Lee BO, Kobune Y, Inazawa J, Oritani K, Itoh M, Ochi T, Ishihara K
Genomics 1995 Apr 10;26(3):527-34
Genomics 1995 Apr 10;26(3):527-34
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
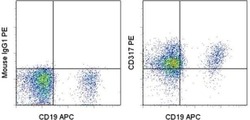
- Experimental details
- Normal human peripheral blood cells were treated with human IFN alpha 2 and then stained with Anti-Human CD19 (HIB19) APC (Product # 17-0199-42) and Mouse IgG1, K Isotype Control PE (Product # 12-4714-81) (left) or Anti-Human CD317 (BST2, PDCA-1) PE (right). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
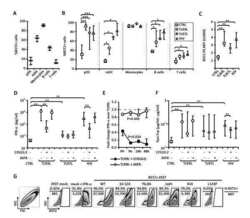
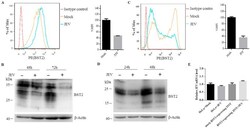
- Fig. 2 Downregulation of endogenous BST2 enhances the release of JEV progeny virions. (A) HeLa cells were transfected with BST2-specific shRNA or control shRNA. At 48 h post infection, cells were harvested and lysed for western blot analysis. (B and C) HeLa cells transfected with BST2 shRNA or non-targeting shRNA were collected at different time points and analyzed by western blot (B) and flow cytometry (C). (D and E) HeLa cells were infected with JEV at an MOI of 25 following 6 h transfection with BST2-specific shRNA or control shRNA, and the titers of supernatant virus (SV) or cell-associated virus (CV) were determined by plaque assay at the indicated time points. (F) HeLa cells were infected with JEV at an MOI of 25 following 6 h transfection with BST2 shRNA or control shRNA. 293T cells were transfected with pBST2 or pcDNA3.1. The levels of protein E in SV or CV were determined by western blot assay at 24 h post infection. Data shown are mean +- SD of three independent experiments with each condition performed in triplicate. Compared to control shRNA, *P < 0.05; **P < 0.01; ***P < 0.001. Fig. 2
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
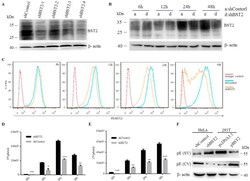
- Fig. 3 JEV infection downregulates the expression of BST2. (A) HeLa cells were mock-infected or infected with JEV at an MOI of 25. At 48 h post infection, cells were fixed and probed with PE-conjugated anti-BST2 antibody. Cells were then analyzed by flow cytometry. The bar graph is mean +- SD of MFI from three independent experiments with one representative histogram being shown. (B) At 48 or 72 h post infection, HeLa cells infected or uninfected with JEV were collected and analyzed by western blot. (C) 293T cells were transfected with plasmid expressing BST2 or empty plasmid. At 6 h post transfection, cells were infected with JEV, collected at 24 h post infection and stained with anti-BST2 antibody for flow cytometry analysis. The bar graph is mean +- SD of MFI from three independent experiments with one representative histogram being shown. (D) At 24 or 48 h post infection, 293T cells infected or uninfected with JEV were collected and analyzed by western blot. One representative experiment out of three is shown. (E) HeLa cells were transfected with control shRNA or BST2 specific shRNA, infected with JEV at an MOI of 25. 293T cells were transfected with empty plasmid or plasmid expressing BST2, infected with JEV at an MOI of 10. The total RNA of the samples was extracted, and the BST2 mRNA was determined by RT-PCR. The mRNA level of GAPDH was scored in parallel and used as an internal control. Data shown are mean +- SD of three independent experiments. Fig. 3
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
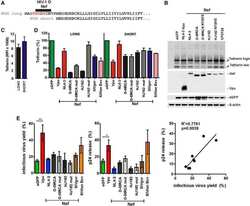
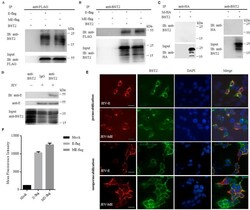
- Fig. 5 JEV protein E physically interacts with BST2. 293T cells were cotransfected with pBST2 and plasmid expressing E-flag, ME-flag or M-HA. At 48 h post transfection, cell lysates were analyzed by co-IP. Co-IP was pulled down using the anti-BST2, anti-flag or anti-HA antibody. Proteins were immunoprecipitated with the anti-flag (A) or anti-BST2 antibody (B) as indicated. (C) Proteins were immunoprecipitated with the anti-HA or anti-BST2 antibody as indicated. (D) HeLa cells were infected with JEV at a MOI of 25. At 48 h post infection, cell lysates were analyzed by co-IP. Co-IP was pulled down using the anti-BST2 antibody. One representative experiment out of three is shown. (E) Colocalization of BST2 with JEV E-flag or ME-flag. 293T cells cotransfected with pBST2 and plasmid expressing E-flag or ME-flag were costained with anti-flag (red) and anti-BST2 (green) antibodies. Nuclei were counterstained with DAPI (blue). Representative confocal images from three independent experiments are shown. Scale bars in all panels represent 10 um. (F) HeLa cells were transfected with plasmid expressing protein E. The surface expression of BST2 was analyzed by flow cytometry. Fig. 5
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry