Antibody data
- Antibody Data
- Antigen structure
- References [15]
- Comments [0]
- Validations
- Immunocytochemistry [3]
- Flow cytometry [1]
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-069A - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- COPII Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-069A detects coatomer-protein II (COPII) from rat, mouse and human cells.
- Concentration
- 1 mg/mL
Submitted references ESCRT dysfunction compromises endoplasmic reticulum maturation and autophagosome biogenesis in Drosophila.
A common human missense mutation of vesicle coat protein SEC23B leads to growth restriction and chronic pancreatitis in mice.
Neuroprogenitor Cells From Patients With TBCK Encephalopathy Suggest Deregulation of Early Secretory Vesicle Transport.
Distributed synthesis of sarcolemmal and sarcoplasmic reticulum membrane proteins in cardiac myocytes.
Imaging of type I procollagen biosynthesis in cells reveals biogenesis in highly organized bodies; Collagenosomes.
Tango1 spatially organizes ER exit sites to control ER export.
α-Synuclein-induced lysosomal dysfunction occurs through disruptions in protein trafficking in human midbrain synucleinopathy models.
A stress assembly that confers cell viability by preserving ERES components during amino-acid starvation.
Two mammalian Sec16 homologues have nonredundant functions in endoplasmic reticulum (ER) export and transitional ER organization.
On and off membrane dynamics of the endoplasmic reticulum-golgi tethering factor p115 in vivo.
Cis-Golgi matrix proteins move directly to endoplasmic reticulum exit sites by association with tubules.
Golgi inheritance in mammalian cells is mediated through endoplasmic reticulum export activities.
Biogenesis of tubular ER-to-Golgi transport intermediates.
Cholesterol is required for efficient endoplasmic reticulum-to-Golgi transport of secretory membrane proteins.
Involvement of BNIP1 in apoptosis and endoplasmic reticulum membrane fusion.
Wang R, Miao G, Shen JL, Fortier TM, Baehrecke EH
Current biology : CB 2022 Mar 28;32(6):1262-1274.e4
Current biology : CB 2022 Mar 28;32(6):1262-1274.e4
A common human missense mutation of vesicle coat protein SEC23B leads to growth restriction and chronic pancreatitis in mice.
Wei W, Liu Z, Zhang C, Khoriaty R, Zhu M, Zhang B
The Journal of biological chemistry 2022 Jan;298(1):101536
The Journal of biological chemistry 2022 Jan;298(1):101536
Neuroprogenitor Cells From Patients With TBCK Encephalopathy Suggest Deregulation of Early Secretory Vesicle Transport.
Moreira DP, Suzuki AM, Silva ALTE, Varella-Branco E, Meneghetti MCZ, Kobayashi GS, Fogo M, Ferrari MFR, Cardoso RR, Lourenço NCV, Griesi-Oliveira K, Zachi EC, Bertola DR, Weinmann KS, de Lima MA, Nader HB, Sertié AL, Passos-Bueno MR
Frontiers in cellular neuroscience 2021;15:803302
Frontiers in cellular neuroscience 2021;15:803302
Distributed synthesis of sarcolemmal and sarcoplasmic reticulum membrane proteins in cardiac myocytes.
Bogdanov V, Soltisz AM, Moise N, Sakuta G, Orengo BH, Janssen PML, Weinberg SH, Davis JP, Veeraraghavan R, Györke S
Basic research in cardiology 2021 Oct 28;116(1):63
Basic research in cardiology 2021 Oct 28;116(1):63
Imaging of type I procollagen biosynthesis in cells reveals biogenesis in highly organized bodies; Collagenosomes.
Stefanovic B, Stefanovic L, Manojlovic Z
Matrix biology plus 2021 Dec;12:100076
Matrix biology plus 2021 Dec;12:100076
Tango1 spatially organizes ER exit sites to control ER export.
Liu M, Feng Z, Ke H, Liu Y, Sun T, Dai J, Cui W, Pastor-Pareja JC
The Journal of cell biology 2017 Apr 3;216(4):1035-1049
The Journal of cell biology 2017 Apr 3;216(4):1035-1049
α-Synuclein-induced lysosomal dysfunction occurs through disruptions in protein trafficking in human midbrain synucleinopathy models.
Mazzulli JR, Zunke F, Isacson O, Studer L, Krainc D
Proceedings of the National Academy of Sciences of the United States of America 2016 Feb 16;113(7):1931-6
Proceedings of the National Academy of Sciences of the United States of America 2016 Feb 16;113(7):1931-6
A stress assembly that confers cell viability by preserving ERES components during amino-acid starvation.
Zacharogianni M, Aguilera-Gomez A, Veenendaal T, Smout J, Rabouille C
eLife 2014 Nov 11;3
eLife 2014 Nov 11;3
Two mammalian Sec16 homologues have nonredundant functions in endoplasmic reticulum (ER) export and transitional ER organization.
Bhattacharyya D, Glick BS
Molecular biology of the cell 2007 Mar;18(3):839-49
Molecular biology of the cell 2007 Mar;18(3):839-49
On and off membrane dynamics of the endoplasmic reticulum-golgi tethering factor p115 in vivo.
Brandon E, Szul T, Alvarez C, Grabski R, Benjamin R, Kawai R, Sztul E
Molecular biology of the cell 2006 Jul;17(7):2996-3008
Molecular biology of the cell 2006 Jul;17(7):2996-3008
Cis-Golgi matrix proteins move directly to endoplasmic reticulum exit sites by association with tubules.
Mardones GA, Snyder CM, Howell KE
Molecular biology of the cell 2006 Jan;17(1):525-38
Molecular biology of the cell 2006 Jan;17(1):525-38
Golgi inheritance in mammalian cells is mediated through endoplasmic reticulum export activities.
Altan-Bonnet N, Sougrat R, Liu W, Snapp EL, Ward T, Lippincott-Schwartz J
Molecular biology of the cell 2006 Feb;17(2):990-1005
Molecular biology of the cell 2006 Feb;17(2):990-1005
Biogenesis of tubular ER-to-Golgi transport intermediates.
Simpson JC, Nilsson T, Pepperkok R
Molecular biology of the cell 2006 Feb;17(2):723-37
Molecular biology of the cell 2006 Feb;17(2):723-37
Cholesterol is required for efficient endoplasmic reticulum-to-Golgi transport of secretory membrane proteins.
Ridsdale A, Denis M, Gougeon PY, Ngsee JK, Presley JF, Zha X
Molecular biology of the cell 2006 Apr;17(4):1593-605
Molecular biology of the cell 2006 Apr;17(4):1593-605
Involvement of BNIP1 in apoptosis and endoplasmic reticulum membrane fusion.
Nakajima K, Hirose H, Taniguchi M, Kurashina H, Arasaki K, Nagahama M, Tani K, Yamamoto A, Tagaya M
The EMBO journal 2004 Aug 18;23(16):3216-26
The EMBO journal 2004 Aug 18;23(16):3216-26
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
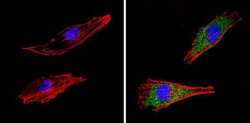
- Experimental details
- Immunofluorescent analysis of COP II (green) showing staining in the cytoplasm of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a COP II polyclonal antibody (Product # PA1-069A) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
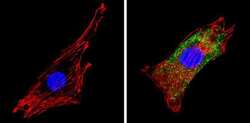
- Experimental details
- Immunofluorescent analysis of COP II (green) showing staining in the cytoplasm of NIH-3T3 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a COP II polyclonal antibody (Product # PA1-069A) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
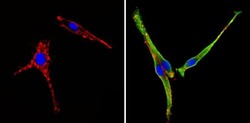
- Experimental details
- Immunofluorescent analysis of COP II (green) showing staining in the cytoplasm of PC12 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a COP II polyclonal antibody (Product # PA1-069A) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
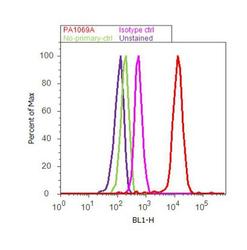
- Experimental details
- Flow cytometry analysis of COP II was done on PC-12 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with COP II Rabbit Polyclonal Antibody (PA1-069A, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10, 000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
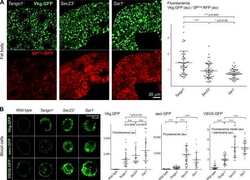
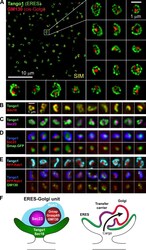
- Figure 1. Imaging of ERES-Golgi units through SIM microscopy. (A) Image of L3 fat body stained with anti-Tango1 and anti-GM130 antibodies obtained through super-resolution SIM imaging. Examples of individual ERES-Golgi units are shown at higher magnification. Images are maximum intensity projections of two to five confocal sections. (B) ERESs visualized through staining with anti-Sec16 (ERES marker) and anti-Tango1. (C) ERES visualized through staining with anti-Sec23 (COPII coat) and anti-Tango1. (D) ERES-Golgi units visualized with anti-Tango1, anti-Sec23, and Gmap.GFP (GFP-trap insertion into Gmap gene). (E) Localization of Rab1 ( Cg>RFP.Rab1 ) in relation to ERESs (anti-Tango1) and cis-Golgi (anti-GM130). (F) Organization of ERES-Golgi units as deduced from SIM images above (A-E).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
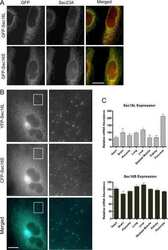
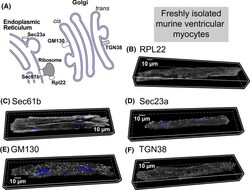
- Fig. 2 A Schematic of protein synthesis machinery. Representative 3D confocal images of adult cardiac myocytes showing B ribosomal protein Rpl22 (associated with actively translating ribosomes), C protein transport protein Sec61b, part of the translocon complex, D protein transport protein Sec23a, component of COPII, E trans-Golgi network protein TGN38, and F Cis-Golgi protein GM130. In all cases, proteins are shown in grayscale with nuclei in blue (3-9 cells per protein from 2 hearts). Quantitative analysis of these data and additional control experiments (along with n's for specific experiments) are included in Supplementary Fig. 6. Antibodies used for these studies are listed in the methods
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry