44-752G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [67]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [2]
- Other assay [35]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-752G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Ser396) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated Tau. The final product is generated by affinity chromatography using a Tau-derived peptide that is phosphorylated at serine 396.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references O-GlcNAcase Inhibitor ASN90 is a Multimodal Drug Candidate for Tau and α-Synuclein Proteinopathies.
Simple, Single-Shot Phosphoproteomic Analysis of Heat-Stable Tau Identifies Age-Related Changes in pS235- and pS396-Tau Levels in Non-human Primates.
Cornel Iridoid Glycoside Protects Against STAT1-Dependent Synapse and Memory Deficits by Increasing N-Methyl-D-aspartate Receptor Expression in a Tau Transgenic Mice.
Aberrant role of ALK in tau proteinopathy through autophagosomal dysregulation.
A new non-aggregative splicing isoform of human Tau is decreased in Alzheimer's disease.
PKR kinase directly regulates tau expression and Alzheimer's disease-related tau phosphorylation.
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
IL-17 triggers the onset of cognitive and synaptic deficits in early stages of Alzheimer's disease.
Estrogens Inhibit Amyloid-β-Mediated Paired Helical Filament-Like Conformation of Tau Through Antioxidant Activity and miRNA 218 Regulation in hTau Mice.
Neurodegeneration of Trigeminal Mesencephalic Neurons by the Tooth Loss Triggers the Progression of Alzheimer's Disease in 3×Tg-AD Model Mice.
Truncation of Tau selectively facilitates its pathological activities.
ACE2 activator diminazene aceturate ameliorates Alzheimer's disease-like neuropathology and rescues cognitive impairment in SAMP8 mice.
Brain-specific repression of AMPKα1 alleviates pathophysiology in Alzheimer's model mice.
Negative Regulator of Ubiquitin-Like Protein 1 modulates the autophagy-lysosomal pathway via p62 to facilitate the extracellular release of tau following proteasome impairment.
Mitophagy Failure in APP and Tau Overexpression Model of Alzheimer's Disease.
Memantine Differentially Regulates Tau Phosphorylation Induced by Chronic Restraint Stress of Varying Duration in Mice.
Subregional Density of Neurons, Neurofibrillary Tangles and Amyloid Plaques in the Hippocampus of Patients With Alzheimer's Disease.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
A unique tau conformation generated by an acetylation-mimic substitution modulates P301S-dependent tau pathology and hyperphosphorylation.
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Targeted degradation of aberrant tau in frontotemporal dementia patient-derived neuronal cell models.
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
A Study of Amyloid-β and Phosphotau in Plaques and Neurons in the Hippocampus of Alzheimer's Disease Patients.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
An inhibitor of the proteasomal deubiquitinating enzyme USP14 induces tau elimination in cultured neurons.
A validated antibody panel for the characterization of tau post-translational modifications.
A Dual Pathogenic Mechanism Links Tau Acetylation to Sporadic Tauopathy.
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Tau deletion promotes brain insulin resistance.
Aβ accumulation causes MVB enlargement and is modelled by dominant negative VPS4A.
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
Resveratrol Attenuates Formaldehyde Induced Hyperphosphorylation of Tau Protein and Cytotoxicity in N2a Cells.
Seed-competent high-molecular-weight tau species accumulates in the cerebrospinal fluid of Alzheimer's disease mouse model and human patients.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
The neuritic plaque facilitates pathological conversion of tau in an Alzheimer's disease mouse model.
Short-term treadmill exercise increased tau insolubility and neuroinflammation in tauopathy model mice.
Epigenetic mechanisms underlying cognitive impairment and Alzheimer disease hallmarks in 5XFAD mice.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
The choice of general anesthetics may not affect neuroinflammation and impairment of learning and memory after surgery in elderly rats.
Fyn inhibition rescues established memory and synapse loss in Alzheimer mice.
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Na, K-ATPase α3 is a death target of Alzheimer patient amyloid-β assembly.
GSK-3β-dependent downregulation of γ-taxilin and αNAC merge to regulate ER stress responses.
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
MicroRNA-922 promotes tau phosphorylation by downregulating ubiquitin carboxy-terminal hydrolase L1 (UCHL1) expression in the pathogenesis of Alzheimer's disease.
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury.
Efficacy and safety of a liposome-based vaccine against protein Tau, assessed in tau.P301L mice that model tauopathy.
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
Dietary resveratrol prevents Alzheimer's markers and increases life span in SAMP8.
Microtubule-associated protein tau in bovine retinal photoreceptor rod outer segments: comparison with brain tau.
A proteomic analysis of MCLR-induced neurotoxicity: implications for Alzheimer's disease.
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
Epigenetic changes in Alzheimer's disease: decrements in DNA methylation.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Increased tau phosphorylation and cleavage in mouse models of type 1 and type 2 diabetes.
Epigenetic differences in cortical neurons from a pair of monozygotic twins discordant for Alzheimer's disease.
Reelin signals through phosphatidylinositol 3-kinase and Akt to control cortical development and through mTor to regulate dendritic growth.
Identification of small molecules that interfere with radial neuronal migration and early cortical plate development.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Permanne B, Sand A, Ousson S, Nény M, Hantson J, Schubert R, Wiessner C, Quattropani A, Beher D
ACS chemical neuroscience 2022 Apr 20;13(8):1296-1314
ACS chemical neuroscience 2022 Apr 20;13(8):1296-1314
Simple, Single-Shot Phosphoproteomic Analysis of Heat-Stable Tau Identifies Age-Related Changes in pS235- and pS396-Tau Levels in Non-human Primates.
Leslie SN, Kanyo J, Datta D, Wilson RS, Zeiss C, Duque A, Lam TT, Arnsten AFT, Nairn AC
Frontiers in aging neuroscience 2021;13:767322
Frontiers in aging neuroscience 2021;13:767322
Cornel Iridoid Glycoside Protects Against STAT1-Dependent Synapse and Memory Deficits by Increasing N-Methyl-D-aspartate Receptor Expression in a Tau Transgenic Mice.
Ma D, Huang R, Guo K, Zhao Z, Wei W, Gu L, Li L, Zhang L
Frontiers in aging neuroscience 2021;13:671206
Frontiers in aging neuroscience 2021;13:671206
Aberrant role of ALK in tau proteinopathy through autophagosomal dysregulation.
Park J, Choi H, Kim YD, Kim SH, Kim Y, Gwon Y, Lee DY, Park SH, Heo WD, Jung YK
Molecular psychiatry 2021 Oct;26(10):5542-5556
Molecular psychiatry 2021 Oct;26(10):5542-5556
A new non-aggregative splicing isoform of human Tau is decreased in Alzheimer's disease.
García-Escudero V, Ruiz-Gabarre D, Gargini R, Pérez M, García E, Cuadros R, Hernández IH, Cabrera JR, García-Escudero R, Lucas JJ, Hernández F, Ávila J
Acta neuropathologica 2021 Jul;142(1):159-177
Acta neuropathologica 2021 Jul;142(1):159-177
PKR kinase directly regulates tau expression and Alzheimer's disease-related tau phosphorylation.
Reimer L, Betzer C, Kofoed RH, Volbracht C, Fog K, Kurhade C, Nilsson E, Överby AK, Jensen PH
Brain pathology (Zurich, Switzerland) 2021 Jan;31(1):103-119
Brain pathology (Zurich, Switzerland) 2021 Jan;31(1):103-119
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
Zhao D, Zhou Y, Huo Y, Meng J, Xiao X, Han L, Zhang X, Luo H, Can D, Sun H, Huang TY, Wang X, Zhang J, Liu FR, Xu H, Zhang YW
Cell death and differentiation 2021 Jan;28(1):337-348
Cell death and differentiation 2021 Jan;28(1):337-348
IL-17 triggers the onset of cognitive and synaptic deficits in early stages of Alzheimer's disease.
Brigas HC, Ribeiro M, Coelho JE, Gomes R, Gomez-Murcia V, Carvalho K, Faivre E, Costa-Pereira S, Darrigues J, de Almeida AA, Buée L, Dunot J, Marie H, Pousinha PA, Blum D, Silva-Santos B, Lopes LV, Ribot JC
Cell reports 2021 Aug 31;36(9):109574
Cell reports 2021 Aug 31;36(9):109574
Estrogens Inhibit Amyloid-β-Mediated Paired Helical Filament-Like Conformation of Tau Through Antioxidant Activity and miRNA 218 Regulation in hTau Mice.
Guglielmotto M, Manassero G, Vasciaveo V, Venezia M, Tabaton M, Tamagno E
Journal of Alzheimer's disease : JAD 2020;77(3):1339-1351
Journal of Alzheimer's disease : JAD 2020;77(3):1339-1351
Neurodegeneration of Trigeminal Mesencephalic Neurons by the Tooth Loss Triggers the Progression of Alzheimer's Disease in 3×Tg-AD Model Mice.
Goto T, Kuramoto E, Dhar A, Wang RP, Seki H, Iwai H, Yamanaka A, Matsumoto SE, Hara H, Michikawa M, Ohyagi Y, Leung WK, Chang RC
Journal of Alzheimer's disease : JAD 2020;76(4):1443-1459
Journal of Alzheimer's disease : JAD 2020;76(4):1443-1459
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
ACE2 activator diminazene aceturate ameliorates Alzheimer's disease-like neuropathology and rescues cognitive impairment in SAMP8 mice.
Duan R, Xue X, Zhang QQ, Wang SY, Gong PY, E Y, Jiang T, Zhang YD
Aging 2020 Jul 23;12(14):14819-14829
Aging 2020 Jul 23;12(14):14819-14829
Brain-specific repression of AMPKα1 alleviates pathophysiology in Alzheimer's model mice.
Zimmermann HR, Yang W, Kasica NP, Zhou X, Wang X, Beckelman BC, Lee J, Furdui CM, Keene CD, Ma T
The Journal of clinical investigation 2020 Jul 1;130(7):3511-3527
The Journal of clinical investigation 2020 Jul 1;130(7):3511-3527
Negative Regulator of Ubiquitin-Like Protein 1 modulates the autophagy-lysosomal pathway via p62 to facilitate the extracellular release of tau following proteasome impairment.
Guarascio R, Salih D, Yasvoina M, Edwards FA, Cheetham ME, van der Spuy J
Human molecular genetics 2020 Jan 1;29(1):80-96
Human molecular genetics 2020 Jan 1;29(1):80-96
Mitophagy Failure in APP and Tau Overexpression Model of Alzheimer's Disease.
Martín-Maestro P, Gargini R, García E, Simón D, Avila J, García-Escudero V
Journal of Alzheimer's disease : JAD 2019;70(2):525-540
Journal of Alzheimer's disease : JAD 2019;70(2):525-540
Memantine Differentially Regulates Tau Phosphorylation Induced by Chronic Restraint Stress of Varying Duration in Mice.
Liu Y, Cao L, Zhang X, Liang Y, Xu Y, Zhu C
Neural plasticity 2019;2019:4168472
Neural plasticity 2019;2019:4168472
Subregional Density of Neurons, Neurofibrillary Tangles and Amyloid Plaques in the Hippocampus of Patients With Alzheimer's Disease.
Furcila D, Domínguez-Álvaro M, DeFelipe J, Alonso-Nanclares L
Frontiers in neuroanatomy 2019;13:99
Frontiers in neuroanatomy 2019;13:99
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
A unique tau conformation generated by an acetylation-mimic substitution modulates P301S-dependent tau pathology and hyperphosphorylation.
Ajit D, Trzeciakiewicz H, Tseng JH, Wander CM, Chen Y, Ajit A, King DP, Cohen TJ
The Journal of biological chemistry 2019 Nov 8;294(45):16698-16711
The Journal of biological chemistry 2019 Nov 8;294(45):16698-16711
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Zhang Y, Wu F, Iqbal K, Gong CX, Hu W, Liu F
Scientific reports 2019 Mar 7;9(1):3789
Scientific reports 2019 Mar 7;9(1):3789
Targeted degradation of aberrant tau in frontotemporal dementia patient-derived neuronal cell models.
Silva MC, Ferguson FM, Cai Q, Donovan KA, Nandi G, Patnaik D, Zhang T, Huang HT, Lucente DE, Dickerson BC, Mitchison TJ, Fischer ES, Gray NS, Haggarty SJ
eLife 2019 Mar 25;8
eLife 2019 Mar 25;8
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Ercan-Herbst E, Ehrig J, Schöndorf DC, Behrendt A, Klaus B, Gomez Ramos B, Prat Oriol N, Weber C, Ehrnhoefer DE
Acta neuropathologica communications 2019 Dec 3;7(1):192
Acta neuropathologica communications 2019 Dec 3;7(1):192
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Hu W, Tung YC, Zhang Y, Liu F, Iqbal K
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
A Study of Amyloid-β and Phosphotau in Plaques and Neurons in the Hippocampus of Alzheimer's Disease Patients.
Furcila D, DeFelipe J, Alonso-Nanclares L
Journal of Alzheimer's disease : JAD 2018;64(2):417-435
Journal of Alzheimer's disease : JAD 2018;64(2):417-435
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
An inhibitor of the proteasomal deubiquitinating enzyme USP14 induces tau elimination in cultured neurons.
Boselli M, Lee BH, Robert J, Prado MA, Min SW, Cheng C, Silva MC, Seong C, Elsasser S, Hatle KM, Gahman TC, Gygi SP, Haggarty SJ, Gan L, King RW, Finley D
The Journal of biological chemistry 2017 Nov 24;292(47):19209-19225
The Journal of biological chemistry 2017 Nov 24;292(47):19209-19225
A validated antibody panel for the characterization of tau post-translational modifications.
Ercan E, Eid S, Weber C, Kowalski A, Bichmann M, Behrendt A, Matthes F, Krauss S, Reinhardt P, Fulle S, Ehrnhoefer DE
Molecular neurodegeneration 2017 Nov 21;12(1):87
Molecular neurodegeneration 2017 Nov 21;12(1):87
A Dual Pathogenic Mechanism Links Tau Acetylation to Sporadic Tauopathy.
Trzeciakiewicz H, Tseng JH, Wander CM, Madden V, Tripathy A, Yuan CX, Cohen TJ
Scientific reports 2017 Mar 13;7:44102
Scientific reports 2017 Mar 13;7:44102
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Chen S, Sun J, Zhao G, Guo A, Chen Y, Fu R, Deng Y
Neurochemical research 2017 Aug;42(8):2326-2335
Neurochemical research 2017 Aug;42(8):2326-2335
Tau deletion promotes brain insulin resistance.
Marciniak E, Leboucher A, Caron E, Ahmed T, Tailleux A, Dumont J, Issad T, Gerhardt E, Pagesy P, Vileno M, Bournonville C, Hamdane M, Bantubungi K, Lancel S, Demeyer D, Eddarkaoui S, Vallez E, Vieau D, Humez S, Faivre E, Grenier-Boley B, Outeiro TF, Staels B, Amouyel P, Balschun D, Buee L, Blum D
The Journal of experimental medicine 2017 Aug 7;214(8):2257-2269
The Journal of experimental medicine 2017 Aug 7;214(8):2257-2269
Aβ accumulation causes MVB enlargement and is modelled by dominant negative VPS4A.
Willén K, Edgar JR, Hasegawa T, Tanaka N, Futter CE, Gouras GK
Molecular neurodegeneration 2017 Aug 23;12(1):61
Molecular neurodegeneration 2017 Aug 23;12(1):61
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
O'Hare Doig RL, Chiha W, Giacci MK, Yates NJ, Bartlett CA, Smith NM, Hodgetts SI, Harvey AR, Fitzgerald M
BMC neuroscience 2017 Aug 14;18(1):62
BMC neuroscience 2017 Aug 14;18(1):62
Resveratrol Attenuates Formaldehyde Induced Hyperphosphorylation of Tau Protein and Cytotoxicity in N2a Cells.
He X, Li Z, Rizak JD, Wu S, Wang Z, He R, Su M, Qin D, Wang J, Hu X
Frontiers in neuroscience 2016;10:598
Frontiers in neuroscience 2016;10:598
Seed-competent high-molecular-weight tau species accumulates in the cerebrospinal fluid of Alzheimer's disease mouse model and human patients.
Takeda S, Commins C, DeVos SL, Nobuhara CK, Wegmann S, Roe AD, Costantino I, Fan Z, Nicholls SB, Sherman AE, Trisini Lipsanopoulos AT, Scherzer CR, Carlson GA, Pitstick R, Peskind ER, Raskind MA, Li G, Montine TJ, Frosch MP, Hyman BT
Annals of neurology 2016 Sep;80(3):355-67
Annals of neurology 2016 Sep;80(3):355-67
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
Manassero G, Guglielmotto M, Zamfir R, Borghi R, Colombo L, Salmona M, Perry G, Odetti P, Arancio O, Tamagno E, Tabaton M
Aging cell 2016 Oct;15(5):914-23
Aging cell 2016 Oct;15(5):914-23
The neuritic plaque facilitates pathological conversion of tau in an Alzheimer's disease mouse model.
Li T, Braunstein KE, Zhang J, Lau A, Sibener L, Deeble C, Wong PC
Nature communications 2016 Jul 4;7:12082
Nature communications 2016 Jul 4;7:12082
Short-term treadmill exercise increased tau insolubility and neuroinflammation in tauopathy model mice.
Elahi M, Motoi Y, Matsumoto SE, Hasan Z, Ishiguro K, Hattori N
Neuroscience letters 2016 Jan 1;610:207-12
Neuroscience letters 2016 Jan 1;610:207-12
Epigenetic mechanisms underlying cognitive impairment and Alzheimer disease hallmarks in 5XFAD mice.
Griñán-Ferré C, Sarroca S, Ivanova A, Puigoriol-Illamola D, Aguado F, Camins A, Sanfeliu C, Pallàs M
Aging 2016 Apr;8(4):664-84
Aging 2016 Apr;8(4):664-84
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Takeda S, Wegmann S, Cho H, DeVos SL, Commins C, Roe AD, Nicholls SB, Carlson GA, Pitstick R, Nobuhara CK, Costantino I, Frosch MP, Müller DJ, Irimia D, Hyman BT
Nature communications 2015 Oct 13;6:8490
Nature communications 2015 Oct 13;6:8490
The choice of general anesthetics may not affect neuroinflammation and impairment of learning and memory after surgery in elderly rats.
Zhang J, Tan H, Jiang W, Zuo Z
Journal of neuroimmune pharmacology : the official journal of the Society on NeuroImmune Pharmacology 2015 Mar;10(1):179-89
Journal of neuroimmune pharmacology : the official journal of the Society on NeuroImmune Pharmacology 2015 Mar;10(1):179-89
Fyn inhibition rescues established memory and synapse loss in Alzheimer mice.
Kaufman AC, Salazar SV, Haas LT, Yang J, Kostylev MA, Jeng AT, Robinson SA, Gunther EC, van Dyck CH, Nygaard HB, Strittmatter SM
Annals of neurology 2015 Jun;77(6):953-71
Annals of neurology 2015 Jun;77(6):953-71
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Wang Y, Yang R, Gu J, Yin X, Jin N, Xie S, Wang Y, Chang H, Qian W, Shi J, Iqbal K, Gong CX, Cheng C, Liu F
Neurobiology of aging 2015 Jan;36(1):188-200
Neurobiology of aging 2015 Jan;36(1):188-200
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Porquet D, Andrés-Benito P, Griñán-Ferré C, Camins A, Ferrer I, Canudas AM, Del Valle J, Pallàs M
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Na, K-ATPase α3 is a death target of Alzheimer patient amyloid-β assembly.
Ohnishi T, Yanazawa M, Sasahara T, Kitamura Y, Hiroaki H, Fukazawa Y, Kii I, Nishiyama T, Kakita A, Takeda H, Takeuchi A, Arai Y, Ito A, Komura H, Hirao H, Satomura K, Inoue M, Muramatsu S, Matsui K, Tada M, Sato M, Saijo E, Shigemitsu Y, Sakai S, Umetsu Y, Goda N, Takino N, Takahashi H, Hagiwara M, Sawasaki T, Iwasaki G, Nakamura Y, Nabeshima Y, Teplow DB, Hoshi M
Proceedings of the National Academy of Sciences of the United States of America 2015 Aug 11;112(32):E4465-74
Proceedings of the National Academy of Sciences of the United States of America 2015 Aug 11;112(32):E4465-74
GSK-3β-dependent downregulation of γ-taxilin and αNAC merge to regulate ER stress responses.
Hotokezaka Y, Katayama I, van Leyen K, Nakamura T
Cell death & disease 2015 Apr 16;6(4):e1719
Cell death & disease 2015 Apr 16;6(4):e1719
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Petry FR, Pelletier J, Bretteville A, Morin F, Calon F, Hébert SS, Whittington RA, Planel E
PloS one 2014;9(5):e94251
PloS one 2014;9(5):e94251
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
Pedrós I, Petrov D, Allgaier M, Sureda F, Barroso E, Beas-Zarate C, Auladell C, Pallàs M, Vázquez-Carrera M, Casadesús G, Folch J, Camins A
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
MicroRNA-922 promotes tau phosphorylation by downregulating ubiquitin carboxy-terminal hydrolase L1 (UCHL1) expression in the pathogenesis of Alzheimer's disease.
Zhao ZB, Wu L, Xiong R, Wang LL, Zhang B, Wang C, Li H, Liang L, Chen SD
Neuroscience 2014 Sep 5;275:232-7
Neuroscience 2014 Sep 5;275:232-7
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Maurin H, Lechat B, Borghgraef P, Devijver H, Jaworski T, Van Leuven F
The European journal of neuroscience 2014 Jul;40(2):2442-53
The European journal of neuroscience 2014 Jul;40(2):2442-53
Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury.
Iliff JJ, Chen MJ, Plog BA, Zeppenfeld DM, Soltero M, Yang L, Singh I, Deane R, Nedergaard M
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Dec 3;34(49):16180-93
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Dec 3;34(49):16180-93
Efficacy and safety of a liposome-based vaccine against protein Tau, assessed in tau.P301L mice that model tauopathy.
Theunis C, Crespo-Biel N, Gafner V, Pihlgren M, López-Deber MP, Reis P, Hickman DT, Adolfsson O, Chuard N, Ndao DM, Borghgraef P, Devijver H, Van Leuven F, Pfeifer A, Muhs A
PloS one 2013;8(8):e72301
PloS one 2013;8(8):e72301
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
Absalon S, Kochanek DM, Raghavan V, Krichevsky AM
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
Dietary resveratrol prevents Alzheimer's markers and increases life span in SAMP8.
Porquet D, Casadesús G, Bayod S, Vicente A, Canudas AM, Vilaplana J, Pelegrí C, Sanfeliu C, Camins A, Pallàs M, del Valle J
Age (Dordrecht, Netherlands) 2013 Oct;35(5):1851-65
Age (Dordrecht, Netherlands) 2013 Oct;35(5):1851-65
Microtubule-associated protein tau in bovine retinal photoreceptor rod outer segments: comparison with brain tau.
Yamazaki A, Nishizawa Y, Matsuura I, Hayashi F, Usukura J, Bondarenko VA
Biochimica et biophysica acta 2013 Oct;1832(10):1549-59
Biochimica et biophysica acta 2013 Oct;1832(10):1549-59
A proteomic analysis of MCLR-induced neurotoxicity: implications for Alzheimer's disease.
Li G, Cai F, Yan W, Li C, Wang J
Toxicological sciences : an official journal of the Society of Toxicology 2012 Jun;127(2):485-95
Toxicological sciences : an official journal of the Society of Toxicology 2012 Jun;127(2):485-95
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
Bolognin S, Blanchard J, Wang X, Basurto-Islas G, Tung YC, Kohlbrenner E, Grundke-Iqbal I, Iqbal K
Acta neuropathologica 2012 Jan;123(1):133-51
Acta neuropathologica 2012 Jan;123(1):133-51
Epigenetic changes in Alzheimer's disease: decrements in DNA methylation.
Mastroeni D, Grover A, Delvaux E, Whiteside C, Coleman PD, Rogers J
Neurobiology of aging 2010 Dec;31(12):2025-37
Neurobiology of aging 2010 Dec;31(12):2025-37
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Increased tau phosphorylation and cleavage in mouse models of type 1 and type 2 diabetes.
Kim B, Backus C, Oh S, Hayes JM, Feldman EL
Endocrinology 2009 Dec;150(12):5294-301
Endocrinology 2009 Dec;150(12):5294-301
Epigenetic differences in cortical neurons from a pair of monozygotic twins discordant for Alzheimer's disease.
Mastroeni D, McKee A, Grover A, Rogers J, Coleman PD
PloS one 2009 Aug 12;4(8):e6617
PloS one 2009 Aug 12;4(8):e6617
Reelin signals through phosphatidylinositol 3-kinase and Akt to control cortical development and through mTor to regulate dendritic growth.
Jossin Y, Goffinet AM
Molecular and cellular biology 2007 Oct;27(20):7113-24
Molecular and cellular biology 2007 Oct;27(20):7113-24
Identification of small molecules that interfere with radial neuronal migration and early cortical plate development.
Zhou L, Jossin Y, Goffinet AM
Cerebral cortex (New York, N.Y. : 1991) 2007 Jan;17(1):211-20
Cerebral cortex (New York, N.Y. : 1991) 2007 Jan;17(1):211-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Peptide Competition. Human recombinant Tau untreated (1) or treated with GSK-3ß (1 µg per µg Tau) for 45 minutes (2-5) was added to background extracts, resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature, then incubated with the Tau (pS396) antibody in a 1% BSA-TBST buffer for two hours at room temperature, following prior incubation with: no peptide (1, 2), the non-phosphopeptide corresponding to the phosphopeptide immunogen (3), a generic phosphoserine-containing peptide (4), or the phosphopeptide immunogen (5). After washing, the membrane was incubated with goat F (ab’)2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and signals were detected using the Pierce SuperSignal™ method. The data show that only the peptide corresponding to Tau (pS396) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show up-regulation of Tau (pS396) upon GSK-3ß treatment.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
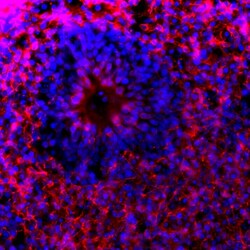
- Experimental details
- Immunofluorescent analysis of Tau pSer396 in human iPSC-derived forebrain organoids derived at Day 40. The organoids were fixed with 4% PFA for 1 hour at room temperature, followed by incubation with 30% sucrose solution overnight at 4°C. The organoids were then embedded in OCT and cryosectioned at 5 µm, permeabilized with 0.2% Triton X-100 for 20 min, and blocked with 10% donkey serum in PBS for 30 min at room temperature. Organoid slices were stained with a rabbit Tau pSer396 polyclonal antibody (red; Product # 44-752G) at a dilution of 1:500 in blocking buffer overnight at 4°C, and then incubated with Donkey anti-Rabbit Alexa Fluor 568 (Product # A10042) at a dilution of 1:1000 as well as DAPI (blue; 1:25000) in blocking solution at room temperature for 1 hour. Images were taken at 20X magnification. Data courtesy of Dr. Zhexing Wen at Emory University.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
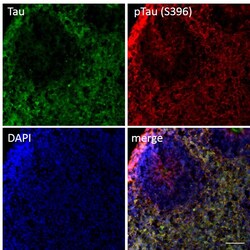
- Experimental details
- Immunofluorescent analysis of p-Tau (S396) and Tau in human iPSC-derived forebrain organoids derived at Day 40. The organoids were fixed with 4% PFA for 1 hour at room temperature, followed by incubation with 30% sucrose solution overnight at 4°C. The organoids were then embedded in OCT and cryosectioned at 5 µm, permeabilized with 0.2% Triton X-100 for 20 min, and blocked with 10% donkey serum in PBS for 30 min at room temperature. Organoid slices were stained with a Mouse Tau (TAU-5) monoclonal antibody (green; Product # AHB0042) at a dilution of 1:500 and a Rabbit p-Tau (Ser396) polyclonal antibody (red; Product # 44-752G) at a dilution of 1:500 in blocking buffer overnight at 4°C, and then incubated with Donkey anti-Mouse Alexa Fluor 488 (Product # R37114), Donkey anti-Rabbit Alexa Fluor 568 (Product # A10042) at a dilution of 1:1000 as well as DAPI (blue; 1:25000) in blocking solution at room temperature for 1 hour. Images were taken at 20X magnification. Scale bar: 50 µm. Data courtesy of Dr. Zhexing Wen at Emory University.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
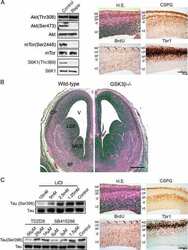
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
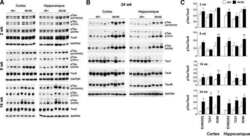
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
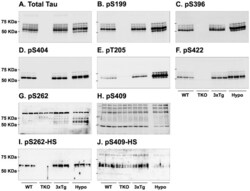
- Figure 7 Analysis of tau signal with polyclonal antibodies by Western blotting. Proteins were extracted from the cortex of 3 mouse lines: control mice (WT and Hypothermic), Tau KO mice and 3xTg-AD mice. Proteins were separated by SDS-PAGE and then identified with the following polyclonal antibodies: A: Total Tau, B: pS199, C: pS396, D: pS404, E: pT205, F: pS422, G: pS262 and H: pS409. Normal anti-rabbit secondary antibodies were used to detect primary antibodies. The heat stable fraction was used to remove non-specificity: I: pS262 and J: pS409. Quantifications of the blots are available in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
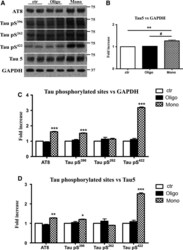
- Figure 3 Abeta1-42 monomers promote phosphorylation at particular sites that have been related to Alzheimer's disease (AD) progression. Representative Western blot of brain extracts (20 mug protein) from control (saline) and treated (Abeta1-42 peptides by ICV for 3 h) mice using antibodies specific for the detection four pathological Tau phosphorylation sites: AT8, pS396, pS262, and pS422. An antibody raised against GAPDH or Tau 5 served as loading control. Densitometric quantification shows an increase of the total protein level of AT8, pS396, and pS422 induced by monomers while monomeric and oligomeric preparations did not change pS262 expression. The data are mean +- standard error of the mean (SEM), * P < 0.05; *** P < 0.001 vs. control by one-way ANOVA followed by Bonferroni post hoc test, n = 6.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
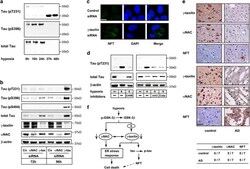
- Figure 7 Hypoxia or gamma -taxilin ablation induces tau hyperphosphorylation and cleavage in neuronal cells. ( a ) Western blot shows hyperphosphorylation at T231 (upper panel) and S396 (middle panel), and tau protein (~55 kDa) (lower panel) in SK-N-SH neuronal cells cultured under hypoxic conditions for indicated durations. Note that cleaved (~35 kDa) tau contains phosphorylated T231, but not phosphorylated S396. ( b ) Western blot shows hyperphosphorylation of tau protein (T231, S396, and S404) in SH-SY5Y neuronal cells cultured in the presence of gamma -taxilin-specific small interfering RNA (siRNA), but not in alpha NAC siRNA-treated cells. Note that total tau increases in amount after gamma -taxilin-specific siRNA addtion. ( c ) Confocal microscopy shows tau aggregations (NFT) in the cytoplasm of gamma -taxilin-specific siRNA-treated SH-SY5Y cells. Scale bar, 10 mu m. ( d ) Tau cleavage is calpain-dependent. Western blot analysis shows that calpain inhibitors (inh3, calpain inhibitor III; or calp, calpeptin) inhibit tau cleavage, but not tau hyperphosphorylation in SK-N-SH neuronal cells (right panels). As expected, GSK-3 beta inhibition with CHIR inhibits tau hyperphosphorylation in the neuronal cells. ( e ) Downregulation of gamma -taxilin and alpha NAC in the brains of patients with AD. Immunohistochemistry shows gamma -taxilin and alpha NAC protein expression in the healthy brain (control, left panels), but not in the brains from patients with AD (right panels). Uppe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
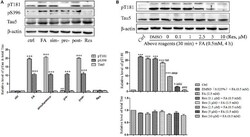
- Figure 4 Res attenuates FA-induced tau protein hyperphosphorylation in N2a cells. (A) N2a cells were pre-treated or post-treated with 10 muM of Res for 30 min before or after 0.5 mM FA exposure for 4 h, or treated simultaneously with 0.5 mM FA for 4 h. (B) N2a cells were pre-treated with Res (0.1, 1, 2.5, 5, and 10 muM) for 30 min, and then treated with 0.5 mM FA for 4 h. Total and phosphorylated tau protein levels were determined by Western blotting (A,B) . Densitometry values were calculated from independent experiments performed in triplicate ( n = 3) and presented as mean +- SEM. * p < 0.05, *** p < 0.001 vs. control. ## p < 0.01, ### p < 0.001 vs. FA treatment group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
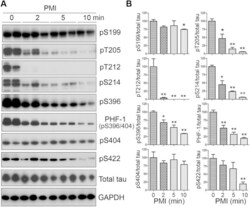
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
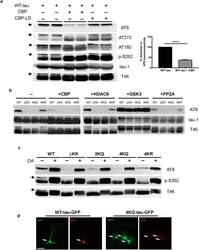
- Figure 1 Tau acetylation modulates tau phosphorylation at specific epitopes. (a-c ) Immunoblot analysis and quantification using the indicated tau antibodies for (a ) cells expressing WT tau-T40 and either an active form of CBP or a catalytically inactive form of CBP (CBP-LD), ( b ) cells co- expressing tau-T40 (WT, DeltaKK, 4KQ, or 4KR) and tau modifying enzymes, and ( c ) cells expressing tau-T40 (WT, DeltaKK, 2KQ, 4KQ, or 4KR) and treated with control (DMSO) or okadaic acid (OA), where indicated. Solid black arrows highlight the ~75 kDa phospho-tau species that is reduced upon acetylation. Statistical significance was assessed using a student t-test (****p < 0.0001). Cropped images from full size immunoblots are provided in panels a-c. Full-length immunoblots are presented in Supplementary Fig. S1 . ( d ) Immunofluorescence microscopy of primary cortical neurons expressing WT-tau-GFP (left) or 4KQ-tau-GFP (right) detecting phosphorylated tau (AT8). White arrows identify transfected neurons. Scale bar, 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
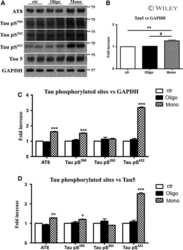
- FIGURE 4 Cornel iridoid glycoside (CIG) inhibited tau phosphorylation and accumulation in the brain of P301S transgenic mice. (A) Representative images of immunohistochemical staining of AT8 in the cerebral cortex, and hippocampus CA1, CA3, and DG areas of mice. Scale bar = 50 mum. Quantitative analysis of integrated optical density (IOD) of AT8 in the cerebral cortex (B) and hippocampus CA1 (C) , CA3 (D) , and DG (E) areas of mice. (F) Representative images of Western blotting for tau5 (total tau), T22 (tau oligomers), monomer (low exposure), and oligomers (high exposure) of phosphorylated (p)-tau at Ser396 (pS396) in the brain of mice. Quantification of Western blotting for tau5 (G) , T22 (H) , pS396-tau oligomers [140~170 kDa, (I) ], and monomer [~55 kDa, (J) ]. Quantification of blots after normalization with GAPDH, and the numerical value of nTg group was taken as 1. Data are expressed as mean +- SEM; n = 3 per group for immunohistochemical staining; n = 4 per group for western blotting. ## P < 0.01, ### P < 0.001, P301S transgenic mice vs nTg control mice; * P < 0.05, ** P < 0.01, drug-treated P301S mice vs saline-treated P301S mice.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
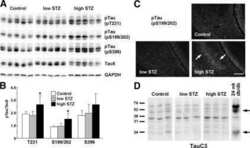
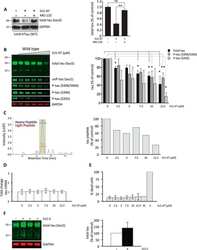
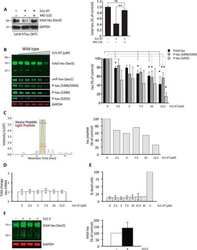
- FIGURE 3 Age-related changes in monkey S235 and S396 validate mass spectrometry results. The 19.5-year-old animal denoted as ""low"" in the proteomics study and western blots is indicated by a gray square in the graphs. (A) Bar graph summarizing a means comparison of pS235-tau between young (light blue, N = 3) and aged (dark blue, N = 11) macaques. Individual data points are plotted for each group. Due to a lack of normal distribution of pS235-tau with age the means were compared using a Mann-Whitney test (* p = 0.0385). (B) Quantification of pS235-tau normalized by GAPDH is plotted by age. Samples were not normally distributed so the correlation coefficient was computed using a Spearman correlation ( r = 0.5809, * p = 0.0319) but a standard linear regression is represented on the graph. (C) Immunohistochemistry revealed robust immunolabeling for pS235-tau in aged macaque dlPFC layer III and layer V/VI, particularly in pyramidal neurons along apical dendrites and in the cell body. Scale bar, 50 mum. (D) A bar graph summarizing a means comparison of pS396-tau between young (light pink, N = 3) and aged (red, N = 11) macaques. Individual data points are plotted for each group. Given the normal distribution of pS235-tau with age but unequal variance, the means were compared using a Welch's t -test (** p = 0.0013). (E) Quantification of pS396-tau normalized by GAPDH is plotted by age. Samples were normally distributed so the correlation coefficient was computed using a Pearson cor
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
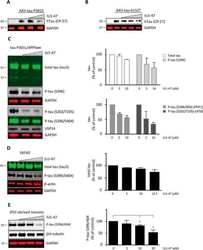
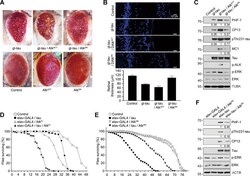
- Fig. 4 ALK regulates tau-mediated neurodegeneration in tauopathy fly models. A Enhancement of the rough eye phenotype and disordered ommatidial morphology in tau transgenic flies expressing active Drosophila Alk (Alk ACT ). External phenotypes of fly eyes were examined under a microscope. Control and experimental genotypes: Control (GMR-GAL4/+), Alk ACT (GMR-GAL4/+; UAS- Alk ACT /+), Alk DN (GMR-GAL4/+; UAS- Alk DN /+), tau (GMR-GAL4/+; gl-tau2.1/+), tau + Alk ACT (GMR-GAL4/+; gl-tau2.1/+; UAS- Alk ACT /+) and tau + Alk DN (GMR-GAL4/+; gl-tau2.1/+; UAS- Alk DN /+). B Exacerbation of retinal degeneration by ALK in tau flies. Internal retinal sections were stained with Hoechst 33342, after which they were observed under a fluorescence microscope (upper). Retinal thickness was quantified ( n = 3) (lower). C Increased tau phosphorylation in tau- and Alk ACT -expressing flies. Heads of the flies expressing tau alone or together with an Alk mutant were examined by western blotting. The signals of total tau and phosphorylated tau on the blots were quantified by densitometric analysis and the values are noted under the blot. D Shortened lifespan of transgenic flies expressing tau and Alk FL in neurons. At least 200 flies of each genotype were collected and assayed for longevity. Control and experimental genotypes: Control (elav-GAL4/+), tau (elav-GAL4/+; UAS-tau), Alk FL (elav-GAL4/+; UAS- Alk FL ) and tau + Alk FL (elav-GAL4/+; UAS-tau; UAS- Alk FL ). E Extended lifespan of transgen
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 ALK is upregulated in AD brains and pharmacological inhibition of ALK rescues memory impairment in 3xTg-AD mice. A Increased expression of ALK and accumulation of p62 in AD brains. Hippocampal lysates from normal elderly controls and patients with AD were analyzed by western blotting (left). Total ALK levels were quantified and normalized to beta-actin. Bars depict mean +- SEM; unpaired two-tailed Student's t test (right). B Colocalization of ALK and phosphorylated tau within the cortex of patients with AD. C-E LDK378 rescues memory in 3xTg-AD mice. Six-month-old 3xTg-AD mice were intraperitoneally injected with 5 mg/kg LDK378 or DMSO (Vehicle) ( n = 7-9) daily for 4 weeks and then analyzed using Y-maze ( C P = 0.0027), novel object recognition ( D P = 0.0046) and passive avoidance ( E ) tests. F-H LDK378 reduces tau phosphorylation and accumulation in the brains of 3xTg-AD mice. Hippocampal extracts from the mice analyzed in ( C - E ) were examined by western blotting ( F ). Levels of total tau (TG5), exogenous human tau (HT7), phosphorylated tau (PHF-1, CP13), p-ERK, and ERK on the blots were quantified and normalized to beta-actin. Bars depict mean +- SEM. PHF-1, P = 0.0059; CP13, P = 0.0319; HT7, P < 0.0001; one-way ANOVA followed by Tukey's test ( G ). The CA1 region in the hippocampus of 8-month-old 3xTg-AD/Vehicle and 3xTg-AD/LDK378 mice were immunostained with anti-MAP2B and AT100 antibodies (Representative confocal images, 200x, Scale bar, 50 mum) ( H ). I The
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
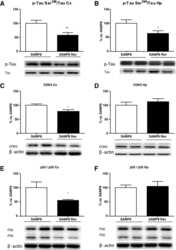
- Fig. 6 Immunoblotting using hTg-Tau and Tau-KO brain lysates confirms the specificity for tau for a panel of antibodies. a ) Scheme of tau (2N4R) showing the epitopes of the Tau12, Tau5, Tau1, HT7, BT2 and Dako antibodies. b ) - d' ) Immunoblots demonstrate strong reactivity of all antibodies with tau bands between 40 and 60 kD. Minor non-specific bands present in Tau-KO brain lysates were detected in d ), e ), i ), h ), p ), x ), y ) and c '). Isoform-specific antibodies in h ), i ), j ) and k ) were validated using a tau ladder with all 6 recombinant human tau isoforms. kDa sizes for the marker are given in e' ). Tg: hTg-Tau mouse, KO: Tau-KO mouse brain lysate, TL: recombinant tau ladder
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
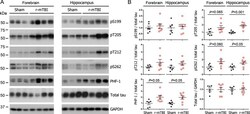
- Fig. 1 Tau phosphorylation increases at multiple sites in the brain following r-mTBI in 3xTg-AD mice. Adult female 3xTg-AD mice, 4-- months of age, were subjected to repetitive mild closed-head brain impacts, totaling five with 48-h inter-impact intervals, and sacrificed 24 h after the final impact. A) Representative western blots showing the level of indicated proteins/phosphorylation sites. B) Densitometric quantification of the blots. Among the phosphorylation sites examined, tau was hyperphosphorylated at pThr 205 , pSer 262 and PHF-1 (pSer 396/404 ) sites. Data are presented as scatter dot plots with mean+-SEM ( n = 6-7 mice each) and analyzed using unpaired t test, with Welch's correction in the case of unequal variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
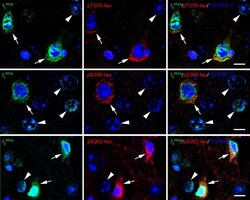
- Fig. 5 Translocated I 2 PP2A /SET is colocalized with hyperphosphorylated tau in the neuronal cytoplasm in cerebral cortex of 3xTg-AD mice with r-mTBI. Brain sections were fluorescently dual immuno-stained for I 2 PP2A and phospho-tau. Translocated I 2 PP2A colocalized with pThr 205 -tau, pSer 396 -tau and with pSer 262 -tau in the cytoplasm of neurons (arrows). However, there was no somatodendritic phospho-tau staining in neurons where cytoplasmic I 2 PP2A was absent (filled arrowheads). Scale bar = 10 mu m for all images.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Compared to Braak 0-I samples, many but not all, tau phosphorylation events are increased in native Braak III-IV samples. Normalized phospho-tau signals obtained from ELISA measurements of samples from a ) entorhinal cortices (EC), b ) hippocampi (Hip) and c ) temporal cortices (TC). Student's t-tests: *, p < 0.05, **, p < 0.01, ***, p < 0.001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
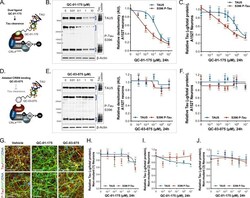
- Experimental details
- Figure 2. Concentration effect of QC-01-175 ( A ) on tau protein levels of A152T and control neurons. Analysis of total tau (TAU5) and phospho-tau (S396 P-tau) levels upon treatment by western blot ( B ) and ELISA ( C ). Analysis of total tau (TAU5) and phospho-tau (S396 P-tau) levels upon treatment with the negative control QC-03-075 ( D ), by western blot ( E ) and ELISA ( F ). Representative western blots are shown ( B, E ) with mean densitometry quantification (bands corresponding to brackets)+-SEM ( n = 3). ( C, F ) For ELISA, data points represent mean tau levels (mug of total protein) normalized to vehicle-treated +- SEM ( n = 4). Both assays show QC-01-175 dose-dependent effect on tau levels, with QC-03-075 minimal effect (~10%). ( G ) IF of A152T neurons treated with vehicle or 10 muM compound, immuno-probed for total tau (K9JA, red), P-tau (PHF-1, red) and the neuronal marker MAP2 (green), scale bar 50 mum. ( H-J ) Tau ELISA of control neurons treated with QC-01-175, which did not show a dose-dependent effect. ( H ) 8330-8-RC1 line; ( I ) MGH2069-RC1 line; ( J ) CTR2-L17-RC2 line. Data points represent mean tau levels (mug of total protein) normalized to vehicle-treated +- SEM ( n = 3). All neurons were differentiated for 6 weeks and treated with compound for 24 hr. Student T-test between each concentration and vehicle-treated tau levels ns p> 0.05, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
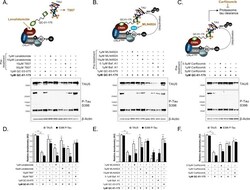
- Experimental details
- Figure 3. Mechanism of QC-01-175 clearance of tau is CRL4 CRBN and UPS-dependent. Neurons were pre-treated for 6 hr with ( A ) either CRBN ligand excess lenalidomide or tau ligand excess T807, ( B ) the NAE inhibitor MLN4924, the autophagy inhibitor Baf.A1, or ( C ) the proteasome inhibitor carfilzomib; followed by 18 hr treatment with QC-01-175 (or negative control QC-03-075), for a total of 24 hr. Total (TAU5) and P-tau S396 levels were analyzed by western blotting. ( A-C ) Representative blots are shown. ( D-F ) Densitometry bars represent tau mean intensity values +- SD ( n = 3), relative to vehicle-treated samples. Student T-test of QC-01-175 samples relative to vehicle treated, and the remainder bars show p-value of each pre-treatment relative to QC-01-175 to assess rescue of clearance effect (***p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
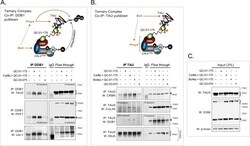
- Experimental details
- Figure 4. Demonstration of ternary complex formation in A152T neurons upon QC-01-175 treatment, by co-IP and western blot analysis. Neurons (6-week differentiated) were treated for 4 hr with 1 muM QC-01-175 +- 30 min pre-treatment with proteasome inhibitors (carfilzomib or bortezomib at 5 muM), with the goal of capturing maximum molecular interactions at 4 hr and halting tau clearance. QC-03-075 is a negative control for CRBN binding. ( A ) Co-IP by DDB1 pulldown and detection of tau in the complex by probing for total tau (TAU5), P-tau S396/S404 (PHF-1), and ubiquitinated proteins (Ubi-1). ( B ) Co-IP by tau pulldown (TAU5) and detection of CRL4 CRBN subunits CRBN, CUL4A and DDB1. Western blot of total tau (K9JA) was used as a control. ( C ) Control western blot analysis with 3% (10 mug) of IP input confirms the effect of QC-01-175 +- proteasome inhibitors on tau and P-tau S396. Red arrows and brackets indicate the predicted bands for each immunoprobed protein ( n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
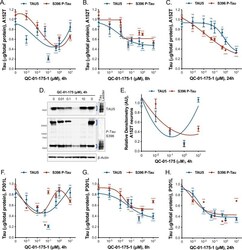
- Experimental details
- Figure 5--figure supplement 1. Degrader concentration and time effect on tau, in A152T and P301L neurons. ( A-C ) Concentration and time effect of QC-01-175 on total tau (TAU5) and S396 P-tau levels, in A152T 6-week differentiated neurons, treated for ( A ) a short 4 hr interval, ( B ) an intermediate 8 hr interval, ( C ) compared to 24 hr treatment. ( D ) Concentration effect of QC-01-175 4 hr treatment in A152T neurons, by western blot analysis of total tau (TAU5) and P-tau S396 (representative blot is shown) with mean densitometry quantification (bands corresponding to brackets)+-SEM ( n = 3). ( F-H ) Concentration and time effect of QC-01-175 on total tau (TAU5) and S396 P-tau levels, in P301L 6-week differentiated neurons treated for ( F ) 4 hr, ( G ) 8 hr and ( H ) 24 hr. ( A-C, F-H ) Data points represent mean levels of tau protein measured by ELISA, normalized to total mug of protein and to vehicle-treated +- SEM ( n = 3). Student T-test between each dose and vehicle-treated tau levels ns p>0.05, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
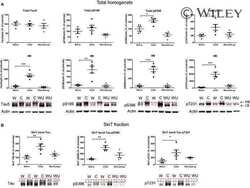
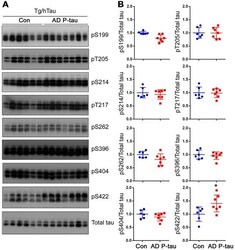
- Figure 3 Tau phosphorylation in the ipsilateral cortex in Tg/hTau mice injected with AD P-tau. Phosphorylation of tau at Ser199, Thr205, Ser214, Thr217, Ser262, Ser396, Ser404 and Ser422 in the ipsilateral cortex was analyzed by Western blots (A) and analyzed with unpaired student t -test. Data are presented as scattered dots with mean +- SD (B) ; * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
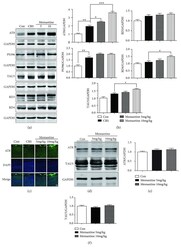
- Figure 5 Memantine enhances tau phosphorylation after 28-day chronic restraint stress (CRS) in the mouse frontal cortex. (a) Results of Western blotting showed that memantine enhances tau phosphorylation at the AT8 site for both doses. Furthermore, the expression of the 3R tau isoform (RD3), 4R tau isoform (RD4), P-tau-PS396, and TAU5 was also detected among the four groups of mice. (b) Densitometric quantification of the bands in (a). (c) Representative areas of AT8-positive immunofluorescent staining in cortices from the four groups of mice. (d) Western blotting analyzed the effects of memantine administration with doses of 5 and 10 mg/kg for 28 days on tau phosphorylation and expression in frontal cortex of mice without CRS. (e, f) Densitometric quantification of the bands in (d). The results are shown as means +- SEM ( n = 4 in each group). GAPDH was used as a loading control. * p < 0.05; ** p < 0.01; *** p < 0.001. Con: control.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot