44-764G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [23]
- Comments [0]
- Validations
- Western blot [2]
- Other assay [17]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-764G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Ser422) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Microglia phenotypes are associated with subregional patterns of concomitant tau, amyloid-β and α-synuclein pathologies in the hippocampus of patients with Alzheimer's disease and dementia with Lewy bodies.
Alzheimer's disease brain-derived extracellular vesicles spread tau pathology in interneurons.
Early detection of Alzheimer's disease using creatine chemical exchange saturation transfer magnetic resonance imaging.
Mixed pathologies in pancreatic β cells from subjects with neurodegenerative diseases and their interaction with prion protein.
Estrogens Inhibit Amyloid-β-Mediated Paired Helical Filament-Like Conformation of Tau Through Antioxidant Activity and miRNA 218 Regulation in hTau Mice.
Amylin as a potential link between type 2 diabetes and alzheimer disease.
Pretangle pathology within cholinergic nucleus basalis neurons coincides with neurotrophic and neurotransmitter receptor gene dysregulation during the progression of Alzheimer's disease.
Rab7A regulates tau secretion.
Functional screening of Alzheimer risk loci identifies PTK2B as an in vivo modulator and early marker of Tau pathology.
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
The neuritic plaque facilitates pathological conversion of tau in an Alzheimer's disease mouse model.
High Resolution Dissection of Reactive Glial Nets in Alzheimer's Disease.
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Reelin immunoreactivity in neuritic varicosities in the human hippocampal formation of non-demented subjects and Alzheimer's disease patients.
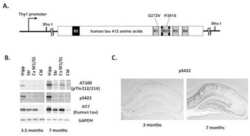
Targeting phospho-Ser422 by active Tau Immunotherapy in the THYTau22 mouse model: a suitable therapeutic approach.
Targeting phospho-Ser422 by active Tau Immunotherapy in the THYTau22 mouse model: a suitable therapeutic approach.
Methylthioninium chloride (methylene blue) induces autophagy and attenuates tauopathy in vitro and in vivo.
Nature of "Tau" immunoreactivity in normal myonuclei and inclusion body myositis.
Increased tau phosphorylation and cleavage in mouse models of type 1 and type 2 diabetes.
Increased tau phosphorylation on mitogen-activated protein kinase consensus sites and cognitive decline in transgenic models for Alzheimer's disease and FTDP-17: evidence for distinct molecular processes underlying tau abnormalities.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Formation of neurofibrillary tangles in P301l tau transgenic mice induced by Abeta 42 fibrils.
Fixemer S, Ameli C, Hammer G, Salamanca L, Uriarte Huarte O, Schwartz C, Gérardy JJ, Mechawar N, Skupin A, Mittelbronn M, Bouvier DS
Acta neuropathologica communications 2022 Mar 16;10(1):36
Acta neuropathologica communications 2022 Mar 16;10(1):36
Alzheimer's disease brain-derived extracellular vesicles spread tau pathology in interneurons.
Ruan Z, Pathak D, Venkatesan Kalavai S, Yoshii-Kitahara A, Muraoka S, Bhatt N, Takamatsu-Yukawa K, Hu J, Wang Y, Hersh S, Ericsson M, Gorantla S, Gendelman HE, Kayed R, Ikezu S, Luebke JI, Ikezu T
Brain : a journal of neurology 2021 Feb 12;144(1):288-309
Brain : a journal of neurology 2021 Feb 12;144(1):288-309
Early detection of Alzheimer's disease using creatine chemical exchange saturation transfer magnetic resonance imaging.
Chen L, van Zijl PCM, Wei Z, Lu H, Duan W, Wong PC, Li T, Xu J
NeuroImage 2021 Aug 1;236:118071
NeuroImage 2021 Aug 1;236:118071
Mixed pathologies in pancreatic β cells from subjects with neurodegenerative diseases and their interaction with prion protein.
Martinez-Valbuena I, Valenti-Azcarate R, Amat-Villegas I, Marcilla I, Marti-Andres G, Caballero MC, Riverol M, Tuñon MT, Fraser PE, Luquin MR
Acta neuropathologica communications 2021 Apr 8;9(1):64
Acta neuropathologica communications 2021 Apr 8;9(1):64
Estrogens Inhibit Amyloid-β-Mediated Paired Helical Filament-Like Conformation of Tau Through Antioxidant Activity and miRNA 218 Regulation in hTau Mice.
Guglielmotto M, Manassero G, Vasciaveo V, Venezia M, Tabaton M, Tamagno E
Journal of Alzheimer's disease : JAD 2020;77(3):1339-1351
Journal of Alzheimer's disease : JAD 2020;77(3):1339-1351
Amylin as a potential link between type 2 diabetes and alzheimer disease.
Martinez-Valbuena I, Valenti-Azcarate R, Amat-Villegas I, Riverol M, Marcilla I, de Andrea CE, Sánchez-Arias JA, Del Mar Carmona-Abellan M, Marti G, Erro ME, Martínez-Vila E, Tuñon MT, Luquin MR
Annals of neurology 2019 Oct;86(4):539-551
Annals of neurology 2019 Oct;86(4):539-551
Pretangle pathology within cholinergic nucleus basalis neurons coincides with neurotrophic and neurotransmitter receptor gene dysregulation during the progression of Alzheimer's disease.
Tiernan CT, Ginsberg SD, He B, Ward SM, Guillozet-Bongaarts AL, Kanaan NM, Mufson EJ, Counts SE
Neurobiology of disease 2018 Sep;117:125-136
Neurobiology of disease 2018 Sep;117:125-136
Rab7A regulates tau secretion.
Rodriguez L, Mohamed NV, Desjardins A, Lippé R, Fon EA, Leclerc N
Journal of neurochemistry 2017 May;141(4):592-605
Journal of neurochemistry 2017 May;141(4):592-605
Functional screening of Alzheimer risk loci identifies PTK2B as an in vivo modulator and early marker of Tau pathology.
Dourlen P, Fernandez-Gomez FJ, Dupont C, Grenier-Boley B, Bellenguez C, Obriot H, Caillierez R, Sottejeau Y, Chapuis J, Bretteville A, Abdelfettah F, Delay C, Malmanche N, Soininen H, Hiltunen M, Galas MC, Amouyel P, Sergeant N, Buée L, Lambert JC, Dermaut B
Molecular psychiatry 2017 Jun;22(6):874-883
Molecular psychiatry 2017 Jun;22(6):874-883
Beta-amyloid 1-42 monomers, but not oligomers, produce PHF-like conformation of Tau protein.
Manassero G, Guglielmotto M, Zamfir R, Borghi R, Colombo L, Salmona M, Perry G, Odetti P, Arancio O, Tamagno E, Tabaton M
Aging cell 2016 Oct;15(5):914-23
Aging cell 2016 Oct;15(5):914-23
The neuritic plaque facilitates pathological conversion of tau in an Alzheimer's disease mouse model.
Li T, Braunstein KE, Zhang J, Lau A, Sibener L, Deeble C, Wong PC
Nature communications 2016 Jul 4;7:12082
Nature communications 2016 Jul 4;7:12082
High Resolution Dissection of Reactive Glial Nets in Alzheimer's Disease.
Bouvier DS, Jones EV, Quesseveur G, Davoli MA, A Ferreira T, Quirion R, Mechawar N, Murai KK
Scientific reports 2016 Apr 19;6:24544
Scientific reports 2016 Apr 19;6:24544
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Takeda S, Wegmann S, Cho H, DeVos SL, Commins C, Roe AD, Nicholls SB, Carlson GA, Pitstick R, Nobuhara CK, Costantino I, Frosch MP, Müller DJ, Irimia D, Hyman BT
Nature communications 2015 Oct 13;6:8490
Nature communications 2015 Oct 13;6:8490
Reelin immunoreactivity in neuritic varicosities in the human hippocampal formation of non-demented subjects and Alzheimer's disease patients.
Notter T, Knuesel I
Acta neuropathologica communications 2013 Jun 26;1:27
Acta neuropathologica communications 2013 Jun 26;1:27
Targeting phospho-Ser422 by active Tau Immunotherapy in the THYTau22 mouse model: a suitable therapeutic approach.
Troquier L, Caillierez R, Burnouf S, Fernandez-Gomez FJ, Grosjean ME, Zommer N, Sergeant N, Schraen-Maschke S, Blum D, Buee L
Current Alzheimer research 2012 May;9(4):397-405
Current Alzheimer research 2012 May;9(4):397-405
Targeting phospho-Ser422 by active Tau Immunotherapy in the THYTau22 mouse model: a suitable therapeutic approach.
Troquier L, Caillierez R, Burnouf S, Fernandez-Gomez FJ, Grosjean ME, Zommer N, Sergeant N, Schraen-Maschke S, Blum D, Buee L
Current Alzheimer research 2012 May;9(4):397-405
Current Alzheimer research 2012 May;9(4):397-405
Methylthioninium chloride (methylene blue) induces autophagy and attenuates tauopathy in vitro and in vivo.
Congdon EE, Wu JW, Myeku N, Figueroa YH, Herman M, Marinec PS, Gestwicki JE, Dickey CA, Yu WH, Duff KE
Autophagy 2012 Apr;8(4):609-22
Autophagy 2012 Apr;8(4):609-22
Nature of "Tau" immunoreactivity in normal myonuclei and inclusion body myositis.
Salajegheh M, Pinkus JL, Nazareno R, Amato AA, Parker KC, Greenberg SA
Muscle & nerve 2009 Oct;40(4):520-8
Muscle & nerve 2009 Oct;40(4):520-8
Increased tau phosphorylation and cleavage in mouse models of type 1 and type 2 diabetes.
Kim B, Backus C, Oh S, Hayes JM, Feldman EL
Endocrinology 2009 Dec;150(12):5294-301
Endocrinology 2009 Dec;150(12):5294-301
Increased tau phosphorylation on mitogen-activated protein kinase consensus sites and cognitive decline in transgenic models for Alzheimer's disease and FTDP-17: evidence for distinct molecular processes underlying tau abnormalities.
Lambourne SL, Sellers LA, Bush TG, Choudhury SK, Emson PC, Suh YH, Wilkinson LS
Molecular and cellular biology 2005 Jan;25(1):278-93
Molecular and cellular biology 2005 Jan;25(1):278-93
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Formation of neurofibrillary tangles in P301l tau transgenic mice induced by Abeta 42 fibrils.
Götz J, Chen F, van Dorpe J, Nitsch RM
Science (New York, N.Y.) 2001 Aug 24;293(5534):1491-5
Science (New York, N.Y.) 2001 Aug 24;293(5534):1491-5
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
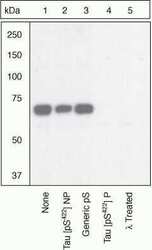
- Up-regulation and Antibody-Peptide Competition. Human recombinant Tau stimulated with GSK-3β (1 µg per µg Tau) for 45 minutes was added to background extracts, resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature and either left untreated (1-4) or treated with lambda phosphatase (5), then incubated with the Tau (pS422) antibody in a 3% BSA-TBST buffer for two hours at room temperature, following prior incubation with: no peptide (1, 5), the non-phosphopeptide corresponding to the phosphopeptide immunogen (2), a generic phosphoserine-containing peptide (3), or the phosphopeptide immunogen (4). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and signals were detected using the Pierce SuperSignal™ method.The data show that only the phosphopeptide corresponding to Tau (pS422) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show that phosphatase stripping eliminates the signal, verifying that the antibody is phospho-specific. (Product # 44-764G)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Up-regulation and Antibody-Peptide Competition. Human recombinant Tau stimulated with GSK-3β (1 µg per µg Tau) for 45 minutes was added to background extracts, resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature and either left untreated (1-4) or treated with lambda phosphatase (5), then incubated with the Tau (pS422) antibody in a 3% BSA-TBST buffer for two hours at room temperature, following prior incubation with: no peptide (1, 5), the non-phosphopeptide corresponding to the phosphopeptide immunogen (2), a generic phosphoserine-containing peptide (3), or the phosphopeptide immunogen (4). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and signals were detected using the Pierce SuperSignal™ method.The data show that only the phosphopeptide corresponding to Tau (pS422) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show that phosphatase stripping eliminates the signal, verifying that the antibody is phospho-specific. (Product # 44-764G)
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
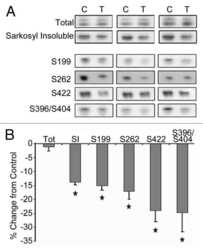
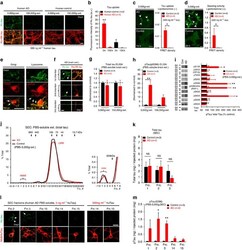
- Figure 6 Neuronal uptake of PBS-soluble HMW tau derived from human AD brain. ( a , b ) Primary neurons were incubated with AD or control brain extracts (cases were matched for age and postmortem interval ( Supplementary Table S1 )) and immunostained at day 2 ( a ). ( b ) Quantification of fluorescence intensity of human tau staining. One-way ANOVA and a subsequent Tukey-Kramer test. ( c , d ) Tau uptake ( c ) and seeding activity ( d ) assay in HEK-tau-biosensor cells. (Mann-Whitney U -test) ( e ) Subcellular localization of human tau taken up by neurons (PBS-3,000 g , 500 ng ml -1 human tau). ( f ) Neuron-to-neuron transfer of tau in a 3-chamber microfluidic device. AD brain extract (PBS-3,000 g , 500 ng ml -1 human tau) was added to the 1st chamber. Human tau positive neurons were detected in both the 1st and 2nd chamber at day 7 (arrow). ( g , h ) Quantification of total-tau ( g ) and phospho-tau ( h ) levels in AD and control brain extract (ELISA). Unpaired t -test. ( i ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. Unpaired t -test. ( j , k ) SEC analysis of PBS-soluble tau from AD and control brain. ( j ) Representative graph of total tau levels (ELISA) in SEC-separated samples. Small peaks for HMW fractions were detected in both groups (right panel). ( k ) Mean total tau levels of HMW SEC fractions. ( l ) Tau uptake from each SEC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
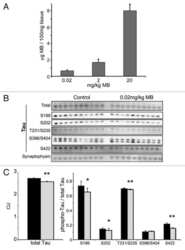
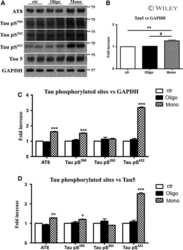
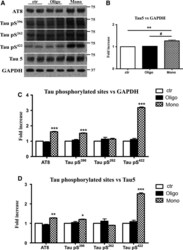
- Figure 3 Abeta1-42 monomers promote phosphorylation at particular sites that have been related to Alzheimer's disease (AD) progression. Representative Western blot of brain extracts (20 mug protein) from control (saline) and treated (Abeta1-42 peptides by ICV for 3 h) mice using antibodies specific for the detection four pathological Tau phosphorylation sites: AT8, pS396, pS262, and pS422. An antibody raised against GAPDH or Tau 5 served as loading control. Densitometric quantification shows an increase of the total protein level of AT8, pS396, and pS422 induced by monomers while monomeric and oligomeric preparations did not change pS262 expression. The data are mean +- standard error of the mean (SEM), * P < 0.05; *** P < 0.001 vs. control by one-way ANOVA followed by Bonferroni post hoc test, n = 6.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
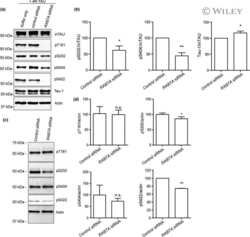
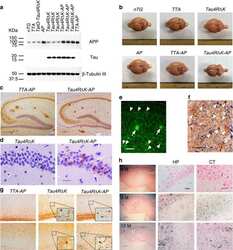
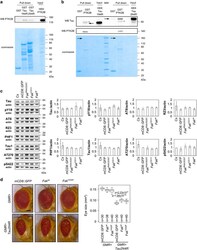
- Figure 3 In vitro biochemical interaction between PTK2B and Tau. ( a ) Pull down of PTK2B with GST, GST-Tau1N4R and GST-Tau2N4R protein constructs. Upper panel: detection of PTK2B in the pull down extract by western blotting. Lower panel: corresponding Coomassie blue gel used as loading control of GST constructs. ( b ) Pull down of Tau1N4R and Tau2N4R with GST and GST-PTK2B protein constructs. Upper panel: detection of Tau (arrows) in the pull down extract by western blotting (an unspecific band is labeled with a star, MW, molecular weight). Lower panel: corresponding Coomassie blue gel used as loading control of GST constructs. As the GST-PTK2B constructs were difficult to produce and visualize with Coomassie blue staining (arrows), we checked the GST-PTK2B construct through detection of PTK2B by western blotting (middle panel). All results shown in panels ( a and b ) are representative of three independent experiments. ( c ) Western blotting analysis of Tau phosphorylation in different Fak conditions in the Drosophila eye. Actin is used as a loading control ( n =2-4). ( d ) Images and quantification of fly eyes co-expressing the 2N4R Tau isoform and a mCD8::GFP construct used as control or wild-type Fak or mutant Fak Y430F (scale bar 0.1 mm).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
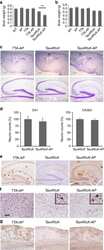
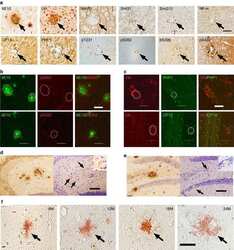
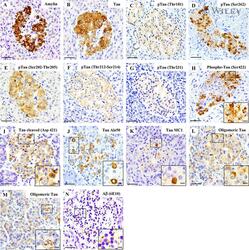
- Amylin, tau, and Abeta inclusions in pancreatic beta cells. Immunohistochemistry is shown for amylin (A), total tau (B), Thr181 phosphorylated tau (AT270; C), Ser262 phosphorylated tau (D), Ser202-Thr205 phosphorylated tau (AT8; E), Thr212-Ser214 phosphorylated tau (AT100; F), Thr231 phosphorylated tau (AT180; G), Ser422 phosphorylated tau (H), Asp421 cleaved tau (I), tau Alz50 (J), tau MC-1 (K), endocrine oligomeric tau (L), exocrine oligomeric tau (M), and Abeta inclusions (N) in pancreatic cells from a 75-year-old male with Alzheimer disease and no history of type 2 diabetes mellitus. Magnification = x40, scale bars = 50mum. The boxed areas (H-N) were examined at a higher magnification: x63 magnification, scale bars = 20mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
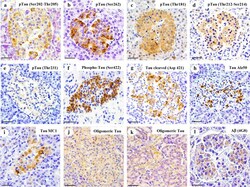
- Fig. 3 Tau and Abeta inclusions in pancreatic beta cells of subjects with Parkinson''s disease, Lewy body dementia and incidental Lewy body disease. Immunohistochemistry for Ser202-Thr205-phosphorylated tau (AT8) ( a ), Ser262-phosphorylated tau ( b ), Thr181-phosphorylated tau (AT270) ( c ), Thr212-Ser214-phosphorylated tau (AT100) ( d ), Thr231-phosphorylated tau (AT180) ( e ), Ser422-phosphorylated tau ( f ), Asp421-cleaved tau ( g ), tau Alz50 ( h ), tau MC-1 ( i ), endocrine oligomeric tau ( j ), exocrine oligomeric tau ( k ) and Abeta ( l ) inclusions in pancreatic cells from subjects with incidental Lewy body disease ( a , b , c , f ), Parkinson''s disease ( d , e , g , h , i , l ) and dementia with Lewy body ( j , k ): a 40 x magnification; scale bar = 50 um
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
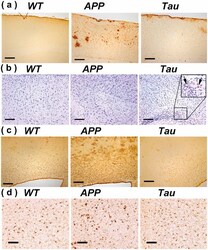
- Experimental details
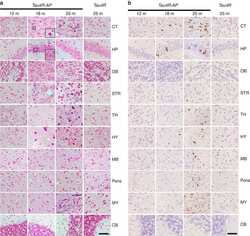
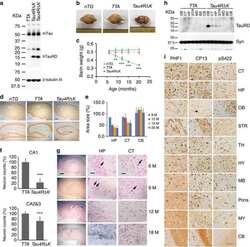
- Fig 6. Brain sections of WT, APP and Tau mice stained by antibodies specific to A beta (6E10) (a), phosphorylated tau: Tau-pS422 (b), reactive astrocytes (GFAP) (c), and microglia (IBA1) (d). Scale bars are 200 mu m in (a-c) and 100 mu m in (d).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
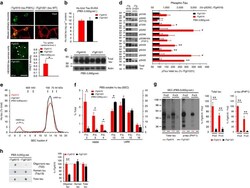
- Experimental details
- Figure 2 Lack of PBS-soluble phosphorylated HMW tau species is associated with low tau uptake in primary neurons. ( a , top) Uptake of human tau from brain extracts from rTg4510 and rTg21221 mice by primary neurons (PBS-3,000 g , 500 ng ml -1 human tau). Neurons were immunostained with human tau-specific antibody (green) and total (human and mouse) tau antibody (red). ( a , bottom) Tau uptake assay in HEK-tau-biosensor cells. Brain extracts (10 mug protein) were applied to the cells (lipofectamie (-)). ( n =4) Unpaired t -test. Scale bar, 50 mum. ( b ) Human tau levels in brain extracts (ELISA). ( c ) Immunoblot analysis of PBS-soluble extracts with total tau antibody (DA9). Up-shifted bands in rTg4510 brain suggest phosphorylation of tau (arrow). ( d ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. ( n =3-4) Unpaired t -test. ( e , f ) SEC analysis of PBS-soluble tau. ( e ) Representative graph of human tau levels (ELISA) in SEC-separated samples ( f ) Mean human tau levels of HMW (Frc. 2-4) and LMW (Frc. 13-16) SEC fractions. ( n =3-6) Unpaired t -test. ( g ) Immunoblot analysis (SDS-PAGE) of SEC-separated fractions from brain extracts (total tau, DAKO). Quantification of band density is also shown (right graphs) ( n =4). Unpaired t -test. ( h ) Dot blot analysis of PBS-soluble brain extracts with tau oligomer-specific antibody (T22), h
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot