AHB0061
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [10]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Immunohistochemistry [2]
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- AHB0061 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Tau (Cleaved Asp421, Asp422) Monoclonal Antibody (TauC3)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This antibody recognizes tau when truncated at Asp421. Several caspases, including caspase-3, caspase-7, and caspase-8, cleave tau at this site, yielding the truncated form of the protein. This antibody does not react with full-length tau, or with tau truncated at Glu391 or Ala429. This antibody provides a tool for studying neuronal cell apoptosis.
- Antibody clone number
- TauC3
- Concentration
- 0.5 mg/mL
Submitted references Targeting MicroRNA-485-3p Blocks Alzheimer's Disease Progression.
A unique tau conformation generated by an acetylation-mimic substitution modulates P301S-dependent tau pathology and hyperphosphorylation.
Early-life exposure to high-fat diet influences brain health in aging mice.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Pimozide reduces toxic forms of tau in TauC3 mice via 5' adenosine monophosphate-activated protein kinase-mediated autophagy.
Modifications of tau protein after cerebral ischemia and reperfusion in rats are similar to those occurring in Alzheimer's disease - Hyperphosphorylation and cleavage of 4- and 3-repeat tau.
Subacute Changes in Cleavage Processing of Amyloid Precursor Protein and Tau following Penetrating Traumatic Brain Injury.
The identification of raft-derived tau-associated vesicles that are incorporated into immature tangles and paired helical filaments.
Carboxy terminus heat shock protein 70 interacting protein reduces tau-associated degenerative changes.
Pericyte loss influences Alzheimer-like neurodegeneration in mice.
Koh HS, Lee S, Lee HJ, Min JW, Iwatsubo T, Teunissen CE, Cho HJ, Ryu JH
International journal of molecular sciences 2021 Dec 4;22(23)
International journal of molecular sciences 2021 Dec 4;22(23)
A unique tau conformation generated by an acetylation-mimic substitution modulates P301S-dependent tau pathology and hyperphosphorylation.
Ajit D, Trzeciakiewicz H, Tseng JH, Wander CM, Chen Y, Ajit A, King DP, Cohen TJ
The Journal of biological chemistry 2019 Nov 8;294(45):16698-16711
The Journal of biological chemistry 2019 Nov 8;294(45):16698-16711
Early-life exposure to high-fat diet influences brain health in aging mice.
Di Meco A, Praticò D
Aging cell 2019 Dec;18(6):e13040
Aging cell 2019 Dec;18(6):e13040
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Pimozide reduces toxic forms of tau in TauC3 mice via 5' adenosine monophosphate-activated protein kinase-mediated autophagy.
Kim YD, Jeong EI, Nah J, Yoo SM, Lee WJ, Kim Y, Moon S, Hong SH, Jung YK
Journal of neurochemistry 2017 Sep;142(5):734-746
Journal of neurochemistry 2017 Sep;142(5):734-746
Modifications of tau protein after cerebral ischemia and reperfusion in rats are similar to those occurring in Alzheimer's disease - Hyperphosphorylation and cleavage of 4- and 3-repeat tau.
Fujii H, Takahashi T, Mukai T, Tanaka S, Hosomi N, Maruyama H, Sakai N, Matsumoto M
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2017 Jul;37(7):2441-2457
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2017 Jul;37(7):2441-2457
Subacute Changes in Cleavage Processing of Amyloid Precursor Protein and Tau following Penetrating Traumatic Brain Injury.
Cartagena CM, Mountney A, Hwang H, Swiercz A, Rammelkamp Z, Boutte AM, Shear DA, Tortella FC, Schmid KE
PloS one 2016;11(7):e0158576
PloS one 2016;11(7):e0158576
The identification of raft-derived tau-associated vesicles that are incorporated into immature tangles and paired helical filaments.
Nishikawa T, Takahashi T, Nakamori M, Hosomi N, Maruyama H, Miyazaki Y, Izumi Y, Matsumoto M
Neuropathology and applied neurobiology 2016 Dec;42(7):639-653
Neuropathology and applied neurobiology 2016 Dec;42(7):639-653
Carboxy terminus heat shock protein 70 interacting protein reduces tau-associated degenerative changes.
Saidi LJ, Polydoro M, Kay KR, Sanchez L, Mandelkow EM, Hyman BT, Spires-Jones TL
Journal of Alzheimer's disease : JAD 2015;44(3):937-47
Journal of Alzheimer's disease : JAD 2015;44(3):937-47
Pericyte loss influences Alzheimer-like neurodegeneration in mice.
Sagare AP, Bell RD, Zhao Z, Ma Q, Winkler EA, Ramanathan A, Zlokovic BV
Nature communications 2013;4:2932
Nature communications 2013;4:2932
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
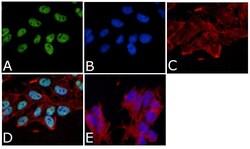
- Experimental details
- Immunofluorescent analysis of Tau (Cleavage site 421/422) Antibody (TauC3) was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Tau (Cleavage site 421/422) Antibody (TauC3) (Product # AHB0061) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Tau (421/422 cleavage site) showing staining in the cytoplasm of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Tau (421/422 cleavage site) monoclonal antibody (Product # AHB0061) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
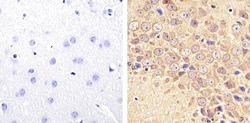
- Experimental details
- Immunohistochemistry analysis of Tau (421/422 cleavage site) showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Tau (421/422 cleavage site) monoclonal antibody (Product # AHB0061) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
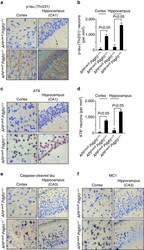
- Figure 7 Tau pathology in pericyte-deficient APP sw/0 Pdgfrbeta +/- mice. ( a - f ) Representative cortex and hippocampus sections stained with antibodies against p-tau (Thr231) ( a ), quantification of p-tau (Thr231)-positive neurons ( b ), p-tau (Ser202/Thr205, AT8) ( c ), quantification of AT8-positive neurons ( d ), caspase-cleaved tau ( e ) and an early pathological tau marker MC1 ( f ) in 9-month-old APP sw/0 Pdgfrbeta +/- mice and age-matched APP sw/0 Pdgfrbeta +/+ littermates. In a , c , e , f , scale bar, 25 mum. CA1 and CA3 denote hippocampal regions. In b , d , values are means+-s.e.m., n =5 mice per group. P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
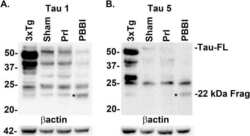
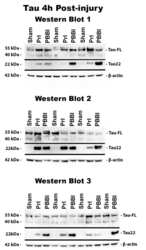
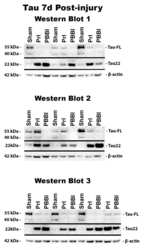
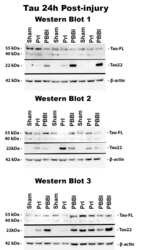
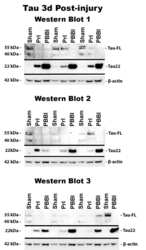
- Fig 6 Detection of FL tau and tau fragments following penetrating TBI. Protein levels from Sham, PrI and PBBI were evaluated by Western blot at time points 4h, 24h, 3d and 7d post injury. Bands were measured and normalized to corresponding betaactin levels. A representative blot at 7d post injury is shown in (A). Quantification corresponding to (B) tau-FL, (C) 40 kDa tau and (D) 22 kDa tau show significant loss of tau-FL and 40 kDa tau and significant increases in 22 kDa tau fragment through 7d. Statistical analysis of changes at each time point was conducted (K-W, * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
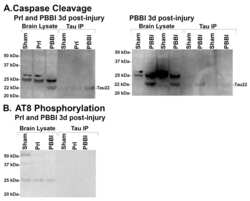
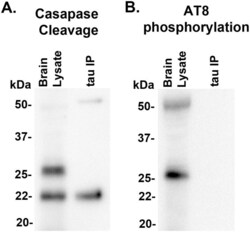
- Fig 9 22 kDa Tau fragment binding confirmation. Protein levels from 7d post-injury PBBI brain lysate and 7d post-PBBI brain lysate that underwent immunoprecipitation with Tau 1 antibody (tau IP) were evaluated for evidence of (A) tau caspase cleavage at Asp421and (B) tau phosphorylation using AT8 antibody. Evidence of caspase cleavage of the 22 kDa tau fragment was seen using the tau Asp421 specific caspase antibody in both the original brain lysate as well as the Tau 1 immunoprecipitation product. Antibody AT8, specific for phosphorylation at sites S202, did not detect the 22 kDa tau fragment. Experiments replicated at least 3 times (sham, PBBI). AT8 phosphorylation experiments replicated twice (sham, PBBI).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
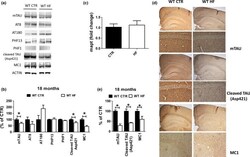
- Figure 3 Effect of gestational high-fat diet on tau brain pathology. (a) Protein levels of tau and its phosphorylated epitopes measured by Western blot in brain cortex of wild-type regular chow (WT CTR) and wild-type high-fat diet (WT HF) offspring at 18 months of age. (b) mRNA levels of tau measured by qPCR in brain cortex of wild-type regular chow (WT CTR) and wild-type high-fat diet (WT HF) offspring at 18 months of age. (c) Immunoreactivity for tau, caspase-3-cleaved tau, and aggregation prone tau (MC1) in brain sections of wild-type regular chow (WT CTR) and wild-type high-fat diet (WT HF) offspring at 18 months of age. (d) Densitometry of immunoreactivities shown in panel a. (e) Quantitative analysis of the immunoreactivities shown in panel c. (* p < .05)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
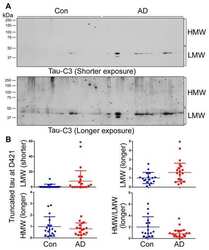
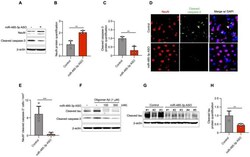
- Figure 4 miR-485-3p antisense oligonucleotide (ASO) reduces apoptosis and truncated tau level. ( A ) Immunoblotting for NeuN and cleaved caspase-3 expression in fractions from the cortex region of control- or miR-485-3p ASO-injected 10-month-old 5XFAD mice. Data are representative of three independent experiments. ( B , C ) Relative protein quantification of NeuN ( B ) and cleaved caspase-3 ( C ) obtained from ( A ). Data obtained across four independent experiments are expressed as mean +- SD. ( D ) Immunohistochemical staining for NeuN and cleaved caspase-3 in coronal brain sections from control- or miR-485-3p ASO-injected 10-month-old 5XFAD mice. Scale bars, 20 mum. Data are representative of three independent experiments. ( E ) Quantification of the NeuN + cleaved caspase-3 + cells from control- ( n = 4) or miR-485-3p ASO- ( n = 5) injected 10-month-old 5XFAD mice. ( F ) Primary cortical neurons were treated with 1 muM oligomeric Abeta (1-42) for 6 h; lysates were immunoblotted with cleaved tau or cleaved caspase-3 antibodies. Data are representative of three independent experiments. ( G ) Immunoblotting for cleaved tau protein expression in control- ( n = 3) or miR-485-3p ASO- ( n = 5) injected 10-month-old 5XFAD mice. ( H ) Relative protein quantification of cleaved tau obtained from ( G ). Data obtained over three independent experiments are expressed as mean +- SD. ** p < 0.01; *** p < 0.001 (two-tailed t -test).
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry