Antibody data
- Antibody Data
- Antigen structure
- References [54]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [32]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 32-2700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Acetyl-alpha Tubulin (Lys40) Monoclonal Antibody (6-11B-1)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This antibody reacts with the ~ 55 kDa acetylated form of alpha-tubulin. This antibody reacts with human, mouse, and rat. Reactivity is confirmed with NIH3T3 and HeLa cells.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 6-11B-1
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Targeting an anchored phosphatase-deacetylase unit restores renal ciliary homeostasis.
Changes in microtubule stability in zebrafish (Danio rerio) embryos after glyphosate exposure.
Sept8/SEPTIN8 involvement in cellular structure and kidney damage is identified by genetic mapping and a novel human tubule hypoxic model.
Adenine nucleotide translocase regulates airway epithelial metabolism, surface hydration and ciliary function.
Polarized Sonic Hedgehog Protein Localization and a Shift in the Expression of Region-Specific Molecules Is Associated With the Secondary Palate Development in the Veiled Chameleon.
Mutations in GRK2 cause Jeune syndrome by impairing Hedgehog and canonical Wnt signaling.
MECP2 mutations affect ciliogenesis: a novel perspective for Rett syndrome and related disorders.
Homogeneous multifocal excitation for high-throughput super-resolution imaging.
Vav3 Mediates Pseudomonas aeruginosa Adhesion to the Cystic Fibrosis Airway Epithelium.
Novel IQCE variations confirm its role in postaxial polydactyly and cause ciliary defect phenotype in zebrafish.
Succinyl-CoA Ligase Deficiency in Pro-inflammatory and Tissue-Invasive T Cells.
Conditional deletion of Nedd4-2 in lung epithelial cells causes progressive pulmonary fibrosis in adult mice.
Synergistic Genetic Interactions between Pkhd1 and Pkd1 Result in an ARPKD-Like Phenotype in Murine Models.
Fibroblast growth factor receptor influences primary cilium length through an interaction with intestinal cell kinase.
Substrate Elasticity Governs Differentiation of Renal Tubule Cells in Prolonged Culture.
Accelerating single molecule localization microscopy through parallel processing on a high-performance computing cluster.
Increased susceptibility of airway epithelial cells from ataxia-telangiectasia to S. pneumoniae infection due to oxidative damage and impaired innate immunity.
Compromised phagosome maturation underlies RPE pathology in cell culture and whole animal models of Smith-Lemli-Opitz Syndrome.
Chemokine expression in the early response to injury in human airway epithelial cells.
Deletion of Kncn Does Not Affect Kinocilium and Stereocilia Bundle Morphogenesis and Mechanotransduction in Cochlear Hair Cells.
Characterizing well-differentiated culture of primary human nasal epithelial cells for use in wound healing assays.
Orbital Shear Stress Regulates Differentiation and Barrier Function of Primary Renal Tubular Epithelial Cells.
Overexpressed HDAC8 in cervical cancer cells shows functional redundancy of tubulin deacetylation with HDAC6.
Regulation of ciliary function by fibroblast growth factor signaling identifies FGFR3-related disorders achondroplasia and thanatophoric dysplasia as ciliopathies.
Ciliogenesis and cell cycle alterations contribute to KIF2A-related malformations of cortical development.
VPS4 is a dynamic component of the centrosome that regulates centrosome localization of γ-tubulin, centriolar satellite stability and ciliogenesis.
The molecular motor Myosin Va interacts with the cilia-centrosomal protein RPGRIP1L.
Identification of Elongated Primary Cilia with Impaired Mechanotransduction in Idiopathic Scoliosis Patients.
Cell-cycle-dependent TGFβ-BMP antagonism regulates neural tube closure by modulating tight junctions.
Identification of Chlamydomonas Central Core Centriolar Proteins Reveals a Role for Human WDR90 in Ciliogenesis.
An inactivating mutation in intestinal cell kinase, ICK, impairs hedgehog signalling and causes short rib-polydactyly syndrome.
Galanin regulates blood glucose level in the zebrafish: a morphological and functional study.
HDAC 1 and 6 modulate cell invasion and migration in clear cell renal cell carcinoma.
A missense mutation in DCDC2 causes human recessive deafness DFNB66, likely by interfering with sensory hair cell and supporting cell cilia length regulation.
ADAMTS9-Mediated Extracellular Matrix Dynamics Regulates Umbilical Cord Vascular Smooth Muscle Differentiation and Rotation.
The Eya1 phosphatase promotes Shh signaling during hindbrain development and oncogenesis.
The role of Arp2/3 in growth cone actin dynamics and guidance is substrate dependent.
Retinoic acid improves morphology of cultured peritoneal mesothelial cells from patients undergoing dialysis.
Analysis of ciliary band formation in the mollusc Ilyanassa obsoleta.
Primary cilia modulate Ihh signal transduction in response to hydrostatic loading of growth plate chondrocytes.
Bioartificial Renal Epithelial Cell System (BRECS): A Compact, Cryopreservable Extracorporeal Renal Replacement Device.
Primary cilia utilize glycoprotein-dependent adhesion mechanisms to stabilize long-lasting cilia-cilia contacts.
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
MicroCT for molecular imaging: quantitative visualization of complete three-dimensional distributions of gene products in embryonic limbs.
Defective Wnt-dependent cerebellar midline fusion in a mouse model of Joubert syndrome.
Subcellular spatial regulation of canonical Wnt signalling at the primary cilium.
Subnuclear development of the zebrafish habenular nuclei requires ER translocon function.
A microfluidic bioreactor with integrated transepithelial electrical resistance (TEER) measurement electrodes for evaluation of renal epithelial cells.
AHI1 is required for photoreceptor outer segment development and is a modifier for retinal degeneration in nephronophthisis.
Impaired Wnt-beta-catenin signaling disrupts adult renal homeostasis and leads to cystic kidney ciliopathy.
Antizyme, a mediator of ubiquitin-independent proteasomal degradation and its inhibitor localize to centrosomes and modulate centriole amplification.
Mutations in the cilia gene ARL13B lead to the classical form of Joubert syndrome.
Differentiated growth of human renal tubule cells on thin-film and nanostructured materials.
Rare incorporation of bone marrow-derived cells into kidney after folic acid-induced injury.
Gopalan J, Omar MH, Roy A, Cruz NM, Falcone J, Jones KN, Forbush KA, Himmelfarb J, Freedman BS, Scott JD
eLife 2021 Jul 12;10
eLife 2021 Jul 12;10
Changes in microtubule stability in zebrafish (Danio rerio) embryos after glyphosate exposure.
Díaz-Martín RD, Valencia-Hernández JD, Betancourt-Lozano M, Yáñez-Rivera B
Heliyon 2021 Jan;7(1):e06027
Heliyon 2021 Jan;7(1):e06027
Sept8/SEPTIN8 involvement in cellular structure and kidney damage is identified by genetic mapping and a novel human tubule hypoxic model.
Keele GR, Prokop JW, He H, Holl K, Littrell J, Deal AW, Kim Y, Kyle PB, Attipoe E, Johnson AC, Uhl KL, Sirpilla OL, Jahanbakhsh S, Robinson M, Levy S, Valdar W, Garrett MR, Solberg Woods LC
Scientific reports 2021 Jan 22;11(1):2071
Scientific reports 2021 Jan 22;11(1):2071
Adenine nucleotide translocase regulates airway epithelial metabolism, surface hydration and ciliary function.
Kliment CR, Nguyen JMK, Kaltreider MJ, Lu Y, Claypool SM, Radder JE, Sciurba FC, Zhang Y, Gregory AD, Iglesias PA, Sidhaye VK, Robinson DN
Journal of cell science 2021 Feb 25;134(4)
Journal of cell science 2021 Feb 25;134(4)
Polarized Sonic Hedgehog Protein Localization and a Shift in the Expression of Region-Specific Molecules Is Associated With the Secondary Palate Development in the Veiled Chameleon.
Hampl M, Dumkova J, Kavkova M, Dosedelova H, Bryjova A, Zahradnicek O, Pyszko M, Macholan M, Zikmund T, Kaiser J, Buchtova M
Frontiers in cell and developmental biology 2020;8:572
Frontiers in cell and developmental biology 2020;8:572
Mutations in GRK2 cause Jeune syndrome by impairing Hedgehog and canonical Wnt signaling.
Bosakova M, Abraham SP, Nita A, Hruba E, Buchtova M, Taylor SP, Duran I, Martin J, Svozilova K, Barta T, Varecha M, Balek L, Kohoutek J, Radaszkiewicz T, Pusapati GV, Bryja V, Rush ET, Thiffault I, Nickerson DA, Bamshad MJ, University of Washington Center for Mendelian Genomics, Rohatgi R, Cohn DH, Krakow D, Krejci P
EMBO molecular medicine 2020 Nov 6;12(11):e11739
EMBO molecular medicine 2020 Nov 6;12(11):e11739
MECP2 mutations affect ciliogenesis: a novel perspective for Rett syndrome and related disorders.
Frasca A, Spiombi E, Palmieri M, Albizzati E, Valente MM, Bergo A, Leva B, Kilstrup-Nielsen C, Bianchi F, Di Carlo V, Di Cunto F, Landsberger N
EMBO molecular medicine 2020 Jun 8;12(6):e10270
EMBO molecular medicine 2020 Jun 8;12(6):e10270
Homogeneous multifocal excitation for high-throughput super-resolution imaging.
Mahecic D, Gambarotto D, Douglass KM, Fortun D, Banterle N, Ibrahim KA, Le Guennec M, Gönczy P, Hamel V, Guichard P, Manley S
Nature methods 2020 Jul;17(7):726-733
Nature methods 2020 Jul;17(7):726-733
Vav3 Mediates Pseudomonas aeruginosa Adhesion to the Cystic Fibrosis Airway Epithelium.
Badaoui M, Zoso A, Idris T, Bacchetta M, Simonin J, Lemeille S, Wehrle-Haller B, Chanson M
Cell reports 2020 Jul 7;32(1):107842
Cell reports 2020 Jul 7;32(1):107842
Novel IQCE variations confirm its role in postaxial polydactyly and cause ciliary defect phenotype in zebrafish.
Estrada-Cuzcano A, Etard C, Delvallée C, Stoetzel C, Schaefer E, Scheidecker S, Geoffroy V, Schneider A, Studer F, Mattioli F, Chennen K, Sigaudy S, Plassard D, Poch O, Piton A, Strahle U, Muller J, Dollfus H
Human mutation 2020 Jan;41(1):240-254
Human mutation 2020 Jan;41(1):240-254
Succinyl-CoA Ligase Deficiency in Pro-inflammatory and Tissue-Invasive T Cells.
Wu B, Qiu J, Zhao TV, Wang Y, Maeda T, Goronzy IN, Akiyama M, Ohtsuki S, Jin K, Tian L, Goronzy JJ, Weyand CM
Cell metabolism 2020 Dec 1;32(6):967-980.e5
Cell metabolism 2020 Dec 1;32(6):967-980.e5
Conditional deletion of Nedd4-2 in lung epithelial cells causes progressive pulmonary fibrosis in adult mice.
Duerr J, Leitz DHW, Szczygiel M, Dvornikov D, Fraumann SG, Kreutz C, Zadora PK, Seyhan Agircan A, Konietzke P, Engelmann TA, Hegermann J, Mulugeta S, Kawabe H, Knudsen L, Ochs M, Rotin D, Muley T, Kreuter M, Herth FJF, Wielpütz MO, Beers MF, Klingmüller U, Mall MA
Nature communications 2020 Apr 24;11(1):2012
Nature communications 2020 Apr 24;11(1):2012
Synergistic Genetic Interactions between Pkhd1 and Pkd1 Result in an ARPKD-Like Phenotype in Murine Models.
Olson RJ, Hopp K, Wells H, Smith JM, Furtado J, Constans MM, Escobar DL, Geurts AM, Torres VE, Harris PC
Journal of the American Society of Nephrology : JASN 2019 Nov;30(11):2113-2127
Journal of the American Society of Nephrology : JASN 2019 Nov;30(11):2113-2127
Fibroblast growth factor receptor influences primary cilium length through an interaction with intestinal cell kinase.
Kunova Bosakova M, Nita A, Gregor T, Varecha M, Gudernova I, Fafilek B, Barta T, Basheer N, Abraham SP, Balek L, Tomanova M, Fialova Kucerova J, Bosak J, Potesil D, Zieba J, Song J, Konik P, Park S, Duran I, Zdrahal Z, Smajs D, Jansen G, Fu Z, Ko HW, Hampl A, Trantirek L, Krakow D, Krejci P
Proceedings of the National Academy of Sciences of the United States of America 2019 Mar 5;116(10):4316-4325
Proceedings of the National Academy of Sciences of the United States of America 2019 Mar 5;116(10):4316-4325
Substrate Elasticity Governs Differentiation of Renal Tubule Cells in Prolonged Culture.
Love HD, Ao M, Jorgensen S, Swearingen L, Ferrell N, Evans R, Gewin L, Harris RC, Zent R, Roy S, Fissell WH
Tissue engineering. Part A 2019 Jul;25(13-14):1013-1022
Tissue engineering. Part A 2019 Jul;25(13-14):1013-1022
Accelerating single molecule localization microscopy through parallel processing on a high-performance computing cluster.
Munro I, García E, Yan M, Guldbrand S, Kumar S, Kwakwa K, Dunsby C, Neil MAA, French PMW
Journal of microscopy 2019 Feb;273(2):148-160
Journal of microscopy 2019 Feb;273(2):148-160
Increased susceptibility of airway epithelial cells from ataxia-telangiectasia to S. pneumoniae infection due to oxidative damage and impaired innate immunity.
Yeo AJ, Henningham A, Fantino E, Galbraith S, Krause L, Wainwright CE, Sly PD, Lavin MF
Scientific reports 2019 Feb 22;9(1):2627
Scientific reports 2019 Feb 22;9(1):2627
Compromised phagosome maturation underlies RPE pathology in cell culture and whole animal models of Smith-Lemli-Opitz Syndrome.
Ramachandra Rao S, Pfeffer BA, Más Gómez N, Skelton LA, Keiko U, Sparrow JR, Rowsam AM, Mitchell CH, Fliesler SJ
Autophagy 2018;14(10):1796-1817
Autophagy 2018;14(10):1796-1817
Chemokine expression in the early response to injury in human airway epithelial cells.
Xie B, Laxman B, Hashemifar S, Stern R, Gilliam TC, Maltsev N, White SR
PloS one 2018;13(3):e0193334
PloS one 2018;13(3):e0193334
Deletion of Kncn Does Not Affect Kinocilium and Stereocilia Bundle Morphogenesis and Mechanotransduction in Cochlear Hair Cells.
Hu Q, Guo L, Li J, Song C, Yu L, He DZZ, Xiong W
Frontiers in molecular neuroscience 2018;11:326
Frontiers in molecular neuroscience 2018;11:326
Characterizing well-differentiated culture of primary human nasal epithelial cells for use in wound healing assays.
Schagen J, Sly PD, Fantino E
Laboratory investigation; a journal of technical methods and pathology 2018 Nov;98(11):1478-1486
Laboratory investigation; a journal of technical methods and pathology 2018 Nov;98(11):1478-1486
Orbital Shear Stress Regulates Differentiation and Barrier Function of Primary Renal Tubular Epithelial Cells.
Ferrell N, Cheng J, Miao S, Roy S, Fissell WH
ASAIO journal (American Society for Artificial Internal Organs : 1992) 2018 Nov Dec;64(6):766-772
ASAIO journal (American Society for Artificial Internal Organs : 1992) 2018 Nov Dec;64(6):766-772
Overexpressed HDAC8 in cervical cancer cells shows functional redundancy of tubulin deacetylation with HDAC6.
Vanaja GR, Ramulu HG, Kalle AM
Cell communication and signaling : CCS 2018 May 2;16(1):20
Cell communication and signaling : CCS 2018 May 2;16(1):20
Regulation of ciliary function by fibroblast growth factor signaling identifies FGFR3-related disorders achondroplasia and thanatophoric dysplasia as ciliopathies.
Kunova Bosakova M, Varecha M, Hampl M, Duran I, Nita A, Buchtova M, Dosedelova H, Machat R, Xie Y, Ni Z, Martin JH, Chen L, Jansen G, Krakow D, Krejci P
Human molecular genetics 2018 Mar 15;27(6):1093-1105
Human molecular genetics 2018 Mar 15;27(6):1093-1105
Ciliogenesis and cell cycle alterations contribute to KIF2A-related malformations of cortical development.
Broix L, Asselin L, Silva CG, Ivanova EL, Tilly P, Gilet JG, Lebrun N, Jagline H, Muraca G, Saillour Y, Drouot N, Reilly ML, Francis F, Benmerah A, Bahi-Buisson N, Belvindrah R, Nguyen L, Godin JD, Chelly J, Hinckelmann MV
Human molecular genetics 2018 Jan 15;27(2):224-238
Human molecular genetics 2018 Jan 15;27(2):224-238
VPS4 is a dynamic component of the centrosome that regulates centrosome localization of γ-tubulin, centriolar satellite stability and ciliogenesis.
Ott C, Nachmias D, Adar S, Jarnik M, Sherman S, Birnbaum RY, Lippincott-Schwartz J, Elia N
Scientific reports 2018 Feb 20;8(1):3353
Scientific reports 2018 Feb 20;8(1):3353
The molecular motor Myosin Va interacts with the cilia-centrosomal protein RPGRIP1L.
Assis LH, Silva-Junior RM, Dolce LG, Alborghetti MR, Honorato RV, Nascimento AF, Melo-Hanchuk TD, Trindade DM, Tonoli CC, Santos CT, Oliveira PS, Larson RE, Kobarg J, Espreafico EM, Giuseppe PO, Murakami MT
Scientific reports 2017 Mar 7;7:43692
Scientific reports 2017 Mar 7;7:43692
Identification of Elongated Primary Cilia with Impaired Mechanotransduction in Idiopathic Scoliosis Patients.
Oliazadeh N, Gorman KF, Eveleigh R, Bourque G, Moreau A
Scientific reports 2017 Mar 14;7:44260
Scientific reports 2017 Mar 14;7:44260
Cell-cycle-dependent TGFβ-BMP antagonism regulates neural tube closure by modulating tight junctions.
Amarnath S, Agarwala S
Journal of cell science 2017 Jan 1;130(1):119-131
Journal of cell science 2017 Jan 1;130(1):119-131
Identification of Chlamydomonas Central Core Centriolar Proteins Reveals a Role for Human WDR90 in Ciliogenesis.
Hamel V, Steib E, Hamelin R, Armand F, Borgers S, Flückiger I, Busso C, Olieric N, Sorzano COS, Steinmetz MO, Guichard P, Gönczy P
Current biology : CB 2017 Aug 21;27(16):2486-2498.e6
Current biology : CB 2017 Aug 21;27(16):2486-2498.e6
An inactivating mutation in intestinal cell kinase, ICK, impairs hedgehog signalling and causes short rib-polydactyly syndrome.
Paige Taylor S, Kunova Bosakova M, Varecha M, Balek L, Barta T, Trantirek L, Jelinkova I, Duran I, Vesela I, Forlenza KN, Martin JH, Hampl A, University of Washington Center for Mendelian Genomics, Bamshad M, Nickerson D, Jaworski ML, Song J, Ko HW, Cohn DH, Krakow D, Krejci P
Human molecular genetics 2016 Sep 15;25(18):3998-4011
Human molecular genetics 2016 Sep 15;25(18):3998-4011
Galanin regulates blood glucose level in the zebrafish: a morphological and functional study.
Podlasz P, Jakimiuk A, Chmielewska-Krzesinska M, Kasica N, Nowik N, Kaleczyc J
Histochemistry and cell biology 2016 Jan;145(1):105-17
Histochemistry and cell biology 2016 Jan;145(1):105-17
HDAC 1 and 6 modulate cell invasion and migration in clear cell renal cell carcinoma.
Ramakrishnan S, Ku S, Ciamporcero E, Miles KM, Attwood K, Chintala S, Shen L, Ellis L, Sotomayor P, Swetzig W, Huang R, Conroy D, Orillion A, Das G, Pili R
BMC cancer 2016 Aug 9;16:617
BMC cancer 2016 Aug 9;16:617
A missense mutation in DCDC2 causes human recessive deafness DFNB66, likely by interfering with sensory hair cell and supporting cell cilia length regulation.
Grati M, Chakchouk I, Ma Q, Bensaid M, Desmidt A, Turki N, Yan D, Baanannou A, Mittal R, Driss N, Blanton S, Farooq A, Lu Z, Liu XZ, Masmoudi S
Human molecular genetics 2015 May 1;24(9):2482-91
Human molecular genetics 2015 May 1;24(9):2482-91
ADAMTS9-Mediated Extracellular Matrix Dynamics Regulates Umbilical Cord Vascular Smooth Muscle Differentiation and Rotation.
Nandadasa S, Nelson CM, Apte SS
Cell reports 2015 Jun 16;11(10):1519-28
Cell reports 2015 Jun 16;11(10):1519-28
The Eya1 phosphatase promotes Shh signaling during hindbrain development and oncogenesis.
Eisner A, Pazyra-Murphy MF, Durresi E, Zhou P, Zhao X, Chadwick EC, Xu PX, Hillman RT, Scott MP, Greenberg ME, Segal RA
Developmental cell 2015 Apr 6;33(1):22-35
Developmental cell 2015 Apr 6;33(1):22-35
The role of Arp2/3 in growth cone actin dynamics and guidance is substrate dependent.
San Miguel-Ruiz JE, Letourneau PC
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Apr 23;34(17):5895-908
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Apr 23;34(17):5895-908
Retinoic acid improves morphology of cultured peritoneal mesothelial cells from patients undergoing dialysis.
Retana C, Sanchez EI, Gonzalez S, Perez-Lopez A, Cruz A, Lagunas-Munoz J, Alfaro-Cruz C, Vital-Flores S, Reyes JL
PloS one 2013;8(11):e79678
PloS one 2013;8(11):e79678
Analysis of ciliary band formation in the mollusc Ilyanassa obsoleta.
Gharbiah M, Nakamoto A, Nagy LM
Development genes and evolution 2013 Jul;223(4):225-35
Development genes and evolution 2013 Jul;223(4):225-35
Primary cilia modulate Ihh signal transduction in response to hydrostatic loading of growth plate chondrocytes.
Shao YY, Wang L, Welter JF, Ballock RT
Bone 2012 Jan;50(1):79-84
Bone 2012 Jan;50(1):79-84
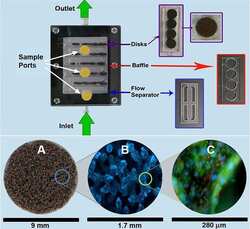
Bioartificial Renal Epithelial Cell System (BRECS): A Compact, Cryopreservable Extracorporeal Renal Replacement Device.
Buffington DA, Pino CJ, Chen L, Westover AJ, Hageman G, Humes HD
Cell medicine 2012 Jan;4(1):33-43
Cell medicine 2012 Jan;4(1):33-43
Primary cilia utilize glycoprotein-dependent adhesion mechanisms to stabilize long-lasting cilia-cilia contacts.
Ott C, Elia N, Jeong SY, Insinna C, Sengupta P, Lippincott-Schwartz J
Cilia 2012 Apr 25;1(1):3
Cilia 2012 Apr 25;1(1):3
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
Roy E, Bruyère J, Flamant P, Bigou S, Ausseil J, Vitry S, Heard JM
Human molecular genetics 2012 Apr 1;21(7):1481-95
Human molecular genetics 2012 Apr 1;21(7):1481-95
MicroCT for molecular imaging: quantitative visualization of complete three-dimensional distributions of gene products in embryonic limbs.
Metscher BD, Müller GB
Developmental dynamics : an official publication of the American Association of Anatomists 2011 Oct;240(10):2301-8
Developmental dynamics : an official publication of the American Association of Anatomists 2011 Oct;240(10):2301-8
Defective Wnt-dependent cerebellar midline fusion in a mouse model of Joubert syndrome.
Lancaster MA, Gopal DJ, Kim J, Saleem SN, Silhavy JL, Louie CM, Thacker BE, Williams Y, Zaki MS, Gleeson JG
Nature medicine 2011 Jun;17(6):726-31
Nature medicine 2011 Jun;17(6):726-31
Subcellular spatial regulation of canonical Wnt signalling at the primary cilium.
Lancaster MA, Schroth J, Gleeson JG
Nature cell biology 2011 Jun;13(6):700-7
Nature cell biology 2011 Jun;13(6):700-7
Subnuclear development of the zebrafish habenular nuclei requires ER translocon function.
Doll CA, Burkart JT, Hope KD, Halpern ME, Gamse JT
Developmental biology 2011 Dec 1;360(1):44-57
Developmental biology 2011 Dec 1;360(1):44-57
A microfluidic bioreactor with integrated transepithelial electrical resistance (TEER) measurement electrodes for evaluation of renal epithelial cells.
Ferrell N, Desai RR, Fleischman AJ, Roy S, Humes HD, Fissell WH
Biotechnology and bioengineering 2010 Nov 1;107(4):707-16
Biotechnology and bioengineering 2010 Nov 1;107(4):707-16
AHI1 is required for photoreceptor outer segment development and is a modifier for retinal degeneration in nephronophthisis.
Louie CM, Caridi G, Lopes VS, Brancati F, Kispert A, Lancaster MA, Schlossman AM, Otto EA, Leitges M, Gröne HJ, Lopez I, Gudiseva HV, O'Toole JF, Vallespin E, Ayyagari R, Ayuso C, Cremers FP, den Hollander AI, Koenekoop RK, Dallapiccola B, Ghiggeri GM, Hildebrandt F, Valente EM, Williams DS, Gleeson JG
Nature genetics 2010 Feb;42(2):175-80
Nature genetics 2010 Feb;42(2):175-80
Impaired Wnt-beta-catenin signaling disrupts adult renal homeostasis and leads to cystic kidney ciliopathy.
Lancaster MA, Louie CM, Silhavy JL, Sintasath L, Decambre M, Nigam SK, Willert K, Gleeson JG
Nature medicine 2009 Sep;15(9):1046-54
Nature medicine 2009 Sep;15(9):1046-54
Antizyme, a mediator of ubiquitin-independent proteasomal degradation and its inhibitor localize to centrosomes and modulate centriole amplification.
Mangold U, Hayakawa H, Coughlin M, Münger K, Zetter BR
Oncogene 2008 Jan 24;27(5):604-13
Oncogene 2008 Jan 24;27(5):604-13
Mutations in the cilia gene ARL13B lead to the classical form of Joubert syndrome.
Cantagrel V, Silhavy JL, Bielas SL, Swistun D, Marsh SE, Bertrand JY, Audollent S, Attié-Bitach T, Holden KR, Dobyns WB, Traver D, Al-Gazali L, Ali BR, Lindner TH, Caspary T, Otto EA, Hildebrandt F, Glass IA, Logan CV, Johnson CA, Bennett C, Brancati F, International Joubert Syndrome Related Disorders Study Group, Valente EM, Woods CG, Gleeson JG
American journal of human genetics 2008 Aug;83(2):170-9
American journal of human genetics 2008 Aug;83(2):170-9
Differentiated growth of human renal tubule cells on thin-film and nanostructured materials.
Fissell WH, Manley S, Westover A, Humes HD, Fleischman AJ, Roy S
ASAIO journal (American Society for Artificial Internal Organs : 1992) 2006 May-Jun;52(3):221-7
ASAIO journal (American Society for Artificial Internal Organs : 1992) 2006 May-Jun;52(3):221-7
Rare incorporation of bone marrow-derived cells into kidney after folic acid-induced injury.
Szczypka MS, Westover AJ, Clouthier SG, Ferrara JL, Humes HD
Stem cells (Dayton, Ohio) 2005;23(1):44-54
Stem cells (Dayton, Ohio) 2005;23(1):44-54
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
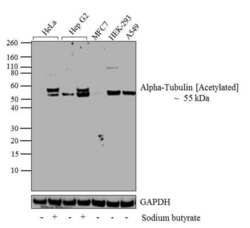
- Experimental details
- Western blot analysis of Alpha-Tubulin (Acetylated) was performed by loading 20 µg of HeLa (lane1), HeLa treated for 12 hr with 5 mM/mL of Sodium Butyrate (lane2), HepG2 (lane3), Hep G2 treated for 12 hr with 5 mM/mL of Sodium Butyrate (lane4), MCF7 (lane5), HEK-293 (lane6) and A549 (lane7) cell lysate using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk for 1 hour at room temperature. Alpha-Tubulin (Acetylated) was detected at ~55 kDa using Alpha-Tubulin (Acetylated) Mouse monoclonal Antibody (Product # 32-2700) at 1-3 µg/mL in 2.5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Mouse IgG - HRP Secondary Antibody (Product # 62-6520) at 1:4000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
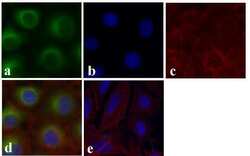
- Experimental details
- Immunofluorescent analysis of Alpha-Tubulin (Acetylated) was done on 70% confluent log phase HeLa cells treated with 5mM sodium butyrate for 24 h. The cells were fixed with 4% paraformaldehyde for 15 minutes; permeabilized with 0.25% Triton X-100 for 10 minutes followed by blocking with 5% BSA for 1 hour at room temperature. The cells were incubated with Alpha-Tubulin (Acetylated) Recombinant Mouse Monoclonal Antibody (Product # 32-2700) at 1 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing Alpha-Tubulin (Acetylated) in cytoplasm and panel e is a no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Human naïve CD4 T cells were activated with anti-CD3/CD28 beads (1:1) (Product # 11132D) for 3 days. Cells were stained with Acetyl-alpha Tubulin (Lys40) Monoclonal Antibody (6-11B-1) (Product # 32-2700) at the concentration of 1:400 for 2 hours on ice, followed by incubation with Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 594 (Product # A-11032) at the concentration of 1:200 for 2 hours on ice. FMO: Fluorescence Minus One. Data courtesy of the antibody data exchange program.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
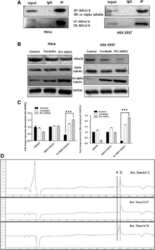
- Experimental details
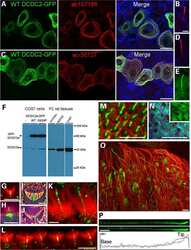
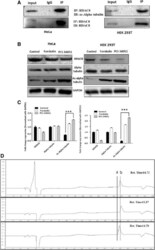
- Fig. 4 a HDAC8 interacts and deacetylates alpha tubulin. a HDAC8 and Acetylated Alpha tubulin Interaction in HeLa and HEK 293 T cells as demonstrated by immunoprecipitation using HDAC8 antibody followed by immunoblot analysis using ac-alpha tubulin. b ac-alpha tubulin levels increase when HDAC8 is inhibited with PCI-34051 (20 muM) or when treated with PKA activator, Forskolin (10 muM) and in HeLa and HEK 293 T cells. c Densitometry analysis of the immunoblot bands showing fold change in expression pattern of HDAC8, Alpha tubulin and acetylated tubulin normalized with GAPDH. d Lower panel of chromatogram represents for Unac-alpha tubulin peptide with a retention time of 14.70 Min. Middle panel of chromatogram for ac-alpha tubulin with retention time of 15.37 Min. Upper panel of chromatogram for GST-HDAC8 (30 mug) + ac-alpha tubulin (500 nM) with a retention time of 14.72 Min. Shift in the retention time from a (15.37) to b (14.72) clearly demonstrates the in vitro deacetylation of ac-alpha tubulin by GST-HDAC8
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 4 a HDAC8 interacts and deacetylates alpha tubulin. a HDAC8 and Acetylated Alpha tubulin Interaction in HeLa and HEK 293 T cells as demonstrated by immunoprecipitation using HDAC8 antibody followed by immunoblot analysis using ac-alpha tubulin. b ac-alpha tubulin levels increase when HDAC8 is inhibited with PCI-34051 (20 muM) or when treated with PKA activator, Forskolin (10 muM) and in HeLa and HEK 293 T cells. c Densitometry analysis of the immunoblot bands showing fold change in expression pattern of HDAC8, Alpha tubulin and acetylated tubulin normalized with GAPDH. d Lower panel of chromatogram represents for Unac-alpha tubulin peptide with a retention time of 14.70 Min. Middle panel of chromatogram for ac-alpha tubulin with retention time of 15.37 Min. Upper panel of chromatogram for GST-HDAC8 (30 mug) + ac-alpha tubulin (500 nM) with a retention time of 14.72 Min. Shift in the retention time from a (15.37) to b (14.72) clearly demonstrates the in vitro deacetylation of ac-alpha tubulin by GST-HDAC8
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
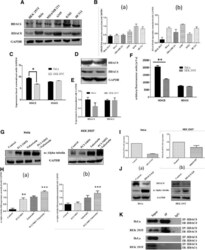
- Fig. 5 HDAC8 might be primary tubulin deacetylase in HeLa cells. a Immunoblot showing the protein expression levels of HDAC8 and HDAC6 in various cancer cell lines. b Densitometric analysis of the (a) HDAC6 bands and (b) HDAC8 bands of immuoblot A. c Real time analysis confirms higher HDAC8 expression compared to HDAC6 in HeLa and HEK 293T normalized with GAPDH * indicates p-value
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
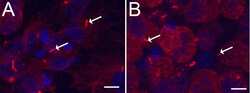
- 1 Supplementary Figure 1. Cilia quantification in MEFs a . Staining for cilia (acetylated tubulin, red, arrow) in serum starved MEFs with reapplication of serum. These results are quantified at the right for at least 200 cells. *** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
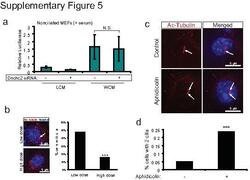
- 5 Supplementary Figure 5. Dnchc2 siRNA and aphidicolin treatment effect on ciliation a . Luciferase activity in MEFs without serum starvation and transfected with 5 pmol Dnchc2 siRNA and treated with WCM or LCM. N.S.=not significant, n=3 independent experiments, Student's t -test. Error bars represent S.E.M. b . Staining for cilia (acetylated tubulin, red, arrow) in MEFs transfected with low dose and high dose (3-fold higher) Dnchc2 siRNA. Hoechst (blue) labels nuclei. These results are quantified in the panel on the right for at least 200 cells. *** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
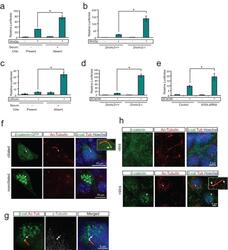
- Figure 1 The primary cilium dampens Wnt activity by regulating beta-catenin a . Luciferase response to Wnt3a conditioned media in a wild-type MEF cell line following serum starvation and re-addition. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
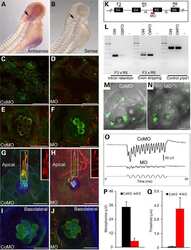
- Figure 3 The primary cilium sequesters Jouberin and beta-catenin away from the nucleus a . 3T3s transfected with two siRNAs to Dnchc2 exhibit accumulation of Jbn-GFP (green) along the length of the cilium (labeled with antibody to acetylated tubulin, red). The nucleus is labeled with Hoechst (blue). Schematic represents the experimental approach and interpretation of these results. Quantification of the green fluorescence along the cilium reveals a shifted localization along the ciliary axoneme in siRNA transfected cells with significantly increased levels of GFP beyond the basal body. Average fluorescence intensities of cells transfected with negative control siRNA (n=7), Dnchc2 siRNA #1 (n=10), or Dnchc2 siRNA #2 (n=11) were normalized and displayed in arbitrary grey value units. Position along the cilium was normalized for total cilia length and results were binned into 20 equal segments. b . Luciferase activity in MEFs transfected with 5 pmol of Dnchc2 siRNA and treated with Wnt3a conditioned media (WCM) or control L-cell conditioned media (LCM). * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
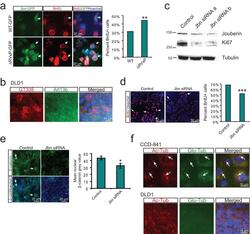
- Figure 5 Regulation of cancer cell proliferation by cilia-localized Jouberin a . BrdU staining (red) in MEFs transfected with GFP tagged wild-type or DeltaRVxP mutant construct (green). Hoechst labels nuclei (blue). Arrows point to GFP positive transfected cells. Quantification of the percent of 200 counted GFP positive cells which stained positive for BrdU is shown at right. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
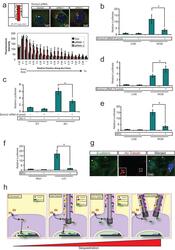
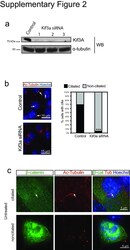
- 2 Supplementary Figure 2. Kif3a knockdown and quantification of ciliation a . Western blot analysis of whole cell lysates from 293Ts transfected with Kif3A siRNA. Alpha-tubulin is shown as a loading control. b . Staining for cilia (acetylated tubulin, red) in MEFs transfected with Kif3a siRNA #3. Arrows point to cilia in control transfected cells. Hoechst (blue) labels nuclei. Results are quantified at the right from at least 200 cells for each. *** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 3 Supplementary Figure 3. beta-catenin and Jbn are sequestered in the presence of cilia a . Staining for endogenous beta-catenin (green) in ciliated (actylated tubulin, red) and nonciliated (arrow) IMCDs revealing increased nuclear (Hoechst, blue, outlined) localization of beta-catenin in nonciliated cells compared with ciliated neighboring cells. b . Quantification of nuclear beta-catenin staining in ciliated and nonciliated IMCDs. Mean grey values were calculated using ImageJ software (NIH) to create a binary mask from Hoechst staining as a nuclear boundary for measurement of beta-catenin levels. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
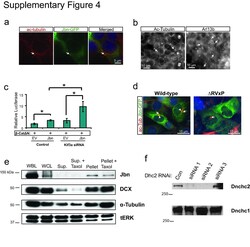
- 4 Supplementary Figure 4. Loss of cilia potentiates Jbn's role in the Wnt pathway a . 293T cells stained for cilia with antibody to acetylated tubulin (red) display clear cilia with Jbn-GFP (green) localized to the basal body similar to previously described in other ciliated cells 2 , 38 . Hoechst was used to stain the nucleus (blue). b . Cilia staining of 293T cells with two independent markers: acetylated tubulin and Arl13b. Arl13b reveals a greater number of cilia (44% of cells) than acetylated tubulin staining (10% of cells). c . Luciferase activity in MEFs transfected with Kif3a siRNA and overexpressed Jbn or EV with activation of the pathway with cotransfected beta-catDeltaN. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
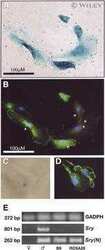
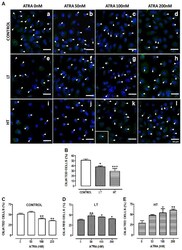
- Figure 8 ATRA increased the number of ciliated cells in HT and LT. ( A ) Immunofluorescence of omentum-derived mesothelial cells (control) and effluent-derived mesothelial cells from LT and HT grown until confluence in the presence of ATRA (0, 50, 100 and 200 nM). Cells were labeled with anti-acetylated alpha-tubulin (green). Nuclei were labeled with ToPro-3 (blue). HT and LT cultures exhibited a reduction in the number of ciliated cells (e and i, respectively). ATRA increased the number of ciliated cells in LT (f, g and h) and HT (j, k and l), being more notable in HT. In control, ATRA decreased the number of ciliated cells. Panel k shows the zoom of a cell with two cilia. (B to E) Quantification of number of ciliated cells. Mean +- SEM of three individual experiments from three different patients are shown. ( B ) * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
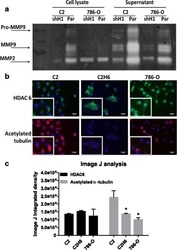
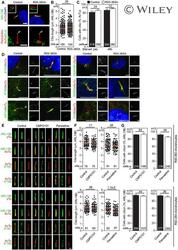
- Fig. 3 HDAC 1 and HDAC 6 increase MMP-2/9 activity and decrease acetylated alpha-tubulin levels, respectively. a ) C2 and 786-0 parental and knocked down HDAC 1 were analyzed for MMP activity by gelatin zymography assay. MMP activity was measured both in cell lysates as well as cell supernatants. b ) Renal tumor cell lines were analyzed for HDAC 6 and acetylated alpha-tubulin expression by immunofluorescence. The staining in red indicates acetylated alpha-tubulin and the staining in green indicates HDAC 6 expression. Scale bar indicates 50 muM distance and images are taken at 20X magnification. c ) Image J analysis measured immunofluorescence by calculating integrated density values of at least three representative fields per cell line. * p < 0.05 indicates statistically significant difference in acetylated alpha-tubulin levels as compared to C2 cells. The error bars represent standard errors from triplicate experiments and p-value was calculated using students t -test
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
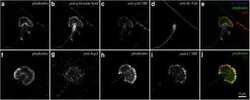
- Figure 5 Data implicating Sept8/ SEPTIN8 in the human tubule hypoxia cell culture model. ( A ) RNAseq values of genes within rat LD block within the RPTEC TERT1 normoxic vs hypoxic conditions. Genes on the right are significant genes. ( B ) TPM values in each of the RNAseq groups for SEPTIN8 in the RPTEC TERT1 cells. ( C ) Expression of SEPTIN8 in single cell datasets (human and mouse). ( D ) Immunohistochemistry from the Human Protein Altas (modified from www.proteinatlas.org/ENSG00000164402-SEPT8/tissue/kidney ) for SEPTIN8 using HPA005665 antibody. Labels are added for tubules and glomerulus. ( E ) Immunofluorescence of SEPTIN8 (red) with acetyl-alpha tubulin, actin, or ZO-1 (green) as labeled. SEPTIN8 localizes with acetyl-alpha tubulin under shear stress with normoxic conditions. ( F ) Under hypoxic conditions, however, SEPTIN8 re-localizes near the actin filaments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
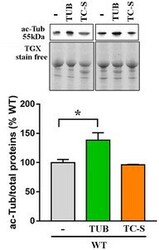
- Figure EV3 Tubulin acetylation is increased by tubacin, but not by the Aurora A inhibitor WT neurons (DIV12) were treated with tubacin (1 muM) or TC-S 7010 (7 nM) for 48 h or left untreated. Acetylated alpha-tubulin (ac-Tub) was measured by Western blot in untreated and tubacin or TC-S-treated cells. Representative bands of acetylated alpha-tubulin are depicted above the histogram, which shows the mean +- SE of the percentage of the expression levels compared to untreated samples ( n = 3, * P < 0.05, one-way ANOVA followed by Dunn's post hoc test). Data are normalized to total protein content, visualized by a TGX stain-free technology.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
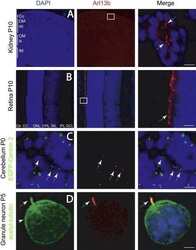
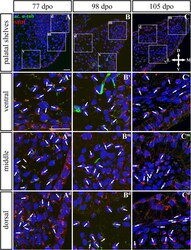
- FIGURE 9 SHH and acetylated alpha-tubulin protein localization in the rostral palatal shelves in pre-hatching chameleons. Immunodetection of SHH (red) and acetylated alpha-tubulin (green) proteins on transversal sections. Lower power pictures overview localization at 77 dpo (A) , 98 dpo (B) , and 105 dpo (C) during pre-hatching development. White rectangles define regions focused on ventral (v), middle (m) and dorsal (d) parts of the palatal shelves. Pictures (A v -C d ) show higher power details from ventral, middle and dorsal regions. White arrowheads indicate polarized colocalization of SHH and primary cilium (detected using acetylated alpha-tubulin) on the same cellular side. Acetylated alpha-tubulin is present not only in primary cilia, but as well in microtubules of mitotic spindle, therefore there was signal in both structures detected in green. Nuclei are counterstained with DRAQ5. Scale bar: 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
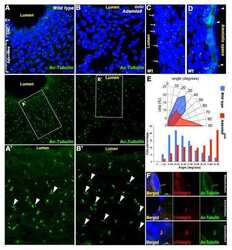
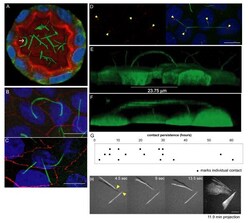
- Figure 2 Prolonged cilia contacts form in cultured cells . ( A ) MDCK cells were cultured in a matrix to form a three-dimensional cyst, then fixed and stained. The arrow indicates an area of overlapping contact (acetylated tubulin - green; Hoechst - blue; actin - red). ( B , C ) MDCK cells were grown on transwell filters, fixed, stained, and imaged using a confocal microscope. These maximum intensity projection images show overlapping ( B ) and point ( C ) contacts between cilia (acetylated tubulin - green; Hoechst - blue; ZO1 - red). ( D) Filter grown MDCK cells were stained and imaged on a confocal microscope (Hoechst - blue, anti-detyrosinated tubulin - green, and anti-gamma tubulin - red). The image presented is a maximum intensity projection (the contrast in the red channel was changed to make the centrosomes more visible). ( E , F ) Filter grown MDCK cells stably expressing tubulin-GFP were imaged by confocal microscopy and used to generate projection images. Cilia from nearby cells appear to be coming together. ( G ) We monitored cells during contact formation and measured how long cilia remained together to determine whether the contacts are transient chance encounters or more persistent inter-cellular interactions. Each point on the graph represents a contact event. The shortest contact duration observed was just over 2 h. ( H ) To test whether primary cilia can move we collected rapid z stacks of primary cilia from TubGFP expressing cells at 4.5 s intervals using a
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 5 Acetylated alpha-tubulin is regulated by HDAC6/ER-alpha interaction in RCC cell lines. a ) HDAC 6, ER-alpha and acetylated alpha-tubulin protein expression were measured by western blot analysis in ccRCC tumors and adjacent non-tumor tissue. b ) A representative ccRCC tumor showed HDAC 6 (in red) and ER-alpha (in green) localization in the cytoplasm. c - d ) Representative immunofluorescent images of acetylated alpha-tubulin (in red) and HDAC 6 (in green) expression in parental and ER-alpha knockdown cell lines are shown. e ) Knockdown of ER-alpha in MCF-7 and renal cell tumors as measured by Western blot analysis. f ) Image J analysis measured immunofluorescence by calculating integrated density values of at least three representative fields per cell line. * p < 0.05 and ** p < 0.01 indicates statistically significant difference of acetylated alpha-tubulin levels in ER-alpha knockdown cells as compared to the parental cell lines. The error bars represent standard errors from triplicate experiments and p-value was calculated using students t -test. g ) C2H6 cells treatment with 10 muM hydroxy tamoxifen and/or 50nM panobinostat for 4 h. Representative immunofluorescence images of acetylated alpha-tubulin (in red), HDAC 6 (in green) and ER-alpha (in green) are shown. h ) Image J analysis measured immunofluorescence by calculating integrated density values of at least three representative fields per cell line. *p < 0.01 indicates statistically different HDAC 6 l
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
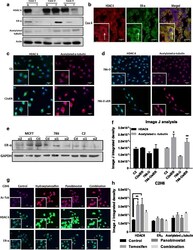
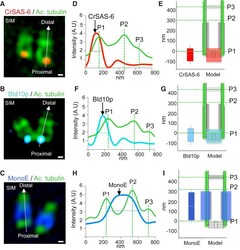
- Figure 2 Localization of Select Centriolar Components (A-C) SIM images of purified Chlamydomonas centrioles stained with acetylated tubulin (green, all panels), highlighting the proximal localization of the cartwheel components CrSAS-6 (A, red), Bld10p (B, blue), and the central core localization of monoglutamylated tubulin (MonoE; C, dark blue). The scale bars represent 100 nm. (D, F, and H) Superimposed plot profiles of acetylated tubulin (green) with CrSAS-6 (red) (D), Bld10p (blue) (F), or monoglutamylated tubulin (dark blue) (MonoE) (H). (E, G, and I) Distribution of CrSAS-6 (E), Bld10p (G), and MonoE (I) as compared to that of acetylated tubulin (left), together with corresponding schematic representations of centriole (right). Heights were as follows: CrSAS-6: 123 +- 20 nm; Bld10p: 133 +- 33 nm; MonoE: 286 +- 33 nm. Error bars represent SD. See also Figure S1 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
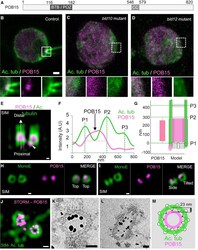
- Figure 4 POB15 Is a Chlamydomonas Central Core Protein (A) Schematic representation of the 820-amino-acid-long POB15 protein highlighting its predicted BTB/POZ and coiled coil (CC) domains. (B-D) Immunofluorescence microscopy of Chlamydomonas control (B), bld10 mutant (C), and bld12 mutant (D) cells stained with antibodies against acetylated tubulin (green) and POB15 (magenta). Here and in other figures, insets are 2-fold magnified views of the region indicated in the lower magnification views, with dashed contours indicating those mutant cases in which centrioles are lacking, shown for visualization purposes, as it is not possible to ascertain where exactly below the plasma membrane centrioles would have been. The scale bar represents 1 mum. (E) SIM image of purified Chlamydomonas centrioles viewed from the side and stained with antibodies against acetylated tubulin (green) and POB15 (magenta). Note the central core localization of the bulk of POB15, as well as the proximal focus (arrowhead). The scale bars represent 100 nm. (F) Superimposed plot profiles of acetylated tubulin (green) and POB15 (magenta). (G) Schematic representation of POB15 localization (height: 229 +- 29 nm; magenta) as compared to that of acetylated tubulin (green). Data are represented as mean +- SD. (H and I) SIM images of centrioles stained with MonoE (green) and POB15 (magenta). The scale bars represent 100 nm. (J) Top view of centrioles imaged using dual SIM/STORM microscopy. Ima
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- EV1 Figure GRK 2 is not required for ciliogenesis or IFT A-C Primary cilia are normal in GRK2 -/- fibroblasts. Cells were serum starved for 24 or 48 h to induce cilia, and (A) immunostained for acetylated tubulin (AcTu, red), ARL13B (green) (upper panel; insets show the individual signals), or AcTu (red), pericentrin (red) and GLI3 (green) (lower panel). Control and GRK2 -/- cilia were positive for AcTu and ARL13B, and showed similar localization of GLI3 to the tips (lower panel, arrows). Acetylated tubulin and pericentrin staining were used to visualize the axoneme and centrioles, respectively. Scale bar, 2 mum. (B) Length distribution of cilia did not differ between control and GRK2 -/- cells. Red bars show medians; dots represent individual cilia. Mann-Whitney U test; number of biological experiments and the total numbers of analyzed cilia are indicated. (C) There was no difference in the efficiency of ciliogenesis between control and GRK2 -/- cells. Mean +- SEM. Welch's t -test; number of biological experiments and the total numbers of analyzed cells are indicated. D Loss of GRK2 did not affect localization of ciliary and IFT components. Immunofluorescence of serum-starved (48 hr) control and GRK2 -/- fibroblasts immunostained with AcTu or detyrosinated tubulin (DetyrTu) to mark the cilium (red), and IFT43, IFT88, KIF3A, TRAF3IP1, WDR34, or ICK (green). No significant differences in staining were found between control and GRK2 -/- cilia, suggesting normal IFT. Scale bars,
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
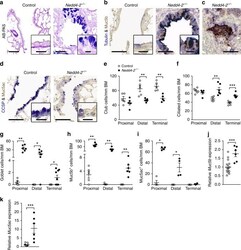
- Fig. 3 Conditional deletion of Nedd4-2 in lung epithelial cells causes peripheral airway remodeling with increased Muc5b production. a - d Representative micrographs and high magnification insets illustrating the airway cell population of terminal bronchioles from conditional Nedd4-2 -/- mice and littermate controls after 3 months of doxycycline induction stained with Alcian blue and periodic acid-Schiff (AB-PAS) (control, n = 5 mice; Nedd4-2 -/- , n = 9 mice) ( a ), or co-stained with anti-Muc5b and anti-acetylated alpha-tubulin antibodies (control, n = 5 mice; Nedd4-2 -/- , n = 6) ( b , c ), or with anti-Muc5ac and anti-CCSP antibodies ( n = 4/group) ( d ). Scale bars, 60 and 15 um (insets). e - i Summary of numeric densities of club cells ( n = 5 mice/group) ( e ), ciliated cells ( n = 6/group and proximal, conditional Nedd4-2 -/- , n = 5 mice) ( f ), goblet cells (proximal, control, n = 5 mice; conditional Nedd4-2 -/- , n = 6 mice; distal, control, n = 5 mice; conditional Nedd4-2 -/- , n = 7 mice, terminal, n = 9/group) ( g ), Muc5b expressing cells (proximal, n = 5/group; distal, control, n = 5 mice; conditional Nedd4-2 -/- , n = 6 mice, terminal, n = 6/group) ( h ), and Muc5ac expressing cells ( n = 4/group) ( i ) in proximal, distal and terminal conducting airways of conditional Nedd4-2 -/- mice and littermate controls after 3 months of doxycycline induction (BM basement membrane). j , k mRNA expression levels of Muc5b ( j ) and Muc5ac (control, n = 15 mice; conditiona
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot