Antibody data
- Antibody Data
- Antigen structure
- References [25]
- Comments [0]
- Validations
- Western blot [3]
- Immunohistochemistry [1]
- Other assay [11]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-698G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PARP1 (cleaved Asp214, Asp215) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody specifically recognizes the 85 kDa fragment of cleaved PARP and can be used as a marker for detecting apoptotic cells. The antibody has been used in western blotting and previous lots have been used in immunostaining applications. Positive controls used in western blotting were Jurkat or HeLa cells treated with staurosporine or etoposide (25 µM for 3 hours).
- Reactivity
- Human, Mouse, Rat, Bovine
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Penta-O-Galloyl-β-D-Glucose in Pistacia integerrima Targets AMPK-ULK1 and ERK/STAT3 Signaling Axes to Induce ROS-Independent Autophagic Cell Death in Human Lung Cancer Cells.
A Metabolomics Investigation of the Metabolic Changes of Raji B Lymphoma Cells Undergoing Apoptosis Induced by Zinc Ions.
Adrenomedullin-CALCRL axis controls relapse-initiating drug tolerant acute myeloid leukemia cells.
Long Non-coding RNA MIAT Knockdown Prevents the Formation of Intracranial Aneurysm by Downregulating ENC1 via MYC.
UHPLC-Q/Orbitrap/MS/MS fingerprinting and antitumoral effects of Prosopis strombulifera (LAM.) BENTH. queous extract on allograft colorectal and melanoma cancer models.
Psoriasin overexpression confers drug resistance to cisplatin by activating ERK in gastric cancer.
The addition of abemaciclib to sunitinib induces regression of renal cell carcinoma xenograft tumors.
Bacterial Carriers for Glioblastoma Therapy.
Disordered IL-33/ST2 activation in decidualizing stromal cells prolongs uterine receptivity in women with recurrent pregnancy loss.
Calpain 3 is important for muscle regeneration: evidence from patients with limb girdle muscular dystrophies.
A gatekeeper residue for NEDD8-activating enzyme inhibition by MLN4924.
Expression of a naturally occurring angiotensin AT(1) receptor cleavage fragment elicits caspase-activation and apoptosis.
BRCA1-IRIS overexpression abrogates UV-induced p38MAPK/p53 and promotes proliferation of damaged cells.
The Bax carboxy-terminal hydrophobic helix does not determine organelle-specific targeting but is essential for maintaining Bax in an inactive state and for stable mitochondrial membrane insertion.
Skin cells and tissue are capable of using L-ergothioneine as an integral component of their antioxidant defense system.
Genistein inhibits radiation-induced activation of NF-kappaB in prostate cancer cells promoting apoptosis and G2/M cell cycle arrest.
CHIR-258: a potent inhibitor of FLT3 kinase in experimental tumor xenograft models of human acute myelogenous leukemia.
Induction of apoptosis in human lung cancer cells by curcumin.
Development of immunomodulatory six base-length non-CpG motif oligonucleotides for cancer vaccination.
RNA interference is a functional pathway with therapeutic potential in human myeloid leukemia cell lines.
Proliferation marker pKi-67 affects the cell cycle in a self-regulated manner.
Peroxisome proliferator-activated receptor gamma ligand troglitazone induces cell cycle arrest and apoptosis of hepatocellular carcinoma cell lines.
Induction of apoptosis and CD10/neutral endopeptidase expression by jaspamide in HL-60 line cells.
Depletion of alveolar macrophages exerts protective effects in pulmonary tuberculosis in mice.
Depletion of alveolar macrophages exerts protective effects in pulmonary tuberculosis in mice.
Balkrishna A, Mulay VP, Verma S, Srivastava J, Lochab S, Varshney A
Frontiers in pharmacology 2022;13:889335
Frontiers in pharmacology 2022;13:889335
A Metabolomics Investigation of the Metabolic Changes of Raji B Lymphoma Cells Undergoing Apoptosis Induced by Zinc Ions.
Yoon N, Lee H, Lee G, Kim EH, Kim SH, Lee JO, Song Y, Park J, Kim SD, Kim Y, Jung BH
Metabolites 2021 Oct 7;11(10)
Metabolites 2021 Oct 7;11(10)
Adrenomedullin-CALCRL axis controls relapse-initiating drug tolerant acute myeloid leukemia cells.
Larrue C, Guiraud N, Mouchel PL, Dubois M, Farge T, Gotanègre M, Bosc C, Saland E, Nicolau-Travers ML, Sabatier M, Serhan N, Sahal A, Boet E, Mouche S, Heydt Q, Aroua N, Stuani L, Kaoma T, Angenendt L, Mikesch JH, Schliemann C, Vergez F, Tamburini J, Récher C, Sarry JE
Nature communications 2021 Jan 18;12(1):422
Nature communications 2021 Jan 18;12(1):422
Long Non-coding RNA MIAT Knockdown Prevents the Formation of Intracranial Aneurysm by Downregulating ENC1 via MYC.
Li X, Zhao H, Liu J, Tong J
Frontiers in physiology 2020;11:572605
Frontiers in physiology 2020;11:572605
UHPLC-Q/Orbitrap/MS/MS fingerprinting and antitumoral effects of Prosopis strombulifera (LAM.) BENTH. queous extract on allograft colorectal and melanoma cancer models.
Persia FA, Troncoso ME, Rinaldini E, Simirgiotis M, Tapia A, Bórquez J, Mackern-Oberti JP, Hapon MB, Gamarra-Luques C
Heliyon 2020 Feb;6(2):e03353
Heliyon 2020 Feb;6(2):e03353
Psoriasin overexpression confers drug resistance to cisplatin by activating ERK in gastric cancer.
Li L, Cui Y, Ye L, Zhao Z, Jiang WG, Ji J
International journal of oncology 2018 Sep;53(3):1171-1182
International journal of oncology 2018 Sep;53(3):1171-1182
The addition of abemaciclib to sunitinib induces regression of renal cell carcinoma xenograft tumors.
Small J, Washburn E, Millington K, Zhu J, Holder SL
Oncotarget 2017 Nov 10;8(56):95116-95134
Oncotarget 2017 Nov 10;8(56):95116-95134
Bacterial Carriers for Glioblastoma Therapy.
Mehta N, Lyon JG, Patil K, Mokarram N, Kim C, Bellamkonda RV
Molecular therapy oncolytics 2017 Mar 17;4:1-17
Molecular therapy oncolytics 2017 Mar 17;4:1-17
Disordered IL-33/ST2 activation in decidualizing stromal cells prolongs uterine receptivity in women with recurrent pregnancy loss.
Salker MS, Nautiyal J, Steel JH, Webster Z, Sućurović S, Nicou M, Singh Y, Lucas ES, Murakami K, Chan YW, James S, Abdallah Y, Christian M, Croy BA, Mulac-Jericevic B, Quenby S, Brosens JJ
PloS one 2012;7(12):e52252
PloS one 2012;7(12):e52252
Calpain 3 is important for muscle regeneration: evidence from patients with limb girdle muscular dystrophies.
Hauerslev S, Sveen ML, Duno M, Angelini C, Vissing J, Krag TO
BMC musculoskeletal disorders 2012 Mar 23;13:43
BMC musculoskeletal disorders 2012 Mar 23;13:43
A gatekeeper residue for NEDD8-activating enzyme inhibition by MLN4924.
Toth JI, Yang L, Dahl R, Petroski MD
Cell reports 2012 Apr 19;1(4):309-16
Cell reports 2012 Apr 19;1(4):309-16
Expression of a naturally occurring angiotensin AT(1) receptor cleavage fragment elicits caspase-activation and apoptosis.
Cook JL, Singh A, DeHaro D, Alam J, Re RN
American journal of physiology. Cell physiology 2011 Nov;301(5):C1175-85
American journal of physiology. Cell physiology 2011 Nov;301(5):C1175-85
BRCA1-IRIS overexpression abrogates UV-induced p38MAPK/p53 and promotes proliferation of damaged cells.
Chock K, Allison JM, Elshamy WM
Oncogene 2010 Sep 23;29(38):5274-85
Oncogene 2010 Sep 23;29(38):5274-85
The Bax carboxy-terminal hydrophobic helix does not determine organelle-specific targeting but is essential for maintaining Bax in an inactive state and for stable mitochondrial membrane insertion.
Brock SE, Li C, Wattenberg BW
Apoptosis : an international journal on programmed cell death 2010 Jan;15(1):14-27
Apoptosis : an international journal on programmed cell death 2010 Jan;15(1):14-27
Skin cells and tissue are capable of using L-ergothioneine as an integral component of their antioxidant defense system.
Markova NG, Karaman-Jurukovska N, Dong KK, Damaghi N, Smiles KA, Yarosh DB
Free radical biology & medicine 2009 Apr 15;46(8):1168-76
Free radical biology & medicine 2009 Apr 15;46(8):1168-76
Genistein inhibits radiation-induced activation of NF-kappaB in prostate cancer cells promoting apoptosis and G2/M cell cycle arrest.
Raffoul JJ, Wang Y, Kucuk O, Forman JD, Sarkar FH, Hillman GG
BMC cancer 2006 Apr 26;6:107
BMC cancer 2006 Apr 26;6:107
CHIR-258: a potent inhibitor of FLT3 kinase in experimental tumor xenograft models of human acute myelogenous leukemia.
Lopes de Menezes DE, Peng J, Garrett EN, Louie SG, Lee SH, Wiesmann M, Tang Y, Shephard L, Goldbeck C, Oei Y, Ye H, Aukerman SL, Heise C
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Jul 15;11(14):5281-91
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Jul 15;11(14):5281-91
Induction of apoptosis in human lung cancer cells by curcumin.
Radhakrishna Pillai G, Srivastava AS, Hassanein TI, Chauhan DP, Carrier E
Cancer letters 2004 May 28;208(2):163-70
Cancer letters 2004 May 28;208(2):163-70
Development of immunomodulatory six base-length non-CpG motif oligonucleotides for cancer vaccination.
Filion MC, Filion B, Roy J, Ménard S, Reader S, Phillips NC
Vaccine 2004 Jun 23;22(19):2480-8
Vaccine 2004 Jun 23;22(19):2480-8
RNA interference is a functional pathway with therapeutic potential in human myeloid leukemia cell lines.
Cioca DP, Aoki Y, Kiyosawa K
Cancer gene therapy 2003 Feb;10(2):125-33
Cancer gene therapy 2003 Feb;10(2):125-33
Proliferation marker pKi-67 affects the cell cycle in a self-regulated manner.
Schmidt MH, Broll R, Bruch HP, Duchrow M
Journal of cellular biochemistry 2002;87(3):334-41
Journal of cellular biochemistry 2002;87(3):334-41
Peroxisome proliferator-activated receptor gamma ligand troglitazone induces cell cycle arrest and apoptosis of hepatocellular carcinoma cell lines.
Yoshizawa K, Cioca DP, Kawa S, Tanaka E, Kiyosawa K
Cancer 2002 Nov 15;95(10):2243-51
Cancer 2002 Nov 15;95(10):2243-51
Induction of apoptosis and CD10/neutral endopeptidase expression by jaspamide in HL-60 line cells.
Cioca DP, Kitano K
Cellular and molecular life sciences : CMLS 2002 Aug;59(8):1377-87
Cellular and molecular life sciences : CMLS 2002 Aug;59(8):1377-87
Depletion of alveolar macrophages exerts protective effects in pulmonary tuberculosis in mice.
Leemans JC, Juffermans NP, Florquin S, van Rooijen N, Vervoordeldonk MJ, Verbon A, van Deventer SJ, van der Poll T
Journal of immunology (Baltimore, Md. : 1950) 2001 Apr 1;166(7):4604-11
Journal of immunology (Baltimore, Md. : 1950) 2001 Apr 1;166(7):4604-11
Depletion of alveolar macrophages exerts protective effects in pulmonary tuberculosis in mice.
Leemans JC, Juffermans NP, Florquin S, van Rooijen N, Vervoordeldonk MJ, Verbon A, van Deventer SJ, van der Poll T
Journal of immunology (Baltimore, Md. : 1950) 2001 Apr 1;166(7):4604-11
Journal of immunology (Baltimore, Md. : 1950) 2001 Apr 1;166(7):4604-11
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of THP1-Nuclear Enriched (lane 1), HeLa-Nuclear Enriched (lane 2), KARPAS-299 (lane 3) and Daudi (lane 4). The blots were probed with Anti-PARP Rabbit Polyclonal Antibody (Product # 44-698G, 1:500-1:2000 dilution) and detected by chemiluminescence oat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.4 µg/mL, 1:2500 dilution). A 113 kDa band corresponding to PARP was observed across cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
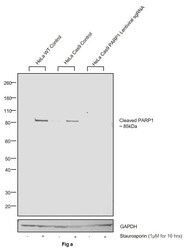
- Experimental details
- CRISPR-Cas9 mediated genome editing ofPARP1 (as confirmed by next generation sequencing) was achieved by using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR978664_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Fig (a) Western blot analysis of PARP1 was performed by loading 30 µg of HeLa Wild Type (Lane 1), HeLa Wild Type treated with Staurosporin (1 µM for 16 hrs) (Lane 2), HeLa Cas9 (Lane 3), HeLa Cas9 treated with Staurosporin (1 µM for 16 hrs) (Lane 4), HeLa Cas9 cells transduced with PARP1 Lentiviral sgRNA (Lane 5) and HeLa Cas9 cells transduced with PARP1 Lentiviral sgRNA treated with Staurosporin (1 µM for 16 hrs) (Lane 6) whole cell extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-PARP1 (cleaved Asp214, Asp215) Polyclonal Antibody (Product # 44-698G) using 1:1,000 dilution and Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036 1:4,000 dilution). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).Even though NGS analysis determine the clone as partial KO, there was complete loss of signal in sgRNA transduced cells using the LentiArray™ CRISP
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-PARP (Cleaved Asp214, Asp215) Polyclonal Antibody (Product # 44-698G) and a 85 kDa band corresponding to PARP (Cleaved Asp214, Asp215) was observed across all cell lines. Nuclear enriched extracts (40 µg lysate) of Jurkat (Lane 1), Jurkat treated with Etoposide (1 µM for 16 hours) (Lane 2), HeLa (Lane 3), HeLa treated with Etoposide (1 µM for 16 hours) (Lane 4), HeLa (Lane 5), HeLa treated with Staurosporine (3 µM for 16 hours) (Lane 6) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
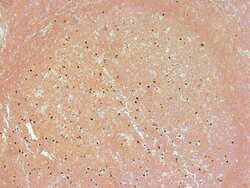
- Experimental details
- PARP (214⁄215) cleavage site specific antibody. Image shows immunohistochemical staining of cleaved PARP in formalin-fixed, paraffin embedded human tonsillar tissue. Antigen retrieval done using citrate buffer.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
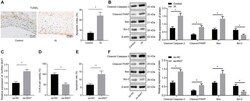
- Experimental details
- Figure 5 Immunohistochemistry of p53-Mediated Apoptosis after Bacterial Carrier Therapy (A) IHC images of brain sections from control and treated non-responder rats to check for expression of p53. Green, human anti-mitochondria antibodies that specifically stain tumor cells derived from U87MG human tumor cell line. Blue, DAPI; and red, p53. More p53 observed within tumor of treated non-responder rats compared to control. (B) Quantification of IHC images showed greater p53 levels in treated non-responder rats compared to control rats with p value of 0.0735. Nine images were taken from each control (n = 4) and treated non-responder rat (n = 4) for this analysis. (C) IHC images of brain sections from control and treated non-responder rats to check for presence of cleaved PARP (indicator of apoptosis). Green, human anti-mitochondria antibodies; blue, DAPI; and red, cleaved PARP. There were more PARP-positive cells in treated non-responder rats compared to control. (D) Quantification of IHC images showed more cleaved PARP in treated non-responder rats compared to controls with p value of 0.6944. Nine images were taken from each control (n = 4) and treated non-responder rat (n = 4) for this analysis. (E) TUNEL staining to check for apoptosis within tumor of control and treated non-responder rats. Green, anti-mitochondria antibodies; blue, DAPI; and red, TUNEL-positive cells. There were more TUNEL-positive cells in the tumor region of treated non-responders compared to control. (F)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
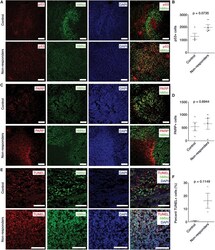
- Experimental details
- FIGURE 2 The endothelial cell apoptosis can be induced by overexpression of MIAT in vitro . (A) Representative images of TUNEL staining (x200) and quantitative analysis of TUNEL-positive cells in patients with IA. (B) The expression of cleaved Caspase-3, cleaved PARP1, Bax, and Bcl-2 in patients with IA measured by Western blot analysis. (C) The expression of MIAT in endothelial cells after overexpressing MIAT determined by RT-qPCR. (D) Viability of endothelial cells in response to oe-MIAT detected by CCK-8 assay. (E) Flow cytometry analysis of apoptosis of endothelial cells in response to oe-MIAT. (F) The expression of apoptosis-related factors (cleaved Caspase-3, cleaved PARP1, Bax, and Bcl-2) in response to oe-MIAT measured by Western blot analysis. * p < 0.05. The measurement data were expressed as mean +- standard derivation. Comparisons between two groups were analyzed by unpaired t test, and the cell experiment was conducted three times independently.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 4 ENC1 expression is augmented in IA patients, and ENC1 silencing negates MIAT-induced endothelial cell apoptosis. (A) ENC1 expression in ruptured IA, unruptured IA and control samples determined by RT-qPCR. (B) Analysis of the correlation between ENC1 expression and MIAT expression. (C) The correlation between the expression of ENC1 and disease-free survival of patients with IA. (D) The correlation between the expression of ENC1 and overall survival of patients with IA. (E) The protein expression of ENC1 in vascular endothelial cells in vitro in response to oe-MIAT and sh-ENC1 alone or in combination measured by Western blot analysis. (F) Flow cytometry analysis for apoptosis of endothelial cells in response to oe-MIAT and sh-ENC1 alone or in combination. (G) The expression of apoptosis-related factors (cleaved Caspase-3, cleaved PARP1, Bax, and Bcl-2) in response to oe-MIAT and sh-ENC1 alone or in combination measured by Western blot analysis. * p < 0.05. The measurement data were expressed as mean +- standard derivation. Comparisons between two groups were analyzed by unpaired t test, while the correlation between the expression of ENC1 in patients with IA and their disease-free survival and overall survival was analyzed by Kaplan-Meier method (long-rank test). Comparisons among multiple groups were analyzed by one-way ANOVA followed by Tukey's post hoc test, and the cell experiment was conducted three times independently.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 MIAT silencing inhibits endothelial cell apoptosis and maintains endothelial cell continuity in IA rats. Rats were induced with IA model and treated with sh-MIAT or sh-NC, with the sham-operated rats as control. (A) The expression of MIAT in rats determined by RT-qPCR. (B) HE staining (x 200) for the ACA/OA branch sections of rats. (C) The representative images of the IA wall of ACA/OA branch of rats (x200) (arrows indicate the arterial wall). (D) Quantitative analysis of cell apoptosis in rats assessed by TUNEL staining. (E) The expression of apoptosis-related factors (cleaved Caspase-3, cleaved PARP1, Bax, and Bcl-2) as determined by Western blot analysis and quantified by the Image J software. * p < 0.05. The measurement data were expressed as mean +- standard derivation. Comparisons among multiple groups were analyzed by one-way ANOVA followed by Tukey's post hoc test, and the cell experiment was conducted three times independently. The sham group: n = 12, the IA + sh-NC group: n = 12, and the IA + sh-MIAT group: n = 6.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- FIGURE 6 Silencing ENC1 reduces endothelial cell apoptosis in vivo to protect rats from IA. Rats were induced with IA model and treated with sh-ENC1 or sh-NC, with the sham-operated rats as control. (A) The expression of ENC1 in rats determined by RT-qPCR. (B) HE staining (x200) for the ACA/OA branch sections of rats. (C) The representative images of TUNEL staining (x200) for apoptosis of rats (arrows indicate the arterial wall). (D) Quantitative analysis of cell apoptosis in rats evaluated by TUNEL staining. (E) The expression of apoptosis-related factors (cleaved Caspase-3, cleaved PARP1, Bax, and Bcl-2) as determined by Western blot analysis and quantified by Image J software. * p < 0.05. The measurement data were expressed as mean +- standard derivation. Comparisons between two groups were analyzed by unpaired t test, and comparisons among multiple groups were analyzed by one-way ANOVA and followed by Tukey's post hoc test. The cell experiment was conducted three times independently. The sham group: n = 12, the IA + sh-NC group: n = 12, and the IA + sh-ENC1 group: n = 6.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
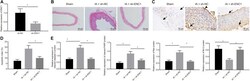
- FIGURE 3 Pistacia integerrima potentiates ROS-independent apoptosis in lung cancer cells. (A) Whole cell lysates of A549 cells post 24 h of treatment at indicated doses were subjected to Western blotting using antibodies against cleaved PARP1 and cleaved caspase 3. beta -Actin served as the loading control. (B) Whole cell lysates of A549 cells treated at 10 mug/ml of PI for 6, 12, and 24 h time period was subjected to Western blotting using antibodies against cleaved PARP1 and cleaved caspase 3. beta -Actin served as a loading control. (C) A549 cells treated at indicated doses of PI for 24 h were stained with DAPI and imaged under a fluorescent microscope. (D) A549 cells post PI treatment for 24 h were stained with DCFDA for 45 min. Fluorescent units were measured using a multi-plate reader and were expressed in relative fold change with respect to untreated. The fold change difference in PI treatments was not-significant (ns) with respect to untreated. (E) DCFDA fluorescence expressed as percent max count was determined in A549 cells with indicated treatments for 24 h using a flow cytometer. H 2 O 2 served as a positive control for DCFDA fluorescence. Error bars represent mean +- SEM; significance of data represented as ** p < 0.005 and # p < 0.0001 and not significant (ns) if p > 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 Bioactivity-guided fractionation of Pistacia integerrima identified penta-O-galloyl- beta -D-glucose (PGG) as a bioactive phytocompound . (A) Chromatogram of total ion count (TIC) versus retention time (min) shows peaks labeled with arrows and numbers. Peaks representing the phytometabolite were identified by UPLC/QToF-MS and detailed in Table 1 . (B) Schematic showing fractionation of PI extract through column chromatography. A total of 34 aliquots were collected as fractions 66,67,68, 69, and 70. UPLC/QToF-MS generated the chromatograms for all the fractions. Peaks 22 and 23 (arrow labeled) corresponding to 1,2,3,4,6-Pentagalloyl Glucose (PGG) and 2-O-digalloyl-1,3,4,6-tetra-O-beta-D-galloyl Glucose were not detected in fraction 66 and 67. (C) Percent cell viability was evaluated in A549 cells treated with different fractions at 10 mug/ml for 24 h. Alamar blue assay determined the percent viability in all treatments ( n = 4). Significance in percent viability in all treatments is calculated with respect to PI. Error bars represent mean +- SEM; significance of data represented as * p < 0.05 and not significant (ns) if p > 0.05. (D) Whole cell lysates of A549 cells, post 24 h of treatments at 10 mug/ml, were subjected to Western blotting using antibodies against cleaved PARP1. beta -Actin served as a loading control. (E) Overlayed chromatogram generated through UHPLC showing the absorbance and retention time of standards, fractions, and PI.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
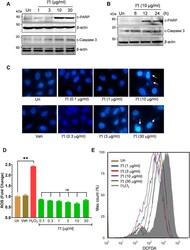
- FIGURE 6 Penta-O-galloyl- beta -D-glucose (PGG) activates AMPK and ERK and inhibits STAT3 signaling in lung cancer cells. (A) Percent cell viability calculated in A549 cells treated for 24 h with 5 and 10 muM of PGG alone or in presence of PI at indicated concentrations. Significance in PI + 5 muM PGG and PI + 10 muM PGG was calculated with respect to PI alone ( n = 3). Error bars represent mean +- SEM; significance of data represented as * p < 0.05, ** p < 0.005, *** p < 0.0005, and # p < 0.0001 and not significant (ns) if p > 0.05. (B) A549 cells post PGG treatment for 24 h were stained with DCFDA for 45 min. Fluorescent units were measured using a multi-plate reader and were expressed in relative fold change with respect to untreated. The fold change difference in PI treatments was non-significant (ns) with respect to untreated. H 2 O 2 served as a positive control for DCFDA fluorescence. Error bars represent mean +- SEM; significance of data represented as # p < 0.0001 and not significant (ns) if p > 0.05. (C) Whole cell lysates of A549 cells, at indicated treatments of PI (10 mug/ml) and PGG (10 muM), were subjected to Western blotting using antibodies against cleaved PARP1 and cleaved caspase 3. beta -Actin served as the loading control. (D) Whole cell lysates of A549 cells, treated with PI (10 mug/ml) and PGG (10 muM), were subjected to Western blotting using antibodies against LC3A/B, pAMPK, pERK1/2, pSTAT3(Y705), STAT3, pULK1(S317), and pULK1(S757). beta -Actin serve
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
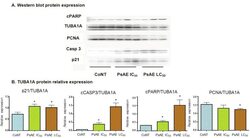
- Figure 6 Western blot analysis of cultured B16-F0 cells. A. After 24 h, protein expression of control, IC 50 and LC 50 PsAE treated cells (0, 2.5 and 9 mug/mL, respectively) are presented. B. Relative expression of p21, caspase 3, PCNA and cPARP with respect to TUBA1A are expressed in terms of mean +- SEM of three assays by triplicated. Results were compared by one-way ANOVA followed by Fisher LSD test. *: indicate significant differences (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
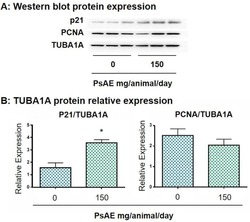
- Figure 7 Western blot analysis of melanoma tumors. A. After 22 days of tumor induction, control and PsAE treated (150 mg/animal/day) groups are presented. B. Relative expression of p21 and PCNA over TUBA1A are expressed in terms of mean +- SEM. Results were compared by Student T test. *: indicates p = 0.0296. Full, non-adjusted images of p21 blot is provided in Supplementary Figure 5; TUBA1A (upper line) and PCNA (lower line) are provided in Supplementary Figure 6. Figure 7
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot