Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- OPA1-04050 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Tyrosine Hydroxylase Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Purifed from natural sources
- Description
- In Western blot, this antibody detects a single ~60 kDa protein representing tyrosine hydroxylase from rat brain lysates of PC-12 cells stimulated by okadaic acid. Immunohistochemical staining of TH in human brain with OPA1-04050 results in intense labeling of the dopaminergic neurons in the substantia nigra.
Submitted references Identifying dominant-negative actions of a dopamine transporter variant in patients with parkinsonism and neuropsychiatric disease.
Unbiased identification of novel transcription factors in striatal compartmentation and striosome maturation.
Locus coeruleus spiking differently correlates with S1 cortex activity and pupil diameter in a tactile detection task.
Failure of Diphtheria Toxin Model to Induce Parkinson-Like Behavior in Mice.
Systemically administered neuregulin-1β1 rescues nigral dopaminergic neurons via the ErbB4 receptor tyrosine kinase in MPTP mouse models of Parkinson's disease.
Transcriptional and structural plasticity of tyrosine hydroxylase expressing neurons in both striatum and nucleus accumbens following dopaminergic denervation.
Brain-resident microglia predominate over infiltrating myeloid cells in activation, phagocytosis and interaction with T-lymphocytes in the MPTP mouse model of Parkinson disease.
Changes in serotoninergic and noradrenergic descending pain pathways during painful diabetic neuropathy: the preventive action of IGF1.
The L-type channel antagonist isradipine is neuroprotective in a mouse model of Parkinson's disease.
Cell type specific sequestration of choline acetyltransferase and tyrosine hydroxylase within Lewy bodies.
Incidental Lewy body disease and preclinical Parkinson disease.
Herborg F, Jensen KL, Tolstoy S, Arends NV, Posselt LP, Shekar A, Aguilar JI, Lund VK, Erreger K, Rickhag M, Lycas MD, Lonsdale MN, Rahbek-Clemmensen T, Sørensen AT, Newman AH, Løkkegaard A, Kjærulff O, Werge T, iPSYCH researchers, Møller LB, Matthies HJ, Galli A, Hjermind LE, Gether U
JCI insight 2021 Sep 22;6(18)
JCI insight 2021 Sep 22;6(18)
Unbiased identification of novel transcription factors in striatal compartmentation and striosome maturation.
Cirnaru MD, Song S, Tshilenge KT, Corwin C, Mleczko J, Galicia Aguirre C, Benlhabib H, Bendl J, Apontes P, Fullard J, Creus-Muncunill J, Reyahi A, Nik AM, Carlsson P, Roussos P, Mooney SD, Ellerby LM, Ehrlich ME
eLife 2021 Oct 5;10
eLife 2021 Oct 5;10
Locus coeruleus spiking differently correlates with S1 cortex activity and pupil diameter in a tactile detection task.
Yang H, Bari BA, Cohen JY, O'Connor DH
eLife 2021 Mar 15;10
eLife 2021 Mar 15;10
Failure of Diphtheria Toxin Model to Induce Parkinson-Like Behavior in Mice.
Valek L, Tegeder I
International journal of molecular sciences 2021 Aug 31;22(17)
International journal of molecular sciences 2021 Aug 31;22(17)
Systemically administered neuregulin-1β1 rescues nigral dopaminergic neurons via the ErbB4 receptor tyrosine kinase in MPTP mouse models of Parkinson's disease.
Depboylu C, Rösler TW, de Andrade A, Oertel WH, Höglinger GU
Journal of neurochemistry 2015 May;133(4):590-7
Journal of neurochemistry 2015 May;133(4):590-7
Transcriptional and structural plasticity of tyrosine hydroxylase expressing neurons in both striatum and nucleus accumbens following dopaminergic denervation.
Depboylu C, Klietz M, Maurer L, Oertel WH, Kobayashi K, Weihe E, Höglinger GU, Schäfer MK
Journal of chemical neuroanatomy 2014 Nov;61-62:169-75
Journal of chemical neuroanatomy 2014 Nov;61-62:169-75
Brain-resident microglia predominate over infiltrating myeloid cells in activation, phagocytosis and interaction with T-lymphocytes in the MPTP mouse model of Parkinson disease.
Depboylu C, Stricker S, Ghobril JP, Oertel WH, Priller J, Höglinger GU
Experimental neurology 2012 Dec;238(2):183-91
Experimental neurology 2012 Dec;238(2):183-91
Changes in serotoninergic and noradrenergic descending pain pathways during painful diabetic neuropathy: the preventive action of IGF1.
Morgado C, Silva L, Pereira-Terra P, Tavares I
Neurobiology of disease 2011 Jul;43(1):275-84
Neurobiology of disease 2011 Jul;43(1):275-84
The L-type channel antagonist isradipine is neuroprotective in a mouse model of Parkinson's disease.
Ilijic E, Guzman JN, Surmeier DJ
Neurobiology of disease 2011 Aug;43(2):364-71
Neurobiology of disease 2011 Aug;43(2):364-71
Cell type specific sequestration of choline acetyltransferase and tyrosine hydroxylase within Lewy bodies.
Dugger BN, Dickson DW
Acta neuropathologica 2010 Nov;120(5):633-9
Acta neuropathologica 2010 Nov;120(5):633-9
Incidental Lewy body disease and preclinical Parkinson disease.
DelleDonne A, Klos KJ, Fujishiro H, Ahmed Z, Parisi JE, Josephs KA, Frigerio R, Burnett M, Wszolek ZK, Uitti RJ, Ahlskog JE, Dickson DW
Archives of neurology 2008 Aug;65(8):1074-80
Archives of neurology 2008 Aug;65(8):1074-80
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of Tyrosine Hydroxylase in rat striatal lysate (10 µg). Showing specific immunolabeling of a ~60 kDa band corresponding to Tyrosine Hydroxylase polyclonal antibody (Product # OPA1-04050).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Tyrosine Hydroxylase Polyclonal Antibody (Product # OPA1-04050) and 58 kDa band corresponding to Tyrosine Hydroxylase was observed. Dexamethasone, a synthetic glucocorticoid, is known to increase the expression of Tyrosine Hydroxylase in neuronal cells. Whole cell extracts (30 µg lysate) of PC-12 (Lane1) and PC-12 treated with dexamethasone (5ug/ml for 6 days) (Lane2) were electrophoresed using Novex® NuPAGE® 4-12% Bis-Tris gel (Product # NP0342BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
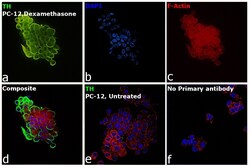
- Experimental details
- Immunofluorescence analysis of Anti-Tyrosine Hydroxylase Polyclonal Antibody (Product # OPA1-04050) was performed using PC-12 cells treated with 1 µg/mL dexamethasone for 6 days. The cells were fixed with 4% Paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 10 minutes at room temperature. The cells were labeled with Tyrosine Hydroxylase Polyclonal Antibody (Product # OPA1-04050) at 1:500 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034, 1:2000 dilution) for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Cytoplasmic localization. Panel e represents untreated PC-12 cells having low expression of TH. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
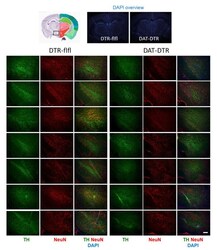
- Experimental details
- Figure 2 Immunofluorescence analysis of tyrosine hydroxylase (TH) as a marker for DA neurons in the substantia nigra (SN). The pan-neuronal marker NeuN was used to label all neurons. DAPI was used as counterstain of nuclei. Mice were treated with 5 x 0.1 ng/g DTx. Mice were 9-13 weeks old at the onset of DTx and were sacrificed 6-7 weeks after the last DTx dose. The image shows examples of n = 4-6 mice per group (one side or both sides). Scale bar 200 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
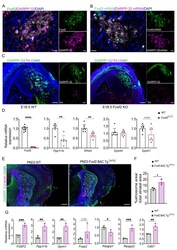
- Experimental details
- Figure 5. Foxf2 is required for striosome compartmentation. ( A ) Representative confocal micrographs of Foxf2 (green), DARPP-32 (magenta) and DAPI (blue) immunolabeling on 16-um-thick coronal sections from wild-type PND3 showing the localization of Foxf2 within the DARPP-32-immunopositive striosomes. Scale bars correspond to 20 um. ( B ) RNAscope analysis in 16-um-thick coronal sections from wild-type PND3 mice shows Foxf2 mRNA (green) co-localization with Ppp1r1b /DARPP-32 mRNA (magenta). Arrow heads indicate vascular localization of Foxf2 mRNA, with absence of DARPP-32 mRNA. Scale bars = 20 um. ( C ) DARPP-32 (green) and tyrosine hydroxylase (TH) (magenta) immunostaining in sagittal sections of E18.5 WT and Foxf2 KO mice show the absence of striosome assembly in Foxf2 KO mice. Scale bars = 100 um ( D ) RT-qPCR from E18.5 Foxf2 WT and littermates striatal RNA indicating that Foxf2 expression was abolished in the mouse and that the levels of Ppp1r1b and Efna5 mRNA were significantly reduced by Foxf2 deletion. n=7 Foxf2 -/- and n=9 WT. Unpaired t-test **p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 DAT-K619N exerts a dominant-negative effect on DAT-WT. ( A ) Evaluation of dominant-negative effects of DAT-K619N in vitro. DA uptake was measured in HEK293 cells, cotransfected with equal amounts (1.5 mug) of DAT-K619N and DAT-WT, and compared with cells transfected only with DAT-WT (1.5 mug + 1.5 mug empty vector), DAT-K619N (1.5 mug + 1.5 mug empty vector), or with 3 mug of DAT-WT as a control. Each experiment was performed in triplicate and normalized to V max of DAT-WT (1.5 mug + 1.5 mug empty vector). The V max of DAT-K619N/DAT-WT cotransfected cells (1.5 mug + 1.5 mug) was reduced relative to DAT-WT (1.5 mug + 1.5 mug empty vector) to a level similar to DAT-K619N alone (* P < 0.016, 1-sample 2-tailed t test, Bonferroni-adjusted significance level [= 0.05/3] n = 4-7) indicating dominant-negative actions of hDAT-K619N. ( B ) Evaluation of dominant-negative effects in vivo performed by overexpressing HA-hDAT-WT or HA-hDAT-K619N selectively in dopaminergic neurons, using bilateral midbrain AAV injections in TH-Cre mice, and performing [ 3 H]-DA uptake on striatal synaptosomes. Injections with AAV encoding mCherry was used for comparison with endogenous DA uptake levels. Uptake curves are average curves of 4 experiments each performed in triplicate. Quantification of V max normalized to mCherry showed HA-hDAT-K619N but not HA-hDAT-WT reduced DA uptake below endogenous uptake capacity (* P < 0.005, 1-sample 2-tailed t test, Bonferroni-adjusted significance level: **
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot