Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-882G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-c-Met (Tyr1003) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Suppressive impact of metronomic chemotherapy using UFT and/or cyclophosphamide on mediators of breast cancer dissemination and invasion.
Hypoxia-Driven Mechanism of Vemurafenib Resistance in Melanoma.
Targeting c-Met in melanoma: mechanism of resistance and efficacy of novel combinatorial inhibitor therapy.
Alternative signaling pathways as potential therapeutic targets for overcoming EGFR and c-Met inhibitor resistance in non-small cell lung cancer.
Anti-c-MET Nanobody - a new potential drug in multiple myeloma treatment.
Expression of hepatocyte epidermal growth factor receptor, FAS and glypican 3 in EpCAM-positive regenerative clusters of hepatocytes, cholangiocytes, and progenitor cells in human liver failure.
MET and phosphorylated MET as potential biomarkers in lung cancer.
MET and phosphorylated MET as potential biomarkers in lung cancer.
The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma.
Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274 and small interfering RNA in non-small cell lung cancer.
A selective small molecule inhibitor of c-Met kinase inhibits c-Met-dependent phenotypes in vitro and exhibits cytoreductive antitumor activity in vivo.
Muñoz R, Hileeto D, Cruz-Muñoz W, Wood GA, Xu P, Man S, Viloria-Petit A, Kerbel RS
PloS one 2019;14(9):e0222580
PloS one 2019;14(9):e0222580
Hypoxia-Driven Mechanism of Vemurafenib Resistance in Melanoma.
Qin Y, Roszik J, Chattopadhyay C, Hashimoto Y, Liu C, Cooper ZA, Wargo JA, Hwu P, Ekmekcioglu S, Grimm EA
Molecular cancer therapeutics 2016 Oct;15(10):2442-2454
Molecular cancer therapeutics 2016 Oct;15(10):2442-2454
Targeting c-Met in melanoma: mechanism of resistance and efficacy of novel combinatorial inhibitor therapy.
Etnyre D, Stone AL, Fong JT, Jacobs RJ, Uppada SB, Botting GM, Rajanna S, Moravec DN, Shambannagari MR, Crees Z, Girard J, Bertram C, Puri N
Cancer biology & therapy 2014 Sep;15(9):1129-41
Cancer biology & therapy 2014 Sep;15(9):1129-41
Alternative signaling pathways as potential therapeutic targets for overcoming EGFR and c-Met inhibitor resistance in non-small cell lung cancer.
Fong JT, Jacobs RJ, Moravec DN, Uppada SB, Botting GM, Nlend M, Puri N
PloS one 2013;8(11):e78398
PloS one 2013;8(11):e78398
Anti-c-MET Nanobody - a new potential drug in multiple myeloma treatment.
Slørdahl TS, Denayer T, Moen SH, Standal T, Børset M, Ververken C, Rø TB
European journal of haematology 2013 Nov;91(5):399-410
European journal of haematology 2013 Nov;91(5):399-410
Expression of hepatocyte epidermal growth factor receptor, FAS and glypican 3 in EpCAM-positive regenerative clusters of hepatocytes, cholangiocytes, and progenitor cells in human liver failure.
Hattoum A, Rubin E, Orr A, Michalopoulos GK
Human pathology 2013 May;44(5):743-9
Human pathology 2013 May;44(5):743-9
MET and phosphorylated MET as potential biomarkers in lung cancer.
Tretiakova M, Salama AK, Karrison T, Ferguson MK, Husain AN, Vokes EE, Salgia R
Journal of environmental pathology, toxicology and oncology : official organ of the International Society for Environmental Toxicology and Cancer 2011;30(4):341-54
Journal of environmental pathology, toxicology and oncology : official organ of the International Society for Environmental Toxicology and Cancer 2011;30(4):341-54
MET and phosphorylated MET as potential biomarkers in lung cancer.
Tretiakova M, Salama AK, Karrison T, Ferguson MK, Husain AN, Vokes EE, Salgia R
Journal of environmental pathology, toxicology and oncology : official organ of the International Society for Environmental Toxicology and Cancer 2011;30(4):341-54
Journal of environmental pathology, toxicology and oncology : official organ of the International Society for Environmental Toxicology and Cancer 2011;30(4):341-54
The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma.
Seiwert TY, Jagadeeswaran R, Faoro L, Janamanchi V, Nallasura V, El Dinali M, Yala S, Kanteti R, Cohen EE, Lingen MW, Martin L, Krishnaswamy S, Klein-Szanto A, Christensen JG, Vokes EE, Salgia R
Cancer research 2009 Apr 1;69(7):3021-31
Cancer research 2009 Apr 1;69(7):3021-31
Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274 and small interfering RNA in non-small cell lung cancer.
Ma PC, Jagadeeswaran R, Jagadeesh S, Tretiakova MS, Nallasura V, Fox EA, Hansen M, Schaefer E, Naoki K, Lader A, Richards W, Sugarbaker D, Husain AN, Christensen JG, Salgia R
Cancer research 2005 Feb 15;65(4):1479-88
Cancer research 2005 Feb 15;65(4):1479-88
A selective small molecule inhibitor of c-Met kinase inhibits c-Met-dependent phenotypes in vitro and exhibits cytoreductive antitumor activity in vivo.
Christensen JG, Schreck R, Burrows J, Kuruganti P, Chan E, Le P, Chen J, Wang X, Ruslim L, Blake R, Lipson KE, Ramphal J, Do S, Cui JJ, Cherrington JM, Mendel DB
Cancer research 2003 Nov 1;63(21):7345-55
Cancer research 2003 Nov 1;63(21):7345-55
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Peptide Competition using Phospho c-Met Tyr1003 antibody (Product # 44-882G): Extracts of H69 cells unstimulated (lane 1) or treated with 40 ng/mL HGF for 7.5 minutes (lanes 2-5) were resolved by SDS-PAGE on a 10% polyacrylamide gel and transferred to a PVDF membrane. The membrane was blocked with a 5% BSA-TBST buffer. Results indicate specificity for phospho c-Met Tyr1003.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
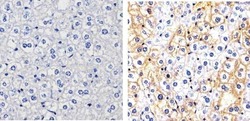
- Experimental details
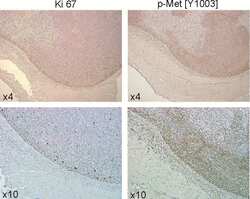
- Immunohistochemistry analysis of Phospho-c-Met (pY1003) showing staining in the cytoplasm and membrane of paraffin-embedded mouse liver tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-c-Met (pY1003) polyclonal antibody (Product # 44-882G) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
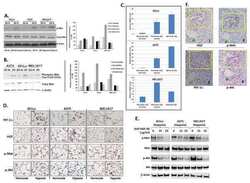
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
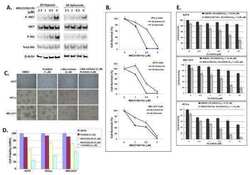
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
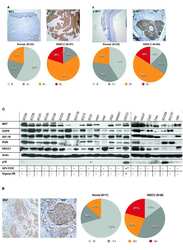
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
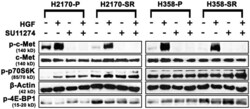
- Figure 3 Differential expression of mTOR pathway proteins in parental and SU11274 resistant H2170 and H358 cell lines by western blotting. Cells were starved overnight and then treated with or without 8.0 uM SU11274 for 24 hours. Cells were stimulated with 40 ng/mL of HGF for 2.5 minutes after which western blot analysis was performed. Downregulation of p-c-Met (Y1003) was seen in both cell lines. Upregulation of p-p70S6kinase (S371) was observed in SR H2170 cells. Upregulation of p-4E-BP1 (T37/46) was also observed in both cells lines +/- SU11274.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
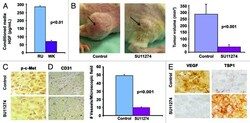
- Figure 1. Intratumoral TKI treatment reduces tumor size in vivo. ( A ) Production of HGF by melanoma cell lines. RU-P cells produced 4-fold higher amounts of HGF compared with WK-P cells in conditioned medium as determined by HGF ELISA kit. ( B ) Five million RU-P melanoma cells were injected subcutaneously into the hind flanks of Rag1 -/- mice. Tumors were allowed to develop for a week after which daily intratumoral doses of SU11274 or vehicle were given for 4 wk. SU11274 treated RU-P tumor xenografts showed a 7-fold reduction in tumor size in comparison to control mice. Seven mice xenografts in each group were evaluated for this study. ( C ) Melanoma tumor sections from mice treated with SU11274 showed downregulation of p-c-Met compared with control mice ( D ) Immunostaining of CD31 in RU-P tumor xenografts in control and SU11274 treated mice. There was a 79.8% (+- 1.5%) ( P < 0.001) decrease in the number of blood vessels when counted in 10 microscopic fields. ( E ) A decrease in VEGF and an increase of TSP1 were found after treatment with SU11274, suggesting decreased angiogenesis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Oral TKI treatment reduces tumor size in vivo. Five million RU-P melanoma cells were injected subcutaneously into the hind flanks of nu/nu mice. Tumors were allowed to develop for a week after which daily oral doses of JNJ38877605 or vehicle were given for 3 wk. ( A )Treatment with JNJ38877605 reduced tumor size by 6-fold when compared with control mice. ( B ) Immunostaining of control and JNJ38877605-treated RU-P tumor xenografts with p-c-Met antibody showed decrease in p-c-Met after treatment with JNJ38877605. ( C ) Immunostaining of control and JNJ38877605 treated RU-P tumor xenografts with CD31 antibody indicate treatment with JNJ38877605 decreased the number of blood vessels in melanoma. There was an 80% (+- 2%) decrease in the number of blood vessels when counted in 10 microscopic fields after treatment with JNJ38877605. ( D ) Immunostaining of control and JNJ38877605-treated RU-P tumor xenografts with VEGF and TSP1 antibody showed a decrease in VEGF and an increase of TSP1 with JNJ38877605 treatment suggesting decreased angiogenesis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
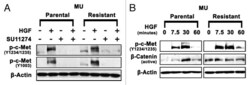
- Figure 4. Upregulation of p-c-Met and active beta-catenin in MU-R cells. ( A ) Cells were starved overnight in media supplemented with 0.5% BSA, and then treated with or without 10 uM SU11274 for 24 h. Cells were stimulated with 40 ng/mL of HGF for 7.5 min, after which immunoblotting analysis was performed. Upregulation of p-c-Met Y1003 (2.4-fold) and p-c-Met Y1234/1235 (1.5-fold) in MU-R cells in absence of HGF was observed, and a 3-5-fold increase in p-c-Met Y1234/1235 was seen after HGF treatment. ( B ) In MU-R cells, HGF induced p-c-Met and active beta-catenin signaling was prolonged by 30 min compared with MU-P cells. Cells were starved for 24 h and then stimulated with 40 ng/ml HGF. Immunoblotting indicated that in MU-R cells, HGF activated p-c-Met (Y1234/1235) and basal levels of active beta-catenin were also 3-fold higher in the absence of HGF and remained high (2.5-fold) for 7.5 min after HGF treatment in MU-R cells compared with those in MU-P cells at 0 min incubation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
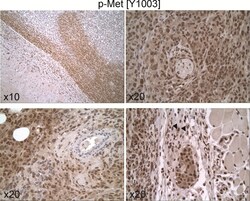
- Experimental details
- 10.1371/journal.pone.0222580.g006 Fig 6 Expression and cellular distribution of p-Met[Y1003] in untreated 231/LM2-4 tumor xenografts. Immunohistochemistry for p-Met[Y1003] shows intense widespread staining in the vital areas of the tumor, with both nuclear and cytoplasmic localization. In certain tumor areas, a large amount of reactive stroma was present, which showed strong cytoplasmic and nuclear staining (down-right arrows).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 10.1371/journal.pone.0222580.g007 Fig 7 Expression of p-Met[Y1003] in control and in treated groups. (A) Tumor section showing peripheral areas with preserved morphology and vast areas of central necrosis. H&E stain. (B) The tumor cells show high pleomorphism, hyperchromasia and increased mitotic activity. H&E stain (C) Immunohistochemistry for p-Met[Y1003]. Untreated controls demonstrate uniform intense immunopositivity in the peripheral vital areas of the tumor (lower left). The necrotic central parts of the tumor are immunonegative (upper right). (D) Untreated controls reveal intense immunopositivity in both nuclear and cytoplasmic distribution. (E, F) UFT+CTX treatment group shows significantly decreased staining intensity with only focal immunopositivity p-Met[Y1003] in a predominantly nuclear pattern. Vast areas of viable tumour are not staining (immunonegative).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 10.1371/journal.pone.0222580.g008 Fig 8 Immunohistochemistry for p-Met[Y1003] H-score results. There was a statistically significant decrease in the expression of p-Met[Y1003] in the UFT+CTX treated group, compared to untreated control (Student's t test, 0.009576 two-tailed p-value).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot