Antibody data
- Antibody Data
- Antigen structure
- References [22]
- Comments [0]
- Validations
- Western blot [2]
- Immunohistochemistry [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-0500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NEFM Monoclonal Antibody (RMO-44)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- RMO-44
- Vial size
- 200 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references An atlas of seven zebrafish hox cluster mutants provides insights into sub/neofunctionalization of vertebrate Hox clusters.
FIGNL1 associates with KIF1Bβ and BICD1 to restrict dynein transport velocity during axon navigation.
A parental requirement for dual-specificity phosphatase 6 in zebrafish.
Asymmetry of an Intracellular Scaffold at Vertebrate Electrical Synapses.
A genetic basis for molecular asymmetry at vertebrate electrical synapses.
Unbiased Proteomics of Early Lewy Body Formation Model Implicates Active Microtubule Affinity-Regulating Kinases (MARKs) in Synucleinopathies.
Ammonium accumulation is a primary effect of 2-methylcitrate exposure in an in vitro model for brain damage in methylmalonic aciduria.
Rapid reverse genetic screening using CRISPR in zebrafish.
Unique function of Kinesin Kif5A in localization of mitochondria in axons.
The C-terminal domains of NF-H and NF-M subunits maintain axonal neurofilament content by blocking turnover of the stationary neurofilament network.
Effect of nicotine and cocaine on neurofilaments and receptors in whole brain tissue and synaptoneurosome preparations.
Characterization and classification of zebrafish brain morphology mutants.
Ammonium alters creatine transport and synthesis in a 3D culture of developing brain cells, resulting in secondary cerebral creatine deficiency.
Whitesnake/sfpq is required for cell survival and neuronal development in the zebrafish.
The zebrafish Iroquois gene iro7 positions the r4/r5 boundary and controls neurogenesis in the rostral hindbrain.
vhnf1 and Fgf signals synergize to specify rhombomere identity in the zebrafish hindbrain.
Abnormal neurofilament transport caused by targeted disruption of neuronal kinesin heavy chain KIF5A.
Ammonium-induced impairment of axonal growth is prevented through glial creatine.
FGF3 and FGF8 mediate a rhombomere 4 signaling activity in the zebrafish hindbrain.
Numerous conglomerate inclusions in slowly progressive familial amyotrophic lateral sclerosis with posterior column involvement.
Numerous conglomerate inclusions in slowly progressive familial amyotrophic lateral sclerosis with posterior column involvement.
Activation of a neurofilament kinase, a tau kinase, and a tau phosphatase by decreased ATP levels in nerve growth factor-differentiated PC-12 cells.
Yamada K, Maeno A, Araki S, Kikuchi M, Suzuki M, Ishizaka M, Satoh K, Akama K, Kawabe Y, Suzuki K, Kobayashi D, Hamano N, Kawamura A
Development (Cambridge, England) 2021 Jun 1;148(11)
Development (Cambridge, England) 2021 Jun 1;148(11)
FIGNL1 associates with KIF1Bβ and BICD1 to restrict dynein transport velocity during axon navigation.
Atkins M, Gasmi L, Bercier V, Revenu C, Del Bene F, Hazan J, Fassier C
The Journal of cell biology 2019 Oct 7;218(10):3290-3306
The Journal of cell biology 2019 Oct 7;218(10):3290-3306
A parental requirement for dual-specificity phosphatase 6 in zebrafish.
Maurer JM, Sagerström CG
BMC developmental biology 2018 Mar 15;18(1):6
BMC developmental biology 2018 Mar 15;18(1):6
Asymmetry of an Intracellular Scaffold at Vertebrate Electrical Synapses.
Marsh AJ, Michel JC, Adke AP, Heckman EL, Miller AC
Current biology : CB 2017 Nov 20;27(22):3561-3567.e4
Current biology : CB 2017 Nov 20;27(22):3561-3567.e4
A genetic basis for molecular asymmetry at vertebrate electrical synapses.
Miller AC, Whitebirch AC, Shah AN, Marsden KC, Granato M, O'Brien J, Moens CB
eLife 2017 May 22;6
eLife 2017 May 22;6
Unbiased Proteomics of Early Lewy Body Formation Model Implicates Active Microtubule Affinity-Regulating Kinases (MARKs) in Synucleinopathies.
Henderson MX, Chung CH, Riddle DM, Zhang B, Gathagan RJ, Seeholzer SH, Trojanowski JQ, Lee VMY
The Journal of neuroscience : the official journal of the Society for Neuroscience 2017 Jun 14;37(24):5870-5884
The Journal of neuroscience : the official journal of the Society for Neuroscience 2017 Jun 14;37(24):5870-5884
Ammonium accumulation is a primary effect of 2-methylcitrate exposure in an in vitro model for brain damage in methylmalonic aciduria.
Cudré-Cung HP, Zavadakova P, do Vale-Pereira S, Remacle N, Henry H, Ivanisevic J, Tavel D, Braissant O, Ballhausen D
Molecular genetics and metabolism 2016 Sep;119(1-2):57-67
Molecular genetics and metabolism 2016 Sep;119(1-2):57-67
Rapid reverse genetic screening using CRISPR in zebrafish.
Shah AN, Davey CF, Whitebirch AC, Miller AC, Moens CB
Nature methods 2015 Jun;12(6):535-40
Nature methods 2015 Jun;12(6):535-40
Unique function of Kinesin Kif5A in localization of mitochondria in axons.
Campbell PD, Shen K, Sapio MR, Glenn TD, Talbot WS, Marlow FL
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Oct 29;34(44):14717-32
The Journal of neuroscience : the official journal of the Society for Neuroscience 2014 Oct 29;34(44):14717-32
The C-terminal domains of NF-H and NF-M subunits maintain axonal neurofilament content by blocking turnover of the stationary neurofilament network.
Rao MV, Yuan A, Campbell J, Kumar A, Nixon RA
PloS one 2012;7(9):e44320
PloS one 2012;7(9):e44320
Effect of nicotine and cocaine on neurofilaments and receptors in whole brain tissue and synaptoneurosome preparations.
Kovacs K, Lajtha A, Sershen H
Brain research bulletin 2010 Apr 29;82(1-2):109-17
Brain research bulletin 2010 Apr 29;82(1-2):109-17
Characterization and classification of zebrafish brain morphology mutants.
Lowery LA, De Rienzo G, Gutzman JH, Sive H
Anatomical record (Hoboken, N.J. : 2007) 2009 Jan;292(1):94-106
Anatomical record (Hoboken, N.J. : 2007) 2009 Jan;292(1):94-106
Ammonium alters creatine transport and synthesis in a 3D culture of developing brain cells, resulting in secondary cerebral creatine deficiency.
Braissant O, Cagnon L, Monnet-Tschudi F, Speer O, Wallimann T, Honegger P, Henry H
The European journal of neuroscience 2008 Apr;27(7):1673-85
The European journal of neuroscience 2008 Apr;27(7):1673-85
Whitesnake/sfpq is required for cell survival and neuronal development in the zebrafish.
Lowery LA, Rubin J, Sive H
Developmental dynamics : an official publication of the American Association of Anatomists 2007 May;236(5):1347-57
Developmental dynamics : an official publication of the American Association of Anatomists 2007 May;236(5):1347-57
The zebrafish Iroquois gene iro7 positions the r4/r5 boundary and controls neurogenesis in the rostral hindbrain.
Lecaudey V, Anselme I, Rosa F, Schneider-Maunoury S
Development (Cambridge, England) 2004 Jul;131(13):3121-31
Development (Cambridge, England) 2004 Jul;131(13):3121-31
vhnf1 and Fgf signals synergize to specify rhombomere identity in the zebrafish hindbrain.
Wiellette EL, Sive H
Development (Cambridge, England) 2003 Aug;130(16):3821-9
Development (Cambridge, England) 2003 Aug;130(16):3821-9
Abnormal neurofilament transport caused by targeted disruption of neuronal kinesin heavy chain KIF5A.
Xia CH, Roberts EA, Her LS, Liu X, Williams DS, Cleveland DW, Goldstein LS
The Journal of cell biology 2003 Apr 14;161(1):55-66
The Journal of cell biology 2003 Apr 14;161(1):55-66
Ammonium-induced impairment of axonal growth is prevented through glial creatine.
Braissant O, Henry H, Villard AM, Zurich MG, Loup M, Eilers B, Parlascino G, Matter E, Boulat O, Honegger P, Bachmann C
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 15;22(22):9810-20
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 15;22(22):9810-20
FGF3 and FGF8 mediate a rhombomere 4 signaling activity in the zebrafish hindbrain.
Maves L, Jackman W, Kimmel CB
Development (Cambridge, England) 2002 Aug;129(16):3825-37
Development (Cambridge, England) 2002 Aug;129(16):3825-37
Numerous conglomerate inclusions in slowly progressive familial amyotrophic lateral sclerosis with posterior column involvement.
Katayama S, Watanabe C, Noda K, Ohishi H, Yamamura Y, Nishisaka T, Inai K, Asayama K, Murayama S, Nakamura S
Journal of the neurological sciences 1999 Dec 1;171(1):72-7
Journal of the neurological sciences 1999 Dec 1;171(1):72-7
Numerous conglomerate inclusions in slowly progressive familial amyotrophic lateral sclerosis with posterior column involvement.
Katayama S, Watanabe C, Noda K, Ohishi H, Yamamura Y, Nishisaka T, Inai K, Asayama K, Murayama S, Nakamura S
Journal of the neurological sciences 1999 Dec 1;171(1):72-7
Journal of the neurological sciences 1999 Dec 1;171(1):72-7
Activation of a neurofilament kinase, a tau kinase, and a tau phosphatase by decreased ATP levels in nerve growth factor-differentiated PC-12 cells.
Bush ML, Miyashiro JS, Ingram VM
Proceedings of the National Academy of Sciences of the United States of America 1995 Mar 14;92(6):1861-5
Proceedings of the National Academy of Sciences of the United States of America 1995 Mar 14;92(6):1861-5
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
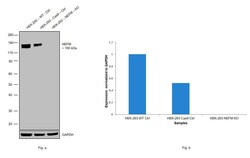
- Experimental details
- Western blot analysis of NEFM was performed by loading 20 µg of HEK-293 wild type (Lane 1), HEK-293 Cas9 control (Lane 2), HEK-293 NEFM knockout (Lane 3) membrane enriched cell extracts. The blot was probed with Anti-NEFM Monoclonal Antibody (Product # 13-0500) (1:1000 dilution) and Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177) (1:4000 dilution). Loss of signal upon CRISPR mediated knockout (KO) confirms that antibody is specific to NEFM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of PC-12 (Lane 1) and PC-12 treated with Nerve Growth Factor (150 ng/mL for 4 days) (Lane 2), tissue extract of Rat Brain (Lane 3). The blot was probed with Anti-NEFM Monoclonal Antibody (RMO-44) (Product # 13-0500, 1:1000 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25 µg/mL, 1:4000 dilution). A 150 kDa band corresponding to NEFM was observed in Rat Brain and enhanced upon treatment with Nerve Growth Factor in PC-12. An additional band at 70 kDa was also observed in PC-12 cell line.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
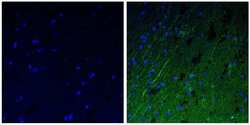
- Experimental details
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament medium chain monoclonal antibody (Product # 13-0500) in 0.3% BSA at a dilution of 1:50 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
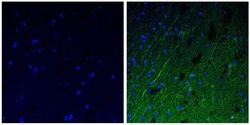
- Experimental details
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament medium chain monoclonal antibody (Product # 13-0500) in 0.3% BSA at a dilution of 1:50 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoprecipitation
Immunoprecipitation