48-4777-42
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [34]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [24]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 48-4777-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (236A/E7), eFluor™ 450, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 236A/E7 antibody reacts with human foxp3 protein also known as FORKHEAD BOX P3, SCURFIN, and JM2; cross reactivity of this antibody to other proteins has not been determined. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of Foxp3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells.
- Antibody clone number
- 236A/E7
- Concentration
- 5 µL/Test
Submitted references Lactobacillus rhamnosus GG-Derived Soluble Mediators Modulate Adaptive Immune Cells.
Increased circulating regulatory T cells in medicated people with schizophrenia.
Multicomponent analysis of the tumour microenvironment reveals low CD8 T cell number, low stromal caveolin-1 and high tenascin-C and their combination as significant prognostic markers in non-small cell lung cancer.
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Identifying functional defects in patients with immune dysregulation due to LRBA and CTLA-4 mutations.
A novel method for assaying human regulatory T cell direct suppression of B cell effector function.
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
TGF-β Affects the Differentiation of Human GM-CSF(+) CD4(+) T Cells in an Activation- and Sodium-Dependent Manner.
Selective Sparing of Human Tregs by Pharmacologic Inhibitors of the Phosphatidylinositol 3-Kinase and MEK Pathways.
A fully human IgG1 anti-PD-L1 MAb in an in vitro assay enhances antigen-specific T-cell responses.
The immune microenvironment of breast ductal carcinoma in situ.
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Differential tumor infiltration by T-cells characterizes intrinsic molecular subtypes in breast cancer.
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Hypomethylation at the regulatory T cell-specific demethylated region in CD25hi T cells is decoupled from FOXP3 expression at the inflamed site in childhood arthritis.
Altered expression of autoimmune regulator in infant down syndrome thymus, a possible contributor to an autoimmune phenotype.
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
IDO and regulatory T cell support are critical for cytotoxic T lymphocyte-associated Ag-4 Ig-mediated long-term solid organ allograft survival.
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Ludwig IS, Broere F, Manurung S, Lambers TT, van der Zee R, van Eden W
Frontiers in immunology 2018;9:1546
Frontiers in immunology 2018;9:1546
Increased circulating regulatory T cells in medicated people with schizophrenia.
Kelly DL, Li X, Kilday C, Feldman S, Clark S, Liu F, Buchanan RW, Tonelli LH
Psychiatry research 2018 Nov;269:517-523
Psychiatry research 2018 Nov;269:517-523
Multicomponent analysis of the tumour microenvironment reveals low CD8 T cell number, low stromal caveolin-1 and high tenascin-C and their combination as significant prognostic markers in non-small cell lung cancer.
Onion D, Isherwood M, Shridhar N, Xenophontos M, Craze ML, Day LJ, García-Márquez MA, Pineda RG, Reece-Smith AM, Saunders JH, Duffy JP, Argent RH, Grabowska AM
Oncotarget 2018 Jan 5;9(2):1760-1771
Oncotarget 2018 Jan 5;9(2):1760-1771
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Kishore M, Cheung KCP, Fu H, Bonacina F, Wang G, Coe D, Ward EJ, Colamatteo A, Jangani M, Baragetti A, Matarese G, Smith DM, Haas R, Mauro C, Wraith DC, Okkenhaug K, Catapano AL, De Rosa V, Norata GD, Marelli-Berg FM
Immunity 2017 Nov 21;47(5):875-889.e10
Immunity 2017 Nov 21;47(5):875-889.e10
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Syed Khaja AS, Toor SM, El Salhat H, Faour I, Ul Haq N, Ali BR, Elkord E
Oncotarget 2017 May 16;8(20):33159-33171
Oncotarget 2017 May 16;8(20):33159-33171
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Zanin-Zhorov A, Weiss JM, Trzeciak A, Chen W, Zhang J, Nyuydzefe MS, Arencibia C, Polimera S, Schueller O, Fuentes-Duculan J, Bonifacio KM, Kunjravia N, Cueto I, Soung J, Fleischmann RM, Kivitz A, Lebwohl M, Nunez M, Woodson J, Smith SL, West RF, Berger M, Krueger JG, Ryan JL, Waksal SD
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Identifying functional defects in patients with immune dysregulation due to LRBA and CTLA-4 mutations.
Hou TZ, Verma N, Wanders J, Kennedy A, Soskic B, Janman D, Halliday N, Rowshanravan B, Worth A, Qasim W, Baxendale H, Stauss H, Seneviratne S, Neth O, Olbrich P, Hambleton S, Arkwright PD, Burns SO, Walker LS, Sansom DM
Blood 2017 Mar 16;129(11):1458-1468
Blood 2017 Mar 16;129(11):1458-1468
A novel method for assaying human regulatory T cell direct suppression of B cell effector function.
Weingartner E, Courneya JP, Keegan A, Golding A
Journal of immunological methods 2017 Feb;441:1-7
Journal of immunological methods 2017 Feb;441:1-7
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Terranova-Barberio M, Thomas S, Ali N, Pawlowska N, Park J, Krings G, Rosenblum MD, Budillon A, Munster PN
Oncotarget 2017 Dec 26;8(69):114156-114172
Oncotarget 2017 Dec 26;8(69):114156-114172
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
TGF-β Affects the Differentiation of Human GM-CSF(+) CD4(+) T Cells in an Activation- and Sodium-Dependent Manner.
Éliás S, Schmidt A, Kannan V, Andersson J, Tegnér J
Frontiers in immunology 2016;7:603
Frontiers in immunology 2016;7:603
Selective Sparing of Human Tregs by Pharmacologic Inhibitors of the Phosphatidylinositol 3-Kinase and MEK Pathways.
Zwang NA, Zhang R, Germana S, Fan MY, Hastings WD, Cao A, Turka LA
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2016 Sep;16(9):2624-38
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2016 Sep;16(9):2624-38
A fully human IgG1 anti-PD-L1 MAb in an in vitro assay enhances antigen-specific T-cell responses.
Grenga I, Donahue RN, Lepone LM, Richards J, Schlom J
Clinical & translational immunology 2016 May;5(5):e83
Clinical & translational immunology 2016 May;5(5):e83
The immune microenvironment of breast ductal carcinoma in situ.
Thompson E, Taube JM, Elwood H, Sharma R, Meeker A, Warzecha HN, Argani P, Cimino-Mathews A, Emens LA
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2016 Mar;29(3):249-58
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2016 Mar;29(3):249-58
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Ghosh S, Taylor A, Chin M, Huang HR, Conery AR, Mertz JA, Salmeron A, Dakle PJ, Mele D, Cote A, Jayaram H, Setser JW, Poy F, Hatzivassiliou G, DeAlmeida-Nagata D, Sandy P, Hatton C, Romero FA, Chiang E, Reimer T, Crawford T, Pardo E, Watson VG, Tsui V, Cochran AG, Zawadzke L, Harmange JC, Audia JE, Bryant BM, Cummings RT, Magnuson SR, Grogan JL, Bellon SF, Albrecht BK, Sims RJ 3rd, Lora JM
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Liang DY, Hou YQ, Luo LJ, Ao L
International journal of molecular medicine 2016 Jul;38(1):131-8
International journal of molecular medicine 2016 Jul;38(1):131-8
Differential tumor infiltration by T-cells characterizes intrinsic molecular subtypes in breast cancer.
Miyan M, Schmidt-Mende J, Kiessling R, Poschke I, de Boniface J
Journal of translational medicine 2016 Jul 29;14(1):227
Journal of translational medicine 2016 Jul 29;14(1):227
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
MacDonald KG, Hoeppli RE, Huang Q, Gillies J, Luciani DS, Orban PC, Broady R, Levings MK
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Kovacsovics-Bankowski M, Kelley TW, Efimova O, Kim SJ, Wilson A, Swierczek S, Prchal J
Experimental hematology & oncology 2015;5:28
Experimental hematology & oncology 2015;5:28
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
Hypomethylation at the regulatory T cell-specific demethylated region in CD25hi T cells is decoupled from FOXP3 expression at the inflamed site in childhood arthritis.
Bending D, Pesenacker AM, Ursu S, Wu Q, Lom H, Thirugnanabalan B, Wedderburn LR
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 15;193(6):2699-708
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 15;193(6):2699-708
Altered expression of autoimmune regulator in infant down syndrome thymus, a possible contributor to an autoimmune phenotype.
Skogberg G, Lundberg V, Lindgren S, Gudmundsdottir J, Sandström K, Kämpe O, Annerén G, Gustafsson J, Sunnegårdh J, van der Post S, Telemo E, Berglund M, Ekwall O
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 1;193(5):2187-95
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 1;193(5):2187-95
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
Ye SB, Li ZL, Luo DH, Huang BJ, Chen YS, Zhang XS, Cui J, Zeng YX, Li J
Oncotarget 2014 Jul 30;5(14):5439-52
Oncotarget 2014 Jul 30;5(14):5439-52
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
Schmidt HH, Ge Y, Hartmann FJ, Conrad H, Klug F, Nittel S, Bernhard H, Domschke C, Schuetz F, Sohn C, Beckhove P
Oncoimmunology 2013 Jun 1;2(6):e24962
Oncoimmunology 2013 Jun 1;2(6):e24962
IDO and regulatory T cell support are critical for cytotoxic T lymphocyte-associated Ag-4 Ig-mediated long-term solid organ allograft survival.
Sucher R, Fischler K, Oberhuber R, Kronberger I, Margreiter C, Ollinger R, Schneeberger S, Fuchs D, Werner ER, Watschinger K, Zelger B, Tellides G, Pilat N, Pratschke J, Margreiter R, Wekerle T, Brandacher G
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 1;188(1):37-46
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 1;188(1):37-46
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Green AM, Mattila JT, Bigbee CL, Bongers KS, Lin PL, Flynn JL
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Kasprzycka M, Zhang Q, Witkiewicz A, Marzec M, Potoczek M, Liu X, Wang HY, Milone M, Basu S, Mauger J, Choi JK, Abrams JT, Hou JS, Rook AH, Vonderheid E, Woetmann A, Odum N, Wasik MA
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Ahmadzadeh M, Rosenberg SA
Blood 2006 Mar 15;107(6):2409-14
Blood 2006 Mar 15;107(6):2409-14
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Roncador G, Brown PJ, Maestre L, Hue S, Martínez-Torrecuadrada JL, Ling KL, Pratap S, Toms C, Fox BC, Cerundolo V, Powrie F, Banham AH
European journal of immunology 2005 Jun;35(6):1681-91
European journal of immunology 2005 Jun;35(6):1681-91
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
Alvaro T, Lejeune M, Salvadó MT, Bosch R, García JF, Jaén J, Banham AH, Roncador G, Montalbán C, Piris MA
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
Roncador G, Garcia JF, Garcia JF, Maestre L, Lucas E, Menarguez J, Ohshima K, Nakamura S, Banham AH, Piris MA
Leukemia 2005 Dec;19(12):2247-53
Leukemia 2005 Dec;19(12):2247-53
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Wolf D, Wolf AM, Rumpold H, Fiegl H, Zeimet AG, Muller-Holzner E, Deibl M, Gastl G, Gunsilius E, Marth C
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
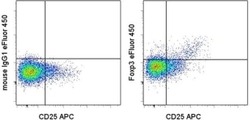
- Experimental details
- Surface staining of normal human peripheral blood cells with Anti-Human CD25 APC (Product # 17-0259-42) followed by fixation and permeabilization with the Foxp3/Transcription Factor Staining Buffers (Product # 00-5523-00) and intracellular staining with Mouse IgG1 K Isotype Control eFluor® 450 (Product # 48-4714-82) (left) or Anti-Human Foxp3 eFluor® 450 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
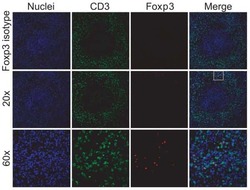
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
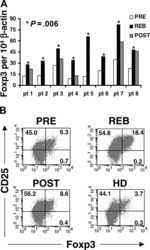
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
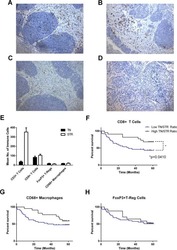
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
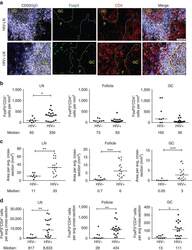
- Experimental details
- Figure 1 T FR expansion in human lymph nodes (LNs) during HIV infection. ( a ) Representative images of immunofluorescently stained LN sections from a subject with chronic, untreated HIV infection ( n =17) and an uninfected control subject ( n =8). LNs were stained with fluorescently labelled antibodies to CD20 (white), IgD (blue), Foxp3 (green) and CD4 (red). Follicle (F) was defined as CD20 + (white line) and germinal centre (GC) was defined as CD20 + IgD - (yellow line). Images were scanned at x 60 magnification and scale bars equal 20 mum. ( b ) Foxp3 + CD4 + cells were quantified in different regions of immunofluorescently stained LN shown in a from uninfected ( n =8) and HIV-infected ( n =17) subjects using visual inspection and quantitative image analysis to determine areas. ( c ) The average areas of total (LN), follicular and GC regions per LN cross-section were determined by quantitative image analysis. ( d ) The average number of CD4 + Foxp3 + cells per LN, F and GC cross-section was calculated by multiplying the frequency of CD4 + Foxp3 + cells per mm 2 ( b ) by the average area of each region ( c ) for each subject. The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Mann-Whitney (Wilcoxon) tests to compare unpaired, nonparametric values and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
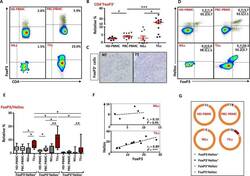
- Experimental details
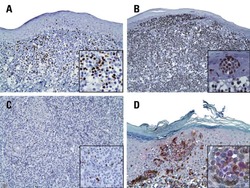
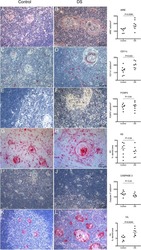
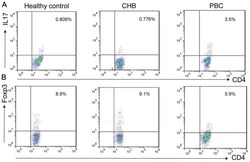
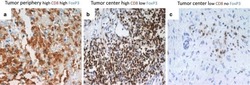
- Figure 6 FoxP3 and Helios expression in CD4 + T cells PBMC from HD and PBC patients, NILs and TILs were stained for CD3 and CD4 antibodies followed by intracellular staining for FoxP3 and Helios. Live cells were first gated using Fixable Viability Dye 660. ( A ). Representative flow cytometric plots of FoxP3 staining from one cancer patient are shown. ( B ). Scatter plot showing the differences in frequencies of FoxP3 + Tregs between different samples. ( C ). IHC staining of FoxP3 + expression in one NT and TT samples. ( D ). Flow cytometric plots of FoxP3 and Helios co-expression in CD4 + T cells from different samples and whisker plots ( E ) showing differences in various FoxP3 and Helios-expressing Treg subsets. ( F ). Non-parametric Spearman's test showing correlations between FoxP3 and Helios expressions in NILs and TILs. ( G ). Pie charts show the relative percentages of different FoxP3 and Helios Treg subsets.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
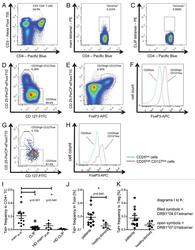
- Experimental details
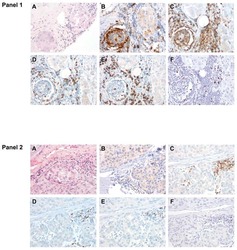
- Figure 3. Tetramer staining of conventional and regulatory T cells from breast cancer patients. ( A-G ) Peripheral blood mononuclear cells (PBMCs) of a breast cancer patient were analyzed, upon gating on living CD3 + CD4 + T cells. The patient sample was stained with tetramers presenting either mam 34-48 ( B ) or the CLIP peptide ( C ). Numbers indicate the percentage of cells in the respective gate, referring to lymphocytes ( A ) or CD3 + CD4 + T cells ( B ) and ( C ). Within CD3 + CD4 + T cells, regulatory T cells (Tregs) were identified as CD25 high CD127 low ( D ) and ( G ) and CD25 high CD127 low FOXP3 + ( E ), ( F ) and ( H ). ( G ) reports the percentage of Tregs within tetramer-positive cells in a representative patient. ( I ) Frequency of mam 34-48 - and CLIP-presenting tetramer-positive cells among CD3 + CD4 + T cells of breast cancer patients and healthy donors (HD) (p values as per Mann-Whitney U tests are indicated). ( J and K ) Frequency of mam 34-48 -specific Tcon ( J ) cells and Tregs ( K ) of breast cancer patients and HDs.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
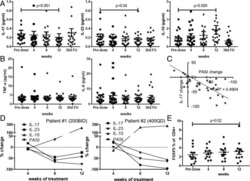
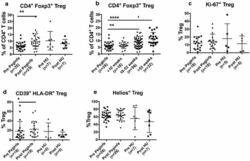
- Fig. 1 PegINFalpha increases Treg and highly suppressive Treg cells in peripheral blood of PV and ET patients. PBMC were collected from patients with PV or ET treated with PegINFalpha or HU for at least 70 days (range 70-616, median 112 for PegINFalpha and range 113-2422, median 175 for HU treated patients). Lymphocytes were analyzed by flow cytometry using surface markers, CD3, CD4, CD25, CD39, HLA-DR and intracellular markers Foxp3, Ki-67 and Helios. a represents CD4 + CD25 + Foxp3 + Treg cells. In b the frequency of Treg was analyzed at different time points after initiation of PegINFalpha treatment. c-e show frequency of Ki-67 + Treg, Helios + Treg, and CD39 + /HLA-DR + ) Treg
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
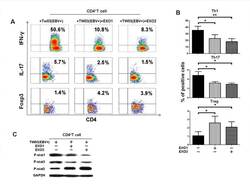
- Figure 3 NPC tumor-released exosomes inhibited Th1 and Th17 cell induction but promoted Tregs by altering STAT protein phosphorylation A. Differentiation of naive CD4 + T cells into Th1 and Th17 cells and Tregs after stimulation with tumor cells with or without EXOs. Purified naive CD4 + T cells from healthy donors were co-cultured with the irradiated NPC cell line TW03 (EBV + ) and then treated with EXOs or left or untreated in the presence of OKT3 in IL-2 medium for 5 days. INFgamma, IL-17, and Foxp3 staining following by FACS analysis was performed after stimulation with PMA/ionomycin. Representative data of five experiments are shown. B. Numerous data showing the mean percentage +- S.E.M. of positive cell subsets in three independent experiments. * means P < 0.05. C. Western blot analysis for the expression of P-STAT1, P-STAT3, and P-STAT5 proteins. Stimulation with EXOs decreased the expression of P-STAT1 and P-STAT3 but increased the expression of P-STAT5 in OKT3 and NPC cells stimulated by CD4 + T cells; the GAPDH gene was included as a control. Representative data of three experiments are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
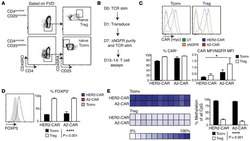
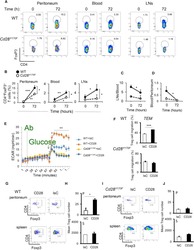
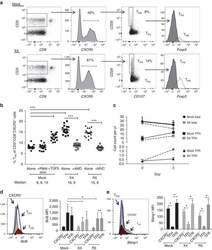
- Figure 3 HIV entry and replication promote T FR expansion. Disaggregated tonsil cells were spinoculated with X4 or R5 HIV and T FR populations were analysed by flow cytometry ( n =15). ( a ) A representative example of tonsil cell flow gating. From viable CD3 + CD8 - cells, T FR are defined as CXCR5 + and CD25 hi CD127 - . T FR cells contain Foxp3 + cells, whereas remaining T FH (CXCR5 + CD25 lo/- ) cells are Foxp3 - . ( b ) Percentages of T FR determined by gating strategies in a are shown. Experimental conditions include mock-spinoculated cells cultured with PMA (50 ng ml -1 ) and ionomycin (1 mug ml -1 ) or exogenous TGF-beta (100 ng ml -1 ) for 24 h and cells pretreated to block CXCR4 (AMD, 200 muM) and CCR5 (MVC, 2 muM). ( c ) Using flow cytometry counting beads, the number of cells per mul were determined for total (CD3 + CD8 - ), T FH (CXCR5 + CD25 lo/- ) and T FR (CXCR5 + CD25 hi CD127 - ) subsets in mock- and X4-spinoculated samples ( n =3). ( d ) Bcl-6 expression is shown in CXCR5- (grey), T FH (blue) and T FR (red) populations after mock-, X4- or R5-spinoculation ( n =5). ( e ) Blimp-1 expression was also determined as in d . The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman nonparametric tests ( b , d , e ) and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
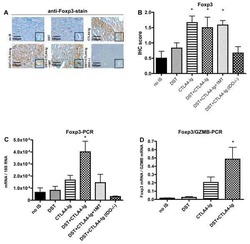
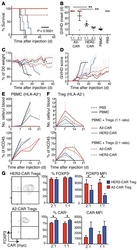
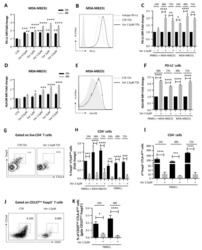
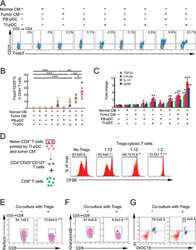
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
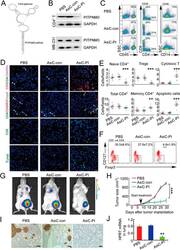
- Experimental details
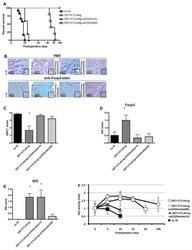
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry