Antibody data
- Antibody Data
- Antigen structure
- References [92]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [2]
- Immunohistochemistry [3]
- Other assay [76]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 40-2300 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ZO-1 Polyclonal Antibody (ZMD.437)
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody reacts with the ZO-1 protein. Based on sequence homology, reactivity with ZO-2 or ZO-3 is not expected. 40-2300 has been successfully used in immunofluorescence and immunohistochemistry. However, for western blotting we recommend the use of monoclonal anti-ZO-1 antibody (Cat. No. 339100), or other polyclonal ZO-1 antibodies (Cat. Nos. 617300 or 402200).
- Antibody clone number
- ZMD.437
- Concentration
- 0.25 mg/mL
Submitted references LINC01094/SPI1/CCL7 Axis Promotes Macrophage Accumulation in Lung Adenocarcinoma and Tumor Cell Dissemination.
Phaseolus vulgaris extract ameliorates high-fat diet-induced colonic barrier dysfunction and inflammation in mice by regulating peroxisome proliferator-activated receptor expression and butyrate levels.
A medium composition containing normal resting glucose that supports differentiation of primary human airway cells.
The evolutionary history of the polyQ tract in huntingtin sheds light on its functional pro-neural activities.
Hydrocephalus in Nfix(-/-) Mice Is Underpinned by Changes in Ependymal Cell Physiology.
A microfluidic chip carrier including temperature control and perfusion system for long-term cell imaging.
Bazedoxifene, a Selective Estrogen Receptor Modulator, Promotes Functional Recovery in a Spinal Cord Injury Rat Model.
Increased Mucosal Thrombin is Associated with Crohn's Disease and Causes Inflammatory Damage through Protease-activated Receptors Activation.
Ruscogenin attenuated tight junction injury and tumor migration in colorectal liver metastasis mice via regulating TRAP1.
Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.
Increased Catecholamine Levels and Inflammatory Mediators Alter Barrier Properties of Brain Microvascular Endothelial Cells in vitro.
Impact of viral disease hypophagia on pig jejunal function and integrity.
Two-Stage Interpretation of Changes in TEER of Intestinal Epithelial Layers Protected by Adhering Bifidobacteria During E. coli Challenges.
R-spondin2 signaling is required for oocyte-driven intercellular communication and follicular growth.
A role for early oral exposure to house dust mite allergens through breast milk in IgE-mediated food allergy susceptibility.
Cellulose Nanofibril Hydrogel Promotes Hepatic Differentiation of Human Liver Organoids.
Antrodia cinnamomea Confers Obesity Resistance and Restores Intestinal Barrier Integrity in Leptin-deficient Obese Mice.
Assembly and Function of a Bioengineered Human Liver for Transplantation Generated Solely from Induced Pluripotent Stem Cells.
Neurotrophic Factors Protect the Intestinal Barrier from Rotavirus Insult in Mice.
Elevated TRPV4 Levels Contribute to Endothelial Damage and Scarring in Experimental Spinal Cord Injury.
Retinal Phenotype in the rd9 Mutant Mouse, a Model of X-Linked RP.
EspF is crucial for Citrobacter rodentium-induced tight junction disruption and lethality in immunocompromised animals.
Non-canonical Wnt signaling regulates junctional mechanocoupling during angiogenic collective cell migration.
ROCK2 inhibition triggers the collective invasion of colorectal adenocarcinomas.
Organoid single cell profiling identifies a transcriptional signature of glomerular disease.
Inhibitory Effects of Sodium Alginate on Hepatic Steatosis in Mice Induced by a Methionine- and Choline-deficient Diet.
Improved In Vitro Model for Intranasal Mucosal Drug Delivery: Primary Olfactory and Respiratory Epithelial Cells Compared with the Permanent Nasal Cell Line RPMI 2650.
Stabilization of HIF-2α impacts pancreas growth.
Characterization of human fetal brain endothelial cells reveals barrier properties suitable for in vitro modeling of the BBB with syngenic co-cultures.
Patient-iPSC-Derived Kidney Organoids Show Functional Validation of a Ciliopathic Renal Phenotype and Reveal Underlying Pathogenetic Mechanisms.
Next generation histology methods for three-dimensional imaging of fresh and archival human brain tissues.
Galectin-3 modulates the polarized surface delivery of β1-integrin in epithelial cells.
Wdpcp promotes epicardial EMT and epicardium-derived cell migration to facilitate coronary artery remodeling.
Cadherin-6B proteolytic N-terminal fragments promote chick cranial neural crest cell delamination by regulating extracellular matrix degradation.
Multifaceted Mechanisms of WY-14643 to Stabilize the Blood-Brain Barrier in a Model of Traumatic Brain Injury.
Targeting Glioma Stem Cell-Derived Pericytes Disrupts the Blood-Tumor Barrier and Improves Chemotherapeutic Efficacy.
Complement C5aR1 Signaling Promotes Polarization and Proliferation of Embryonic Neural Progenitor Cells through PKCζ.
Massively Increased Caries Susceptibility in an Irf6 Cleft Lip/Palate Model.
In Vitro Study of Receptor-Mediated Silica Nanoparticles Delivery across Blood-Brain Barrier.
Contractile forces at tricellular contacts modulate epithelial organization and monolayer integrity.
E-cadherin cleavage by MT2-MMP regulates apical junctional signaling and epithelial homeostasis in the intestine.
Systemic Injection of RPE65-Programmed Bone Marrow-Derived Cells Prevents Progression of Chronic Retinal Degeneration.
Silencing microRNA-143 protects the integrity of the blood-brain barrier: implications for methamphetamine abuse.
Enhanced Re-Endothelialization of Decellularized Rat Lungs.
Intrinsic FGF2 and FGF5 promotes angiogenesis of human aortic endothelial cells in 3D microfluidic angiogenesis system.
Isolation, culture and characterization of primary mouse RPE cells.
Murine junctional adhesion molecules JAM-B and JAM-C mediate endothelial and stellate cell interactions during hepatic fibrosis.
MICAL2 is a novel human cancer gene controlling mesenchymal to epithelial transition involved in cancer growth and invasion.
Toll-like receptor 4-mediated lymphocyte influx induces neonatal necrotizing enterocolitis.
Evaluation of Planar-Cell-Polarity Phenotypes in Ciliopathy Mouse Mutant Cochlea.
Lung Microtissue Array to Screen the Fibrogenic Potential of Carbon Nanotubes.
A Novel Dynamic Neonatal Blood-Brain Barrier on a Chip.
Liver Sinusoidal Endothelial Cells Escape Senescence by Loss of p19ARF.
The pH-sensing receptor OGR1 improves barrier function of epithelial cells and inhibits migration in an acidic environment.
Immunohistochemical expression of VDR is associated with reduced integrity of tight junction complex in psoriatic skin.
Upregulation of Shiga toxin receptor CD77/Gb3 and interleukin-1β expression in the brain of EHEC patients with hemolytic uremic syndrome and neurologic symptoms.
Acid sphingomyelinase inhibition protects mice from lung edema and lethal Staphylococcus aureus sepsis.
Constraint of gene expression by the chromatin remodelling protein CHD4 facilitates lineage specification.
Impaired intestinal mucosal barrier upon ischemia-reperfusion: "patching holes in the shield with a simple surgical method".
Phosphorylation decreases ubiquitylation of the thiazide-sensitive cotransporter NCC and subsequent clathrin-mediated endocytosis.
Retinal pigment epithelial cell death by the alternative complement cascade: role of membrane regulatory proteins, calcium, PKC, and oxidative stress.
Twist1-induced dissemination preserves epithelial identity and requires E-cadherin.
Vinculin directly binds zonula occludens-1 and is essential for stabilizing connexin-43-containing gap junctions in cardiac myocytes.
Disruption of astrocyte-vascular coupling and the blood-brain barrier by invading glioma cells.
An in vitro blood-brain barrier model combining shear stress and endothelial cell/astrocyte co-culture.
Intestinal and systemic inflammatory responses are positively associated with sulfidogenic bacteria abundance in high-fat-fed male C57BL/6J mice.
MUC4 overexpression augments cell migration and metastasis through EGFR family proteins in triple negative breast cancer cells.
Polarity, cell division, and out-of-equilibrium dynamics control the growth of epithelial structures.
Luminal mitosis drives epithelial cell dispersal within the branching ureteric bud.
Heme modulates intestinal epithelial cell activation: involvement of NADPHox-derived ROS signaling.
Echovirus 1 entry into polarized Caco-2 cells depends on dynamin, cholesterol, and cellular factors associated with macropinocytosis.
Evidence for phenotypic plasticity in aggressive triple-negative breast cancer: human biology is recapitulated by a novel model system.
The LKB1 tumor suppressor controls spindle orientation and localization of activated AMPK in mitotic epithelial cells.
C-reactive protein increases BBB permeability: implications for obesity and neuroinflammation.
Integrin-linked kinase deletion in the developing lens leads to capsule rupture, impaired fiber migration and non-apoptotic epithelial cell death.
Liver X receptor activation enhances blood-brain barrier integrity in the ischemic brain and increases the abundance of ATP-binding cassette transporters ABCB1 and ABCC1 on brain capillary cells.
The tight junction scaffolding protein cingulin regulates neural crest cell migration.
Lung self-assembly is modulated by tissue surface tensions.
Cx50 requires an intact PDZ-binding motif and ZO-1 for the formation of functional intercellular channels.
Renal ischemia-reperfusion injury causes intercalated cell-specific disruption of occludin in the collecting duct.
S-Adenosylmethionine regulates connexins sub-types expressed by hepatocytes.
Hypoxia stimulates hepatocyte epithelial to mesenchymal transition by hypoxia-inducible factor and transforming growth factor-beta-dependent mechanisms.
Embryonic vascular endothelial cells are malleable to reprogramming via Prox1 to a lymphatic gene signature.
Adenovirus-driven overexpression of proteinases in organ-cultured normal human corneas leads to diabetic-like changes.
The canonical NF-kappaB pathway governs mammary tumorigenesis in transgenic mice and tumor stem cell expansion.
Normalization of wound healing and diabetic markers in organ cultured human diabetic corneas by adenoviral delivery of c-Met gene.
Zonula occludens-1, occludin, and E-cadherin protein expression in biliary tract cancers.
Depletion of passenger leukocytes from corneal grafts: an effective means of promoting transplant survival?
Disruptions of occludin and claudin-5 in brain endothelial cells in vitro and in brains of mice with acute liver failure.
Beta-catenin is necessary to keep cells of ureteric bud/Wolffian duct epithelium in a precursor state.
ZO-1 is required for protein kinase C gamma-driven disassembly of connexin 43.
Expression of zonula occludens-1 (ZO-1) and the transcription factor ZO-1-associated nucleic acid-binding protein (ZONAB)-MsY3 in glial cells and colocalization at oligodendrocyte and astrocyte gap junctions in mouse brain.
Wu Z, Bai X, Lu Z, Liu S, Jiang H
Journal of immunology research 2022;2022:6450721
Journal of immunology research 2022;2022:6450721
Phaseolus vulgaris extract ameliorates high-fat diet-induced colonic barrier dysfunction and inflammation in mice by regulating peroxisome proliferator-activated receptor expression and butyrate levels.
Avagliano C, De Caro C, Cuozzo M, Liguori FM, La Rana G, Micheli L, Di Cesare Mannelli L, Ghelardini C, Paciello O, Russo R
Frontiers in pharmacology 2022;13:930832
Frontiers in pharmacology 2022;13:930832
A medium composition containing normal resting glucose that supports differentiation of primary human airway cells.
Morgan R, Manfredi C, Easley KF, Watkins LD, Hunt WR, Goudy SL, Sorscher EJ, Koval M, Molina SA
Scientific reports 2022 Jan 27;12(1):1540
Scientific reports 2022 Jan 27;12(1):1540
The evolutionary history of the polyQ tract in huntingtin sheds light on its functional pro-neural activities.
Iennaco R, Formenti G, Trovesi C, Rossi RL, Zuccato C, Lischetti T, Bocchi VD, Scolz A, Martínez-Labarga C, Rickards O, Pacifico M, Crottini A, Møller AP, Chen RZ, Vogt TF, Pavesi G, Horner DS, Saino N, Cattaneo E
Cell death and differentiation 2022 Feb;29(2):293-305
Cell death and differentiation 2022 Feb;29(2):293-305
Hydrocephalus in Nfix(-/-) Mice Is Underpinned by Changes in Ependymal Cell Physiology.
Harkins D, Harvey TJ, Atterton C, Miller I, Currey L, Oishi S, Kasherman M, Davila RA, Harris L, Green K, Piper H, Parton RG, Thor S, Cooper HM, Piper M
Cells 2022 Aug 2;11(15)
Cells 2022 Aug 2;11(15)
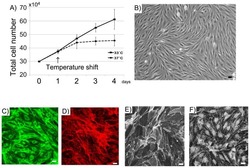
A microfluidic chip carrier including temperature control and perfusion system for long-term cell imaging.
Cantoni F, Werr G, Barbe L, Porras AM, Tenje M
HardwareX 2021 Oct;10:e00245
HardwareX 2021 Oct;10:e00245
Bazedoxifene, a Selective Estrogen Receptor Modulator, Promotes Functional Recovery in a Spinal Cord Injury Rat Model.
Kim Y, Roh EJ, Joshi HP, Shin HE, Choi H, Kwon SY, Sohn S, Han I
International journal of molecular sciences 2021 Oct 12;22(20)
International journal of molecular sciences 2021 Oct 12;22(20)
Increased Mucosal Thrombin is Associated with Crohn's Disease and Causes Inflammatory Damage through Protease-activated Receptors Activation.
Motta JP, Palese S, Giorgio C, Chapman K, Denadai-Souza A, Rousset P, Sagnat D, Guiraud L, Edir A, Seguy C, Alric L, Bonnet D, Bournet B, Buscail L, Gilletta C, Buret AG, Wallace JL, Hollenberg MD, Oswald E, Barocelli E, Le Grand S, Le Grand B, Deraison C, Vergnolle N
Journal of Crohn's & colitis 2021 May 4;15(5):787-799
Journal of Crohn's & colitis 2021 May 4;15(5):787-799
Ruscogenin attenuated tight junction injury and tumor migration in colorectal liver metastasis mice via regulating TRAP1.
Lv Y, Wu X, Chen J, Shao F
Translational cancer research 2021 Mar;10(3):1470-1483
Translational cancer research 2021 Mar;10(3):1470-1483
Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.
Salman MM, Marsh G, Kusters I, Delincé M, Di Caprio G, Upadhyayula S, de Nola G, Hunt R, Ohashi KG, Gray T, Shimizu F, Sano Y, Kanda T, Obermeier B, Kirchhausen T
Frontiers in bioengineering and biotechnology 2020;8:573775
Frontiers in bioengineering and biotechnology 2020;8:573775
Increased Catecholamine Levels and Inflammatory Mediators Alter Barrier Properties of Brain Microvascular Endothelial Cells in vitro.
Ittner C, Burek M, Störk S, Nagai M, Förster CY
Frontiers in cardiovascular medicine 2020;7:73
Frontiers in cardiovascular medicine 2020;7:73
Impact of viral disease hypophagia on pig jejunal function and integrity.
Helm ET, Curry SM, De Mille CM, Schweer WP, Burrough ER, Gabler NK
PloS one 2020;15(1):e0227265
PloS one 2020;15(1):e0227265
Two-Stage Interpretation of Changes in TEER of Intestinal Epithelial Layers Protected by Adhering Bifidobacteria During E. coli Challenges.
Yuan L, van der Mei HC, Busscher HJ, Peterson BW
Frontiers in microbiology 2020;11:599555
Frontiers in microbiology 2020;11:599555
R-spondin2 signaling is required for oocyte-driven intercellular communication and follicular growth.
De Cian MC, Gregoire EP, Le Rolle M, Lachambre S, Mondin M, Bell S, Guigon CJ, Chassot AA, Chaboissier MC
Cell death and differentiation 2020 Oct;27(10):2856-2871
Cell death and differentiation 2020 Oct;27(10):2856-2871
A role for early oral exposure to house dust mite allergens through breast milk in IgE-mediated food allergy susceptibility.
Rekima A, Bonnart C, Macchiaverni P, Metcalfe J, Tulic MK, Halloin N, Rekima S, Genuneit J, Zanelli S, Medeiros S, Palmer DJ, Prescott S, Verhasselt V
The Journal of allergy and clinical immunology 2020 May;145(5):1416-1429.e11
The Journal of allergy and clinical immunology 2020 May;145(5):1416-1429.e11
Cellulose Nanofibril Hydrogel Promotes Hepatic Differentiation of Human Liver Organoids.
Krüger M, Oosterhoff LA, van Wolferen ME, Schiele SA, Walther A, Geijsen N, De Laporte L, van der Laan LJW, Kock LM, Spee B
Advanced healthcare materials 2020 Mar;9(6):e1901658
Advanced healthcare materials 2020 Mar;9(6):e1901658
Antrodia cinnamomea Confers Obesity Resistance and Restores Intestinal Barrier Integrity in Leptin-deficient Obese Mice.
Tsai YT, Ruan JW, Chang CS, Ko ML, Chou HC, Lin CC, Lin CM, Huang CT, Wei YS, Liao EC, Chen HY, Kao CY, Chan HL
Nutrients 2020 Mar 10;12(3)
Nutrients 2020 Mar 10;12(3)
Assembly and Function of a Bioengineered Human Liver for Transplantation Generated Solely from Induced Pluripotent Stem Cells.
Takeishi K, Collin de l'Hortet A, Wang Y, Handa K, Guzman-Lepe J, Matsubara K, Morita K, Jang S, Haep N, Florentino RM, Yuan F, Fukumitsu K, Tobita K, Sun W, Franks J, Delgado ER, Shapiro EM, Fraunhoffer NA, Duncan AW, Yagi H, Mashimo T, Fox IJ, Soto-Gutierrez A
Cell reports 2020 Jun 2;31(9):107711
Cell reports 2020 Jun 2;31(9):107711
Neurotrophic Factors Protect the Intestinal Barrier from Rotavirus Insult in Mice.
Hagbom M, De Faria FM, Winberg ME, Westerberg S, Nordgren J, Sharma S, Keita ÅV, Loitto V, Magnusson KE, Svensson L
mBio 2020 Jan 21;11(1)
mBio 2020 Jan 21;11(1)
Elevated TRPV4 Levels Contribute to Endothelial Damage and Scarring in Experimental Spinal Cord Injury.
Kumar H, Lim CS, Choi H, Joshi HP, Kim KT, Kim YH, Park CK, Kim HM, Han IB
The Journal of neuroscience : the official journal of the Society for Neuroscience 2020 Feb 26;40(9):1943-1955
The Journal of neuroscience : the official journal of the Society for Neuroscience 2020 Feb 26;40(9):1943-1955
Retinal Phenotype in the rd9 Mutant Mouse, a Model of X-Linked RP.
Falasconi A, Biagioni M, Novelli E, Piano I, Gargini C, Strettoi E
Frontiers in neuroscience 2019;13:991
Frontiers in neuroscience 2019;13:991
EspF is crucial for Citrobacter rodentium-induced tight junction disruption and lethality in immunocompromised animals.
Xia X, Liu Y, Hodgson A, Xu D, Guo W, Yu H, She W, Zhou C, Lan L, Fu K, Vallance BA, Wan F
PLoS pathogens 2019 Jun;15(6):e1007898
PLoS pathogens 2019 Jun;15(6):e1007898
Non-canonical Wnt signaling regulates junctional mechanocoupling during angiogenic collective cell migration.
Carvalho JR, Fortunato IC, Fonseca CG, Pezzarossa A, Barbacena P, Dominguez-Cejudo MA, Vasconcelos FF, Santos NC, Carvalho FA, Franco CA
eLife 2019 Jun 27;8
eLife 2019 Jun 27;8
ROCK2 inhibition triggers the collective invasion of colorectal adenocarcinomas.
Libanje F, Raingeaud J, Luan R, Thomas Z, Zajac O, Veiga J, Marisa L, Adam J, Boige V, Malka D, Goéré D, Hall A, Soazec JY, Prall F, Gelli M, Dartigues P, Jaulin F
The EMBO journal 2019 Jul 15;38(14):e99299
The EMBO journal 2019 Jul 15;38(14):e99299
Organoid single cell profiling identifies a transcriptional signature of glomerular disease.
Harder JL, Menon R, Otto EA, Zhou J, Eddy S, Wys NL, O'Connor C, Luo J, Nair V, Cebrian C, Spence JR, Bitzer M, Troyanskaya OG, Hodgin JB, Wiggins RC, Freedman BS, Kretzler M, European Renal cDNA Bank (ERCB), Nephrotic Syndrome Study Network (NEPTUNE)
JCI insight 2019 Jan 10;4(1)
JCI insight 2019 Jan 10;4(1)
Inhibitory Effects of Sodium Alginate on Hepatic Steatosis in Mice Induced by a Methionine- and Choline-deficient Diet.
Kawauchi S, Horibe S, Sasaki N, Tanahashi T, Mizuno S, Hamaguchi T, Rikitake Y
Marine drugs 2019 Feb 9;17(2)
Marine drugs 2019 Feb 9;17(2)
Improved In Vitro Model for Intranasal Mucosal Drug Delivery: Primary Olfactory and Respiratory Epithelial Cells Compared with the Permanent Nasal Cell Line RPMI 2650.
Ladel S, Schlossbauer P, Flamm J, Luksch H, Mizaikoff B, Schindowski K
Pharmaceutics 2019 Aug 1;11(8)
Pharmaceutics 2019 Aug 1;11(8)
Stabilization of HIF-2α impacts pancreas growth.
Flores-Martínez A, García-Núñez A, Rojas A, Cano DA
Scientific reports 2018 Sep 12;8(1):13713
Scientific reports 2018 Sep 12;8(1):13713
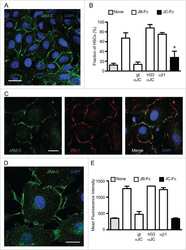
Characterization of human fetal brain endothelial cells reveals barrier properties suitable for in vitro modeling of the BBB with syngenic co-cultures.
Andrews AM, Lutton EM, Cannella LA, Reichenbach N, Razmpour R, Seasock MJ, Kaspin SJ, Merkel SF, Langford D, Persidsky Y, Ramirez SH
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2018 May;38(5):888-903
Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2018 May;38(5):888-903
Patient-iPSC-Derived Kidney Organoids Show Functional Validation of a Ciliopathic Renal Phenotype and Reveal Underlying Pathogenetic Mechanisms.
Forbes TA, Howden SE, Lawlor K, Phipson B, Maksimovic J, Hale L, Wilson S, Quinlan C, Ho G, Holman K, Bennetts B, Crawford J, Trnka P, Oshlack A, Patel C, Mallett A, Simons C, Little MH
American journal of human genetics 2018 May 3;102(5):816-831
American journal of human genetics 2018 May 3;102(5):816-831
Next generation histology methods for three-dimensional imaging of fresh and archival human brain tissues.
Lai HM, Liu AKL, Ng HHM, Goldfinger MH, Chau TW, DeFelice J, Tilley BS, Wong WM, Wu W, Gentleman SM
Nature communications 2018 Mar 14;9(1):1066
Nature communications 2018 Mar 14;9(1):1066
Galectin-3 modulates the polarized surface delivery of β1-integrin in epithelial cells.
Hönig E, Ringer K, Dewes J, von Mach T, Kamm N, Kreitzer G, Jacob R
Journal of cell science 2018 Jun 11;131(11)
Journal of cell science 2018 Jun 11;131(11)
Wdpcp promotes epicardial EMT and epicardium-derived cell migration to facilitate coronary artery remodeling.
Liu X, Wang Y, Liu F, Zhang M, Song H, Zhou B, Lo CW, Tong S, Hu Z, Zhang Z
Science signaling 2018 Feb 27;11(519)
Science signaling 2018 Feb 27;11(519)
Cadherin-6B proteolytic N-terminal fragments promote chick cranial neural crest cell delamination by regulating extracellular matrix degradation.
Schiffmacher AT, Adomako-Ankomah A, Xie V, Taneyhill LA
Developmental biology 2018 Dec 1;444 Suppl 1(Suppl 1):S237-S251
Developmental biology 2018 Dec 1;444 Suppl 1(Suppl 1):S237-S251
Multifaceted Mechanisms of WY-14643 to Stabilize the Blood-Brain Barrier in a Model of Traumatic Brain Injury.
Neuhaus W, Krämer T, Neuhoff A, Gölz C, Thal SC, Förster CY
Frontiers in molecular neuroscience 2017;10:149
Frontiers in molecular neuroscience 2017;10:149
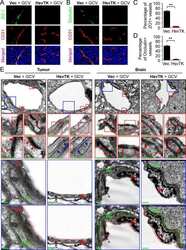
Targeting Glioma Stem Cell-Derived Pericytes Disrupts the Blood-Tumor Barrier and Improves Chemotherapeutic Efficacy.
Zhou W, Chen C, Shi Y, Wu Q, Gimple RC, Fang X, Huang Z, Zhai K, Ke SQ, Ping YF, Feng H, Rich JN, Yu JS, Bao S, Bian XW
Cell stem cell 2017 Nov 2;21(5):591-603.e4
Cell stem cell 2017 Nov 2;21(5):591-603.e4
Complement C5aR1 Signaling Promotes Polarization and Proliferation of Embryonic Neural Progenitor Cells through PKCζ.
Coulthard LG, Hawksworth OA, Li R, Balachandran A, Lee JD, Sepehrband F, Kurniawan N, Jeanes A, Simmons DG, Wolvetang E, Woodruff TM
The Journal of neuroscience : the official journal of the Society for Neuroscience 2017 May 31;37(22):5395-5407
The Journal of neuroscience : the official journal of the Society for Neuroscience 2017 May 31;37(22):5395-5407
Massively Increased Caries Susceptibility in an Irf6 Cleft Lip/Palate Model.
Tamasas B, Cox TC
Journal of dental research 2017 Mar;96(3):315-322
Journal of dental research 2017 Mar;96(3):315-322
In Vitro Study of Receptor-Mediated Silica Nanoparticles Delivery across Blood-Brain Barrier.
Song Y, Du D, Li L, Xu J, Dutta P, Lin Y
ACS applied materials & interfaces 2017 Jun 21;9(24):20410-20416
ACS applied materials & interfaces 2017 Jun 21;9(24):20410-20416
Contractile forces at tricellular contacts modulate epithelial organization and monolayer integrity.
Salomon J, Gaston C, Magescas J, Duvauchelle B, Canioni D, Sengmanivong L, Mayeux A, Michaux G, Campeotto F, Lemale J, Viala J, Poirier F, Minc N, Schmitz J, Brousse N, Ladoux B, Goulet O, Delacour D
Nature communications 2017 Jan 13;8:13998
Nature communications 2017 Jan 13;8:13998
E-cadherin cleavage by MT2-MMP regulates apical junctional signaling and epithelial homeostasis in the intestine.
Gómez-Escudero J, Moreno V, Martín-Alonso M, Hernández-Riquer MV, Feinberg T, Colmenar Á, Calvo E, Camafeita E, Martínez F, Oudhoff MJ, Weiss SJ, Arroyo AG
Journal of cell science 2017 Dec 1;130(23):4013-4027
Journal of cell science 2017 Dec 1;130(23):4013-4027
Systemic Injection of RPE65-Programmed Bone Marrow-Derived Cells Prevents Progression of Chronic Retinal Degeneration.
Qi X, Pay SL, Yan Y, Thomas J Jr, Lewin AS, Chang LJ, Grant MB, Boulton ME
Molecular therapy : the journal of the American Society of Gene Therapy 2017 Apr 5;25(4):917-927
Molecular therapy : the journal of the American Society of Gene Therapy 2017 Apr 5;25(4):917-927
Silencing microRNA-143 protects the integrity of the blood-brain barrier: implications for methamphetamine abuse.
Bai Y, Zhang Y, Hua J, Yang X, Zhang X, Duan M, Zhu X, Huang W, Chao J, Zhou R, Hu G, Yao H
Scientific reports 2016 Oct 21;6:35642
Scientific reports 2016 Oct 21;6:35642
Enhanced Re-Endothelialization of Decellularized Rat Lungs.
Stabler CT, Caires LC Jr, Mondrinos MJ, Marcinkiewicz C, Lazarovici P, Wolfson MR, Lelkes PI
Tissue engineering. Part C, Methods 2016 May;22(5):439-50
Tissue engineering. Part C, Methods 2016 May;22(5):439-50
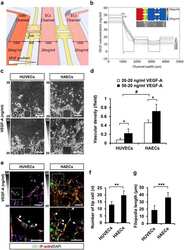
Intrinsic FGF2 and FGF5 promotes angiogenesis of human aortic endothelial cells in 3D microfluidic angiogenesis system.
Seo HR, Jeong HE, Joo HJ, Choi SC, Park CY, Kim JH, Choi JH, Cui LH, Hong SJ, Chung S, Lim DS
Scientific reports 2016 Jun 30;6:28832
Scientific reports 2016 Jun 30;6:28832
Isolation, culture and characterization of primary mouse RPE cells.
Fernandez-Godino R, Garland DL, Pierce EA
Nature protocols 2016 Jul;11(7):1206-18
Nature protocols 2016 Jul;11(7):1206-18
Murine junctional adhesion molecules JAM-B and JAM-C mediate endothelial and stellate cell interactions during hepatic fibrosis.
Hintermann E, Bayer M, Ehser J, Aurrand-Lions M, Pfeilschifter JM, Imhof BA, Christen U
Cell adhesion & migration 2016 Jul 3;10(4):419-33
Cell adhesion & migration 2016 Jul 3;10(4):419-33
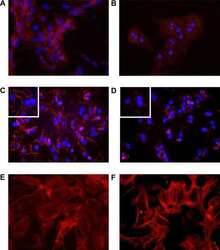
MICAL2 is a novel human cancer gene controlling mesenchymal to epithelial transition involved in cancer growth and invasion.
Mariotti S, Barravecchia I, Vindigni C, Pucci A, Balsamo M, Libro R, Senchenko V, Dmitriev A, Jacchetti E, Cecchini M, Roviello F, Lai M, Broccoli V, Andreazzoli M, Mazzanti CM, Angeloni D
Oncotarget 2016 Jan 12;7(2):1808-25
Oncotarget 2016 Jan 12;7(2):1808-25
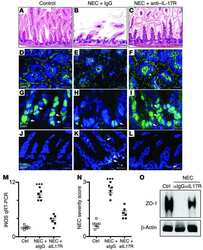
Toll-like receptor 4-mediated lymphocyte influx induces neonatal necrotizing enterocolitis.
Egan CE, Sodhi CP, Good M, Lin J, Jia H, Yamaguchi Y, Lu P, Ma C, Branca MF, Weyandt S, Fulton WB, Niño DF, Prindle T Jr, Ozolek JA, Hackam DJ
The Journal of clinical investigation 2016 Feb;126(2):495-508
The Journal of clinical investigation 2016 Feb;126(2):495-508
Evaluation of Planar-Cell-Polarity Phenotypes in Ciliopathy Mouse Mutant Cochlea.
May-Simera H
Journal of visualized experiments : JoVE 2016 Feb 21;(108):53559
Journal of visualized experiments : JoVE 2016 Feb 21;(108):53559
Lung Microtissue Array to Screen the Fibrogenic Potential of Carbon Nanotubes.
Chen Z, Wang Q, Asmani M, Li Y, Liu C, Li C, Lippmann JM, Wu Y, Zhao R
Scientific reports 2016 Aug 11;6:31304
Scientific reports 2016 Aug 11;6:31304
A Novel Dynamic Neonatal Blood-Brain Barrier on a Chip.
Deosarkar SP, Prabhakarpandian B, Wang B, Sheffield JB, Krynska B, Kiani MF
PloS one 2015;10(11):e0142725
PloS one 2015;10(11):e0142725
Liver Sinusoidal Endothelial Cells Escape Senescence by Loss of p19ARF.
Koudelkova P, Weber G, Mikulits W
PloS one 2015;10(11):e0142134
PloS one 2015;10(11):e0142134
The pH-sensing receptor OGR1 improves barrier function of epithelial cells and inhibits migration in an acidic environment.
de Vallière C, Vidal S, Clay I, Jurisic G, Tcymbarevich I, Lang S, Ludwig MG, Okoniewski M, Eloranta JJ, Kullak-Ublick GA, Wagner CA, Rogler G, Seuwen K
American journal of physiology. Gastrointestinal and liver physiology 2015 Sep 15;309(6):G475-90
American journal of physiology. Gastrointestinal and liver physiology 2015 Sep 15;309(6):G475-90
Immunohistochemical expression of VDR is associated with reduced integrity of tight junction complex in psoriatic skin.
Visconti B, Paolino G, Carotti S, Pendolino AL, Morini S, Richetta AG, Calvieri S
Journal of the European Academy of Dermatology and Venereology : JEADV 2015 Oct;29(10):2038-42
Journal of the European Academy of Dermatology and Venereology : JEADV 2015 Oct;29(10):2038-42
Upregulation of Shiga toxin receptor CD77/Gb3 and interleukin-1β expression in the brain of EHEC patients with hemolytic uremic syndrome and neurologic symptoms.
Hagel C, Krasemann S, Löffler J, Püschel K, Magnus T, Glatzel M
Brain pathology (Zurich, Switzerland) 2015 Mar;25(2):146-56
Brain pathology (Zurich, Switzerland) 2015 Mar;25(2):146-56
Acid sphingomyelinase inhibition protects mice from lung edema and lethal Staphylococcus aureus sepsis.
Peng H, Li C, Kadow S, Henry BD, Steinmann J, Becker KA, Riehle A, Beckmann N, Wilker B, Li PL, Pritts T, Edwards MJ, Zhang Y, Gulbins E, Grassmé H
Journal of molecular medicine (Berlin, Germany) 2015 Jun;93(6):675-89
Journal of molecular medicine (Berlin, Germany) 2015 Jun;93(6):675-89
Constraint of gene expression by the chromatin remodelling protein CHD4 facilitates lineage specification.
O'Shaughnessy-Kirwan A, Signolet J, Costello I, Gharbi S, Hendrich B
Development (Cambridge, England) 2015 Aug 1;142(15):2586-97
Development (Cambridge, England) 2015 Aug 1;142(15):2586-97
Impaired intestinal mucosal barrier upon ischemia-reperfusion: "patching holes in the shield with a simple surgical method".
Rosero O, Ónody P, Kovács T, Molnár D, Lotz G, Tóth S, Turóczi Z, Fülöp A, Garbaisz D, Harsányi L, Szijártó A
BioMed research international 2014;2014:210901
BioMed research international 2014;2014:210901
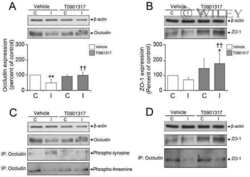
Phosphorylation decreases ubiquitylation of the thiazide-sensitive cotransporter NCC and subsequent clathrin-mediated endocytosis.
Rosenbaek LL, Kortenoeven ML, Aroankins TS, Fenton RA
The Journal of biological chemistry 2014 May 9;289(19):13347-61
The Journal of biological chemistry 2014 May 9;289(19):13347-61
Retinal pigment epithelial cell death by the alternative complement cascade: role of membrane regulatory proteins, calcium, PKC, and oxidative stress.
Yang P, Baciu P, Kerrigan BC, Etheridge M, Sung E, Toimil BA, Berchuck JE, Jaffe GJ
Investigative ophthalmology & visual science 2014 May 6;55(5):3012-21
Investigative ophthalmology & visual science 2014 May 6;55(5):3012-21
Twist1-induced dissemination preserves epithelial identity and requires E-cadherin.
Shamir ER, Pappalardo E, Jorgens DM, Coutinho K, Tsai WT, Aziz K, Auer M, Tran PT, Bader JS, Ewald AJ
The Journal of cell biology 2014 Mar 3;204(5):839-56
The Journal of cell biology 2014 Mar 3;204(5):839-56
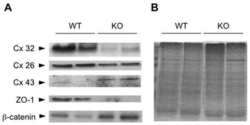
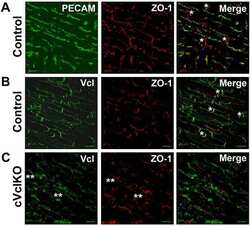
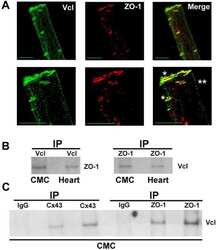
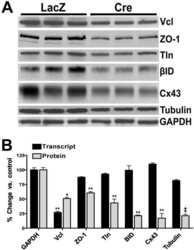
Vinculin directly binds zonula occludens-1 and is essential for stabilizing connexin-43-containing gap junctions in cardiac myocytes.
Zemljic-Harpf AE, Godoy JC, Platoshyn O, Asfaw EK, Busija AR, Domenighetti AA, Ross RS
Journal of cell science 2014 Mar 1;127(Pt 5):1104-16
Journal of cell science 2014 Mar 1;127(Pt 5):1104-16
Disruption of astrocyte-vascular coupling and the blood-brain barrier by invading glioma cells.
Watkins S, Robel S, Kimbrough IF, Robert SM, Ellis-Davies G, Sontheimer H
Nature communications 2014 Jun 19;5:4196
Nature communications 2014 Jun 19;5:4196
An in vitro blood-brain barrier model combining shear stress and endothelial cell/astrocyte co-culture.
Takeshita Y, Obermeier B, Cotleur A, Sano Y, Kanda T, Ransohoff RM
Journal of neuroscience methods 2014 Jul 30;232:165-72
Journal of neuroscience methods 2014 Jul 30;232:165-72
Intestinal and systemic inflammatory responses are positively associated with sulfidogenic bacteria abundance in high-fat-fed male C57BL/6J mice.
Shen W, Wolf PG, Carbonero F, Zhong W, Reid T, Gaskins HR, McIntosh MK
The Journal of nutrition 2014 Aug;144(8):1181-7
The Journal of nutrition 2014 Aug;144(8):1181-7
MUC4 overexpression augments cell migration and metastasis through EGFR family proteins in triple negative breast cancer cells.
Mukhopadhyay P, Lakshmanan I, Ponnusamy MP, Chakraborty S, Jain M, Pai P, Smith LM, Lele SM, Batra SK
PloS one 2013;8(2):e54455
PloS one 2013;8(2):e54455
Polarity, cell division, and out-of-equilibrium dynamics control the growth of epithelial structures.
Cerruti B, Puliafito A, Shewan AM, Yu W, Combes AN, Little MH, Chianale F, Primo L, Serini G, Mostov KE, Celani A, Gamba A
The Journal of cell biology 2013 Oct 28;203(2):359-72
The Journal of cell biology 2013 Oct 28;203(2):359-72
Luminal mitosis drives epithelial cell dispersal within the branching ureteric bud.
Packard A, Georgas K, Michos O, Riccio P, Cebrian C, Combes AN, Ju A, Ferrer-Vaquer A, Hadjantonakis AK, Zong H, Little MH, Costantini F
Developmental cell 2013 Nov 11;27(3):319-30
Developmental cell 2013 Nov 11;27(3):319-30
Heme modulates intestinal epithelial cell activation: involvement of NADPHox-derived ROS signaling.
Barcellos-de-Souza P, Moraes JA, de-Freitas-Junior JC, Morgado-Díaz JA, Barja-Fidalgo C, Arruda MA
American journal of physiology. Cell physiology 2013 Jan 15;304(2):C170-9
American journal of physiology. Cell physiology 2013 Jan 15;304(2):C170-9
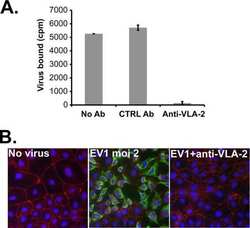
Echovirus 1 entry into polarized Caco-2 cells depends on dynamin, cholesterol, and cellular factors associated with macropinocytosis.
Krieger SE, Kim C, Zhang L, Marjomaki V, Bergelson JM
Journal of virology 2013 Aug;87(16):8884-95
Journal of virology 2013 Aug;87(16):8884-95
Evidence for phenotypic plasticity in aggressive triple-negative breast cancer: human biology is recapitulated by a novel model system.
D'Amato NC, Ostrander JH, Bowie ML, Sistrunk C, Borowsky A, Cardiff RD, Bell K, Young LJ, Simin K, Bachelder RE, Delrow J, Dawson A, Yee LD, Mrózek K, Clay TM, Osada T, Seewaldt VL
PloS one 2012;7(9):e45684
PloS one 2012;7(9):e45684
The LKB1 tumor suppressor controls spindle orientation and localization of activated AMPK in mitotic epithelial cells.
Wei C, Bhattaram VK, Igwe JC, Fleming E, Tirnauer JS
PloS one 2012;7(7):e41118
PloS one 2012;7(7):e41118
C-reactive protein increases BBB permeability: implications for obesity and neuroinflammation.
Hsuchou H, Kastin AJ, Mishra PK, Pan W
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2012;30(5):1109-19
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2012;30(5):1109-19
Integrin-linked kinase deletion in the developing lens leads to capsule rupture, impaired fiber migration and non-apoptotic epithelial cell death.
Cammas L, Wolfe J, Choi SY, Dedhar S, Beggs HE
Investigative ophthalmology & visual science 2012 May 17;53(6):3067-81
Investigative ophthalmology & visual science 2012 May 17;53(6):3067-81
Liver X receptor activation enhances blood-brain barrier integrity in the ischemic brain and increases the abundance of ATP-binding cassette transporters ABCB1 and ABCC1 on brain capillary cells.
ElAli A, Hermann DM
Brain pathology (Zurich, Switzerland) 2012 Mar;22(2):175-87
Brain pathology (Zurich, Switzerland) 2012 Mar;22(2):175-87
The tight junction scaffolding protein cingulin regulates neural crest cell migration.
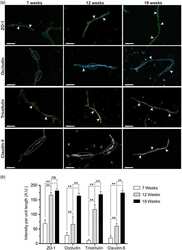
Wu CY, Jhingory S, Taneyhill LA
Developmental dynamics : an official publication of the American Association of Anatomists 2011 Oct;240(10):2309-23
Developmental dynamics : an official publication of the American Association of Anatomists 2011 Oct;240(10):2309-23
Lung self-assembly is modulated by tissue surface tensions.
Schwarz MA, Zheng H, Legan S, Foty RA
American journal of respiratory cell and molecular biology 2011 May;44(5):682-91
American journal of respiratory cell and molecular biology 2011 May;44(5):682-91
Cx50 requires an intact PDZ-binding motif and ZO-1 for the formation of functional intercellular channels.
Chai Z, Goodenough DA, Paul DL
Molecular biology of the cell 2011 Dec;22(23):4503-12
Molecular biology of the cell 2011 Dec;22(23):4503-12
Renal ischemia-reperfusion injury causes intercalated cell-specific disruption of occludin in the collecting duct.
Lee SY, Shin JA, Kwon HM, Weiner ID, Han KH
Histochemistry and cell biology 2011 Dec;136(6):637-47
Histochemistry and cell biology 2011 Dec;136(6):637-47
S-Adenosylmethionine regulates connexins sub-types expressed by hepatocytes.
Yamaji S, Droggiti A, Lu SC, Martinez-Chantar ML, Warner A, Varela-Rey M
European journal of cell biology 2011 Apr;90(4):312-22
European journal of cell biology 2011 Apr;90(4):312-22
Hypoxia stimulates hepatocyte epithelial to mesenchymal transition by hypoxia-inducible factor and transforming growth factor-beta-dependent mechanisms.
Copple BL
Liver international : official journal of the International Association for the Study of the Liver 2010 May;30(5):669-82
Liver international : official journal of the International Association for the Study of the Liver 2010 May;30(5):669-82
Embryonic vascular endothelial cells are malleable to reprogramming via Prox1 to a lymphatic gene signature.
Kim H, Nguyen VP, Petrova TV, Cruz M, Alitalo K, Dumont DJ
BMC developmental biology 2010 Jun 28;10:72
BMC developmental biology 2010 Jun 28;10:72
Adenovirus-driven overexpression of proteinases in organ-cultured normal human corneas leads to diabetic-like changes.
Saghizadeh M, Kramerov AA, Yaghoobzadeh Y, Hu J, Ljubimova JY, Black KL, Castro MG, Ljubimov AV
Brain research bulletin 2010 Feb 15;81(2-3):262-72
Brain research bulletin 2010 Feb 15;81(2-3):262-72
The canonical NF-kappaB pathway governs mammary tumorigenesis in transgenic mice and tumor stem cell expansion.
Liu M, Sakamaki T, Casimiro MC, Willmarth NE, Quong AA, Ju X, Ojeifo J, Jiao X, Yeow WS, Katiyar S, Shirley LA, Joyce D, Lisanti MP, Albanese C, Pestell RG
Cancer research 2010 Dec 15;70(24):10464-73
Cancer research 2010 Dec 15;70(24):10464-73
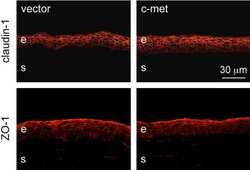
Normalization of wound healing and diabetic markers in organ cultured human diabetic corneas by adenoviral delivery of c-Met gene.
Saghizadeh M, Kramerov AA, Yu FS, Castro MG, Ljubimov AV
Investigative ophthalmology & visual science 2010 Apr;51(4):1970-80
Investigative ophthalmology & visual science 2010 Apr;51(4):1970-80
Zonula occludens-1, occludin, and E-cadherin protein expression in biliary tract cancers.
Németh Z, Szász AM, Somorácz A, Tátrai P, Németh J, Gyorffy H, Szíjártó A, Kupcsulik P, Kiss A, Schaff Z
Pathology oncology research : POR 2009 Sep;15(3):533-9
Pathology oncology research : POR 2009 Sep;15(3):533-9
Depletion of passenger leukocytes from corneal grafts: an effective means of promoting transplant survival?
Zhang X, Shen L, Jin Y, Saban DR, Chauhan SK, Dana R
Investigative ophthalmology & visual science 2009 Jul;50(7):3137-44
Investigative ophthalmology & visual science 2009 Jul;50(7):3137-44
Disruptions of occludin and claudin-5 in brain endothelial cells in vitro and in brains of mice with acute liver failure.
Chen F, Ohashi N, Li W, Eckman C, Nguyen JH
Hepatology (Baltimore, Md.) 2009 Dec;50(6):1914-23
Hepatology (Baltimore, Md.) 2009 Dec;50(6):1914-23
Beta-catenin is necessary to keep cells of ureteric bud/Wolffian duct epithelium in a precursor state.
Marose TD, Merkel CE, McMahon AP, Carroll TJ
Developmental biology 2008 Feb 1;314(1):112-26
Developmental biology 2008 Feb 1;314(1):112-26
ZO-1 is required for protein kinase C gamma-driven disassembly of connexin 43.
Akoyev V, Takemoto DJ
Cellular signalling 2007 May;19(5):958-67
Cellular signalling 2007 May;19(5):958-67
Expression of zonula occludens-1 (ZO-1) and the transcription factor ZO-1-associated nucleic acid-binding protein (ZONAB)-MsY3 in glial cells and colocalization at oligodendrocyte and astrocyte gap junctions in mouse brain.
Penes MC, Li X, Nagy JI
The European journal of neuroscience 2005 Jul;22(2):404-18
The European journal of neuroscience 2005 Jul;22(2):404-18
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
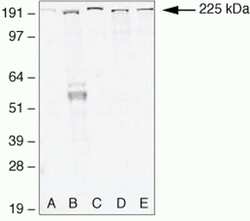
- Experimental details
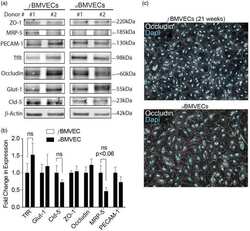
- Western blot analysis of (A) MDCKII, (B) A431, (C) Caco-2, (D) Rat-1, and (E) NRK-52 E cell lysates using Rabbit anti-ZO-1 (N-term) Polyclonal Antibody (Product # 40-2300).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
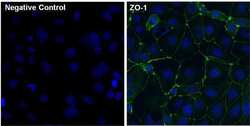
- Experimental details
- Immunofluorescent analysis of ZO-1 (green) in Caco-2 cells. The cells were fixed with 4% paraformaldehyde for 15 minutes and blocked with 3% Blocker BSA (Product # 37525) in PBS for 30 minutes at room temperature. Cells were stained with or without ZO-1 rabbit polyclonal antibody (Product # 40-2300), at a concentration of 5 µg/mL for 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:1000 for 1 hour at room temperature (both panels, green). Nuclei (both panels, blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
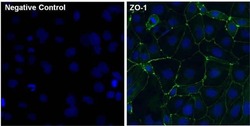
- Experimental details
- Immunofluorescent analysis of ZO-1 (green) in Caco-2 cells. The cells were fixed with 4% paraformaldehyde for 15 minutes and blocked with 3% Blocker BSA (Product # 37525) in PBS for 30 minutes at room temperature. Cells were stained with or without ZO-1 rabbit polyclonal antibody (Product # 40-2300), at a concentration of 5 µg/mL for 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:1000 for 1 hour at room temperature (both panels, green). Nuclei (both panels, blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
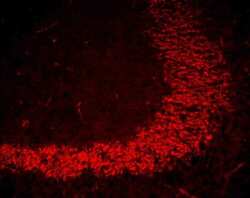
- Experimental details
- Immunofluorescent staining of mouse brain mossy fiber terminals using Rb anti-ZO-1 (N-term) (Product # 40-2300). Image courtesy of James I. Nagy, PhD, University of Manitoba, Canada.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent staining of blood vessels in mouse heart using Rb anti-ZO-1 (N-term) (Product # 40-2300). Image courtesy of James I. Nagy, PhD, University of Manitoba, Canada.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
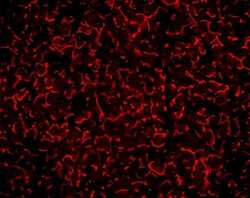
- Experimental details
- Immunofluorescent staining of mouse liver tissue using Rb anti-ZO-1 (N-term) (Product # 40-2300). Image courtesy of James I. Nagy, PhD, University of Manitoba, Canada.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
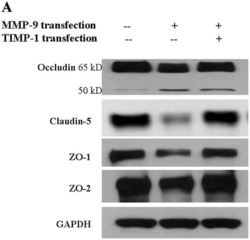
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
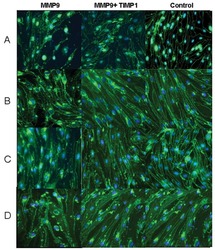
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
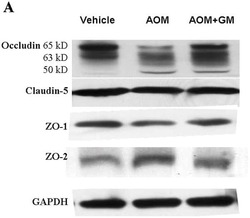
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
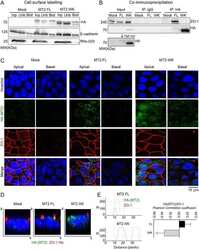
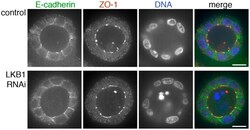
- Figure 3 ZO-1 and E-cadherin localization are unaffected by LKB1 RNAi in MDCK cell cysts. Representative images of ZO-1 (red) and E-cadherin (green) immunofluorescence in control MDCK cell cysts and in cysts with LKB1 RNAi are shown. Sections were taken through the midpoint of the cyst to show the hollow lumen as well as the apical and lateral surfaces of cells at the widest part of the cyst structure. Bars, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
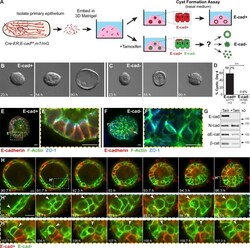
- Figure 1. E-cad deletion induced loss of simple epithelial architecture. (A) E-cad deletion was induced in half of Cre-ER;E-cad fl/fl organoids with tamoxifen. (B) Control, E-cad + organoids (-Tam) formed cysts. (C) E-cad - organoids (+Tam) failed to form cysts (28/42 movies across three biological replicates) or transiently established and then lost lumens (14/42 movies). (D) E-cad deletion blocked cyst formation. n , total number of organoids; r , number of biological replicates. Error bars indicate SD. ***, P = 0.0004, two-tailed Student's t test with equal variance. (E) Control organoids formed cysts with enrichment of E-cad and ZO-1 along apicolateral membranes (E'). (F) E-cad - organoids were multilayered, lacked E-cad immunoreactivity, and displayed abnormal ZO-1 localization. Arrow indicates rare E-cad + cells. (G) By Western blot, E-cad deletion (+Tam) resulted in complete loss of E-cad protein and significant reductions in alphaE-catenin and beta-catenin (see also Fig. S1, A and B ). Whole cell lysate samples were loaded for equal protein based on BCA analysis. (H) The Cre biosensor mT/mG was used to observe E-cad - cell behaviors by confocal microscopy ( Video 1 ). Cre + , E-cad - cells (green) changed shape, from columnar to round, before shifting apically (H' and H'', arrowheads). Gamma adjustments were performed in E and F to improve image clarity. Bars: (B and C) 20 um; (E, F, and H) 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
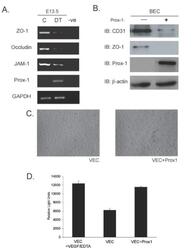
- Experimental details
- Figure 5 Overexpression of Prox1 results in changes in junctional proteins in vivo and in vitro . (A) RT-PCR analysis was performed on yolk sacs derived from E13.5 embryos to targets such as ZO-1, Occludin and JAM-1. (B) Similarly, in vitro analysis shows that overexpresion of Prox1 in vascular endothelial cells decreases junctional protein levels. (C) Bright field microscopy of VECs transfected with Prox1 (10x mag) (D) Vascular endothelial cells that overexpress Prox1 display an increase in permeability measured by FITC-albumin transit in a Boyden chamber assay. Figure is representative of three experiments with all samples run in triplicate. Error bars represent standard deviation of the mean. P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
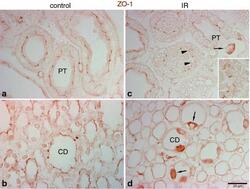
- Experimental details
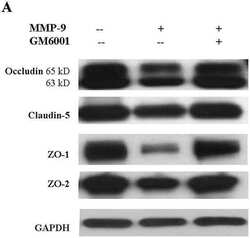
- Fig. 5 Effects of IR injury on ZO-1 expression in the outer medulla. Representative micrographs of ZO-1 localization in the outer medulla in control ( n = 5) and IR ( n = 5) kidneys. In control kidneys ( a , b ), ZO-1 ( brown ), immunoreactivity was observed in the proximal tubule ( PT ) as well as in the collecting duct ( CD ). In ischemic kidneys ( c , d ), ZO-1 localization was distorted and abnormally localized in the cytoplasm ( arrowheads , inset). Occasionally, a combination of both delocalization and upregulation of ZO-1 expression was observed in some cells ( arrows ) in the PT and CD
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 MUC4 contributes to a altered phenotype. ( A ) The control or MUC4 knockdown cells were seeded in 2% Matrigel on top of a 100% Matrigel layer, and fed with media every 3 days. After 7 days, acini-like structures were photographed under a phase-contrast microscope. The acini-like structures (examples shown in the boxes) that were regular (smooth and spherical shape) or irregular (irregular outline, multi-lobular) were counted and plotted as a percentage of the total count (p = 0.0005 for regular and p = 0.002 for irregular). A minimum of 120 structures was counted for each of control cells or MUC4 knockdown cells. Reduced irregular outline, multi-lobular and increased smooth and spherical shape colonies were found in MUC4 knock down cells when compared with control cells. ( B ) Structures were stained with the anti-ZO-1 antibody. 4,6-diamidino-2-phenylindole (DAPI) was used for nuclei staining. Optical sections (0.7-0.9 um) were captured using a laser scanning confocal microscope. The images presented here are the central planes of the acini. Bar = 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
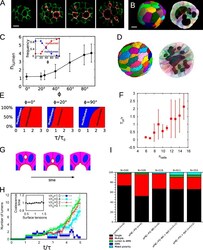
- Experimental details
- Figure 6. The ratio of lumen coalescence to cell division times controls the monolumen or multilumen phenotypes. (A) We treated cysts with aPKC-PSi to induce polarity disruption and spatially disordered spindle positioning. Cells were seeded in growth medium supplemented with 50 uM aPKC-PSi, and fixed 3 d later. Confocal scans for cysts treated with aPKC-PSi, which lack control over mitotic spindle positioning; stained for beta-catenin (green), labeling cell membranes; and ZO-1 (white) and PCX/gp135 (red), labeling luminal interfaces. Notice the multilumen morphology. Time from seeding: 3 d. Bar, 10 um. (B) 3D reconstructions of the cyst in A from the full set of confocal sections. Several disconnected lumens are visible in the cross-section (right). Bar, 10 um. (C) Mean number of luminal volumes and fraction of cysts with single lumen/multiple lumen phenotypes (inset), obtained from numerical simulations as a function of the tightness of the control over orientation of the cleavage plane. ph = 0deg, mitotic planes always orthogonal to the luminal surface. ph = 90deg, division planes chosen isotropically in three dimensions. Note that even for ph = 0deg, it is still possible to get more than one lumen. Simulated cysts have 32 cells. Error bars indicate standard deviations. (D) Numerical simulation of a multiple-lumen phenotype. The cleavage plane has been chosen at random, isotropically in 3D space. Equivalent time from seeding is around 5 d. (E) Mean fraction of cysts with s
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
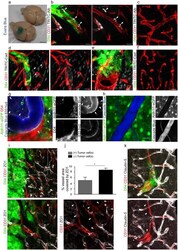
- Experimental details
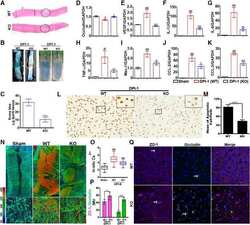
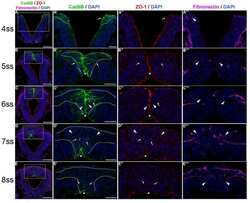
- Figure 3 Perivascular glioma cell co-option causes a breakdown of the blood-brain barrier The tracer, Evans blue, permeates into the brain parenchyma in tumor-bearing ( n =3 animals) (bottom), but not in sham mice 3 weeks post surgery ( n =3 animals) (top), Scale 5 mm. (a) . Immunofluorescence of CD31 and intravenously injected tracers, tetramethylrhodamine-albumin (TRITC-alb, white) or -cadaverine (TRITC-cad, white), and implanted eGFP-expressing human glioma cells ( D54 ) allow analysis of tracer leakage in relation to tumor burden. The large MW 70 kDa TRITC-albumin (b) , as well as the small MW 950 Da TRITC-cadaverine (d) can be found outside the vasculature (arrows) (b-f , Suppl. Fig.6 ) . Note that leaked TRITC-cadaverine is taken up by nearby neurons (arrows) (d,e) . No extravasation was seen in the absence of tumor cells (c,f) . To assess extravasation of the tracer Cascade blue (MW 10 kDa), Aldh1l1-eGFP-scid (eGFP) immunodeficient mice, which were previously implanted with TdTomato-expressing human glioma cells ( D54 ), were retro-orbitally injected with Alexa Fluor 633 hydrazide dye (633 hyd) and Cascade blue (g,h) . Accumulation of Cascade blue occurs in the brain's parenchyma indicating breakdown of the blood-brain barrier (BBB), where perivascular astrocytes have been displaced by glioma cell co-option (g) . Vessels lacking tumor cells do not show extravasation of the dye (h) . Immunohistochemistry for the TJ proteins zonula occludens-1 (ZO-1) (i) and claudin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
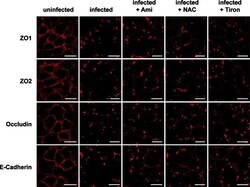
- Fig. 4 Endothelial cells were infected for 2 h with S. aureus (MOI 10:1) or left uninfected. As indicated, cells were pretreated for 20 min with amitriptyline (Ami) (20 muM), Tiron (10 mM), or NAC (10 mM) before infection with S. aureus . Immunofluorescence stainings were performed with antibodies against ZO1, ZO2, occludin, or E-cadherin for determination of the degradation of these TJ proteins. The presented pictures are representative of the results of at least three independent experiments. Scale bar is 25 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
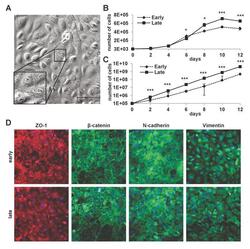
- Fig 1 Properties of cultured mLSECs. (A) Phase contrast microscopy of p19 ARF-/- mLSECs at passage 5. Inset show cells at higher magnification. (B) Proliferation kinetics by cell density analysis (upper panel) and proliferation kinetics by determination of cumulative cell numbers (lower panel) of mLSECs at early (p5) and late passages (p20). (C) Immunofluorescence analysis showing endothelial junction and intermediate filament proteins expressed in early and late passaged mLSECs. *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
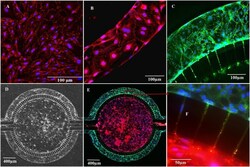
- Fig 4 Tight junction formation by neonatal RBEC under static and flow conditions as indicated by immunofluorescence staining of ZO-1. RBEC cultured for 5 days under static conditions were stained with ZO-1 (red) and Hoechst 33342 (blue) (A). RBEC cultured for 5 days under flow of RBEC medium in B 3 C were stained with ZO-1 (red) and Hoechst 33342 (blue) (B). RBEC cultured under flow of ACM for 5 days in B 3 C were stained with ZO-1 (green) and Hoechst 33342 (blue) (C). Bright field image of B 3 C showing RBEC in the vascular channels after 5 days of co-culture with rat astrocytes in the tissue compartment of B 3 C (D). Immunofluorescence staining of RBEC in vascular channel for ZO-1 (green) and astrocytes in tissue compartment for GFAP (red) (E). Magnified view of interface with pores from panel E showing staining of cells inside the pores, ZO-1 (green), GFAP (red) and nuclei (blue) (F).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
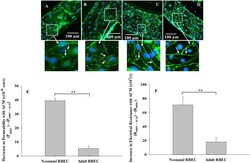
- Fig 8 Neonatal RBEC exhibit distinct barrier structure and function compared to adult RBEC in B 3 C. Neonatal RBEC alone (A) and neonatal RBEC + ACM (B) exhibit distinctly weaker and less organized ZO-1 staining (arrows) compared to adult RBEC alone (C) and adult RBEC + ACM (D). In the presence of ACM, neonatal RBEC exhibit discontinuous bands of ZO-1 staining (B) compared to neonatal RBEC in the absence of ACM where ZO-1 staining is discontinuous and granular (A). Presence of ACM has a significantly larger impact on both permeability (E) and electrical resistance (F) in neonatal RBEC compared to adult RBEC. Inset panels show higher magnification of white squared regions. (** indicates significant difference p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Abating MICAL2 in 786-O kidney cancer cells in vitro induces MET ( A ), Morphological analysis in light-transmitted microscopy. Scale bar: 100 mum. ( B ), F-actin staining. IF of: E-cadherin, ZO-1, catenin-beta. In KD2 and KD14 cells, these three markers relocated at cell perimeters (as in epithelial cells), rather than being diffusedly cytoplasmic. IF of vimentin with Vim13.2 antibody. In MIC2-KD cells it decorated short and disorganized filaments instead of long meshes observed in control cells. Scale bar of all IF images: 20 mum. Inset magnification: 3x. ( C ), Vimentin epitope recognized by V9 antibody was undetectable in KD2 and KD14 cells. ( D ), Agarose gel run of SNAI1, SNAI2, 18s5 RT-PCR products. ( E ), KD14 cells transfected with HA-MICAL2 cDNA. The elongated phenotype showed dose-dependency on HA-MICAL2 expression. One-way Anova test and Tukey's Multiple Comparison post-hoc test. In all graph bars, horizontal lines denote mean and SEM. N.s: non significant. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
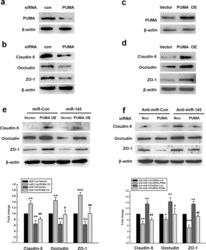
- Figure 5 miR-143 regulated the permeability of endothelial cells by targeting PUMA. (a,b) The transduction of cells with the PUMA siRNA lentivirus decreased the PUMA expression and the expression of tight junction proteins (claudin-5, occludin, and ZO-1) in HBMECs. (c,d) The transduction of HBMECs with the PUMA OE lentivirus increased the expression of PUMA and tight junction proteins (claudin-5, occludin, and ZO-1) in HBMECs. (e) The transduction of cells with miR-143 failed to decrease the level of tight junction proteins in the cells co-transduced with the PUMA OE lentivirus, as determined by western blot analysis. (f) The transduction of cells with the PUMA siRNA lentivirus significantly inhibited the anti-miR-143-induced increase in the expression of tight junction proteins, as determined by western blot analysis. All data are presented as the mean +- SD of three independent experiments. *p < 0.05, **p < 0.01 and ***p < 0.001 vs. the miR-Con/Vector group or the anti-miR-Con/siRNA-Con group; + p < 0.05 and ## p < 0.01 vs. the miR-143/Vector group or the anti-miR-143/siRNA-Con group using one-way ANOVA followed by the Holm-Sidak test. Meth, methamphetamine.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
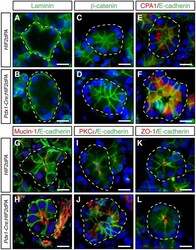
- Figure 4 Acini disorganization in HIF2alpha-overexpressing mice. No changes in the expression or localization of laminin ( A , B ), beta-catenin ( C , D ), and E-cadherin ( E - L ) between acini of two-week-old Pdx1-Cre;HIF2dPA and control mice. Carboxypeptidase A1 (CPA1) is localized close to the lumen of control acini ( E ). CPA1 does not show a clear apical localization in Pdx1-Cre;HIF2dPA acinar cells ( F ). Intracellular accumulation of mucin-1 ( H ), and PKCepsilon ( J ), in Pdx1-Cre;HIF2dPA acini while markers are normally localized at the apical region in control acini ( G , I ). Zonula occludens 1 (ZO-1) immunofluorescence show lumen dilation in Pdx1-Cre;HIF2dPA ( L ) acini compared to control acini ( K ). Nuclei are stained with DAPI (blue). Individual acini are outlined in yellow. Scale bars = 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
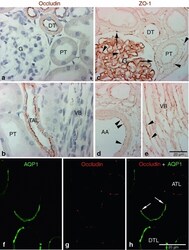
- Fig. 1 Occludin and ZO-1 expression in control kidney. Representative of occludin ( a , b ), ZO-1( c - e ), and occludin-AQP1 confocal ( f - h ) immunostaining images obtained from sham-operated control kidneys. Occludin ( brown ) immunoreactivity was observed as dot-and-line patterns at the apical end of the lateral membrane in tubular epithelial cells in the cortex ( a ) and outer medulla ( b ). Occludin was expressed primarily in the distal tubule ( DT ) including the thick ascending limb ( TAL ). No occludin immunolabel was detected in glomeruli ( G ), proximal tubule ( PT ), or blood vessels, including the vascular bundles ( VB ). ZO-1 ( brown ) immunoreactivity was observed in all tubular segments, including the proximal tubule ( PT ) and distal tubule ( DT ) ( c - e ). ZO-1 labeling also appeared in the glomerular parietal epithelial cells ( arrows ) and in endothelial cells ( arrowheads ) of all blood vessels including the glomerular capillary, peritubular capillary, arcuate artery ( AA ), and vascular bundle ( VB ). To identify the thin limbs of the loop of Henle, confocal immunofluorescence staining for occludin ( red ) and AQP1 ( green ) were performed ( f - h ). Occasionally, transition points ( white arrows ) from the AQP1-positive descending thin limb ( DTL ) to AQP1-negative ascending thin limb ( ATL ) were observed and occludin immunoreactivity appeared in both thin DTL and ATL segments ( g , h ). Results are representative of findings in the five control rat
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
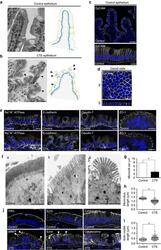
- Figure 1 Cell organization defects occur in the intestinal epithelium of CTE patients. ( a , b ) Histological analysis of hematoxylin-eosin stained paraffin sections of duodenal biopsies from control ( a ) and CTE patients ( b ). Scale bars, 10 mum. For each condition, a corresponding scheme of the epithelial organization is presented. CTE intestinal epithelium displays tufts (black arrowheads). ( c , d ) Confocal microscopy analysis of EpCAM distribution in duodenal control biopsy ( c ) or in Caco2 cells ( d ). Intestinal epithelium baseline is demarcated by dotted white line ( c ). Scale bars, upper panel 50 mum ( c ), lower panel 10 mum ( c ) and 10 mum ( d ). ( e ), Confocal microscopy analyses of Na + /K + -ATPase, E-cadherin, claudin-7 and ZO-1 distribution in control or CTE biopsies. N (Control) =6 biopsies, N (CTE) =6 biopsies. Scale bars, 10 mum. ( f ) Transmission electron microscopy (TEM) ultrastructural analysis of enterocyte apical membranes in control ( a ) and CTE ( b , c ) biopsies, showing actin rootlets ( black arrowheads ). Scale bars, 5 mum. N (Control) =3 biopsies, N (CTE) =3 biopsies. ( g ) Statistical analysis of microvillus density in control and CTE enterocytes. t test, * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2 EpCAM silencing leads to unusual tight junction positioning defects at tricellular junctions. ( a ) Western blot analysis of the EpCAM expression level in control ( Caco2 shNT ) or EpCAM-deprived ( Caco2 shEpCAM #1 and Caco2 shEpCAM #2 ) clones. GAPDH was used as loading control. ( b , c ) Confocal microscopy analysis of E-cadherin ( b ) and Scribble ( c ) on the apical or lateral sides in control and EpCAM-depleted cells. Scale bars, 5 mum. Nuclei were detected with Hoechst 33342 staining (blue). ( d , e ) 3D-SIM microscopy analysis of E-cadherin localization in control ( d ) and EpCAM-deprived ( e ) cells. Scale bars, 2 mum. ( f ) Cell shape analysis after E-cadherin immunostaining in control or EpCAM-deprived cells. Scale bars, 5 mum. ( g ), Statistical analysis of polygonal shape in control or EpCAM-deprived cells. Three independent experiments were carried out. N (Caco2shNT)=517 cells, N (Caco2shEpCAM#1)=550 cells, N (Caco2shEpCAM#2)=427 cells. Caco2 shNT cells with two cell edges=0%, three cell edges=0.455+-0.787%, four cell edges=8.673+-3.406%, five cell edges =48.154+-11.123%, six cell edges=35.652+-8.888%, more than six cell edges=7.067+-3.851 cells%. Caco2 shEpCAM#1 cells with two cell edges=1.822+-0.733 cells%, three cell edges=22.031+-5.906%, four cell edges=46.061+-3.851%, five cell edges=24.728+-4.344%, six cell edges=5.027+-2.210%, more than six cell edges=0.330+-0.572%. Caco2 shEpCAM#2 cells with two cell edges=0.966+-0.848%, three cell edges=12.542+-
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
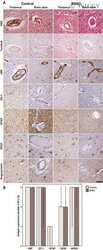
- Figure 3 Representative photographs of claudin-2, claudin-3, claudin-4, and zonula occludens-1 immunostained in longitudinal jejunal sections of the three experimental groups. Claudin-2 immunoreaction in the sham-operated group (a) was low and was mainly found in the cytoplasm of the intestinal epithelial cells. Claudin-2 expression in the groups exposed to ischemia-reperfusion was substantially higher and was principally found in the lateral membrane of the epithelial cells (b, c). The sham-operated group presented strong membrane claudin-3 immunostaining (d), which was remarkably decreased in the ischemia-reperfusion- (IR-) group (e), but not in the postconditioned- (PC-) group (f). Minimal claudin-4 immunostaining was observed in the jejunal samples of the sham-operated animals (g). The two groups challenged with 60 minutes of mesenteric ischemia and 6 hours of reperfusion (h, i) showed high membrane claudin-4 expression, which was limited to the tips of the villi. Zonula occludens-1 was strongly immunostained in the jejunal samples of the sham-operated animals (j). Following small intestinal ischemia-reperfusion, intestinal epithelial cell with zonula occludens-1 membrane immunostaining notably decreased in the IR-group (k); in the PC-group minimal loss of zonula occludens-1 membrane immunostaining was detected (l). Each photograph was taken with the same magnification. Scale bar: 20 mu m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
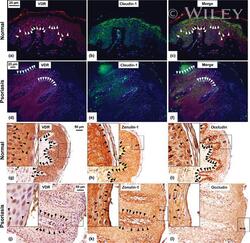
- Immunolabelling experiments of Vitamin D receptor (VDR) and tight junction proteins in normal and psoriatic skin. Photomicrographs of skin specimens labelled by double immunofluorescence (a-f). Immunofluorescent staining of VDR (red) (a,d) and claudin-1 (green) (b,e) in normal (a-c) and psoriatic (d-f) skin showed a larger number of claudin-1- and VDR-positive cells (arrowheads) in normal compared to psoriatic skin. VDR and zonulin-1/occludin immunohistochemistry (g-l). Immunohistochemical staining in serial section from normal (g-i) and psoriatic (j-l) skin showed an altered expression of zonulin-1/occludin localization in psoriasis compared to that in normal skin. The number of clearly positive cells (arrowheads) is slightly larger in normal compared to psoriatic skin for both zonulin-1 (h,k) and occludin (i,l). However, the distribution of zonulin- 1/occludin involved a thicker region of the upper layers of psoriatic epidermis compared to normal one (double-headed arrows). Zonulin-1 and occludin decorated plasma membrane of keratinocytes with a discontinuous pattern in psoriatic compared to normal skin (arrowheads, high power fields, h,i,k,l). VDR-positive cells were mainly localized in the same areas in which zonulin-1- and occludin-positive cells were found (arrowheads, g,j, standard and high power fields). Original magnification, x400 (a-c), x200 (d-l); high power fields (x400) calibration bar: 25 mum (a-f), 50 mum (g-l). VDR was stained in red using an ALEXA 568-labe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
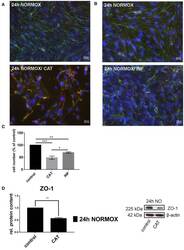
- Figure 1 Effects of catecholamines and inflammatory mediators under normoxia conditions on cEND cells. Cells were grown to confluence with subsequent differentiation. Catecholamines (CAT) or inflammatory mediators (INF) were applied for 24 h under normoxia conditions (24 h NORMOX). (A,B) Immunofluorescence staining of tight junction proteins claudin-5 (green) and ZO-1 (red) as markers of morphological changes of the endothelial cell monolayer. DAPI (blue) was used for staining of nuclei. Magnification 400 times, scale bar 20 mum. (C) Cell number after CAT and INF treatment normalized to control. (D) CAT induced changes of ZO-1 protein expression in cEND cells investigated by Western blot. Data is presented as the means (+- SEM) of n = 5 independent experiments. Altered protein expression was normalized to beta-actin. ZO-1 protein level changes are expressed as fold over untreated control, which was set 1. * P < 0.05, ** P < 0.01, *** P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
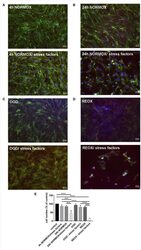
- Figure 2 Loss of morphological integrity of cEND cells exposed to elevated catecholamine levels and inflammatory mediators. Immunofluorescence staining of tight junction proteins claudin-5 (green) and ZO-1 (red) as markers of morphological changes of the endothelial cell monolayer. DAPI (blue) was used for staining nuclei. Cells were grown on cover slips to confluence. After differentiation cEND cells were treated with combination of catecholamines and inflammatory mediators (stress factors). Stress factors were administered under different incubation conditions. Magnification 400 times, scale bar 20 mum. (A) Stress factor application for 4h under normoxia conditions (4 h NORMOX). (B) Stress factor application for 24 h under normoxia conditions (24h NORMOX). (C) . Stress factor application for 4 h under oxygen glucose deprivation conditions (OGD). (D) Stress factor application for 4 h under OGD conditions with 20 h of subsequent reoxygenation under normoxia conditions (REOX). (E) Cell number in treatments shown in (A-D) normalized to control. *** P < 0.001, **** P < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
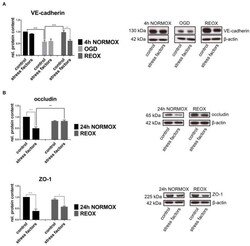
- Figure 3 Protein expression in cEND cells is changed by elevated catecholamine levels and inflammatory mediators under oxygen glucose deprivation conditions. Cells were grown to confluence with subsequent differentiation. Stress factors were applied under different incubation conditions. Protein levels were examined by Western blot. Changed protein expression was normalized to beta-actin. Changes of protein levels determined by densitometric analysis are expressed as fold over untreated control, which was set as 1. (A) VE-cadherin protein expression after stress factor administration under oxygen glucose deprivation conditions (OGD) and subsequent reoxygenation (REOX) in comparison with 4 h of normoxia (4 h NORMOX). Values are the means (+- SEM) of n = 8 independent experiments. (B) Occludin and ZO-1 protein levels changed by stress factors after reoxygenation conditions (REOX) compared to 24 h of normoxia conditions (24 h NORMOX). Values are the means (+- SEM) of n = 10 independent experiments. * P < 0.05, ** P < 0.01, *** P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
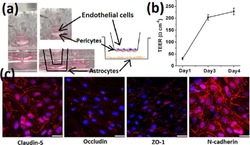
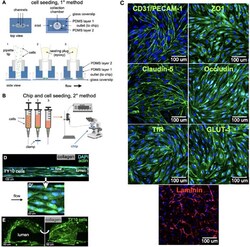
- FIGURE 2 Cell seeding procedures and formation of a human brainmicrovessel using TY10 cells. (A) Cell seeding, 1st method. Representation of the steps used to increase the cell concentration before injection into the chip. Cells are allowed to settle by gravity (left and central panels) in the cell seeder and then injected as a bolus into the hollow lumen. Prior to injection, the chamber is capped with a plugged pipette tip (central panel). (B) Cell seeding, 2nd method. Representation of the steps used to inject cells into the chip in a more controlled and uniform manner compared to the 1st method. Cells are allowed to settle at the bottom of a syringe, and are then delivered into the hollow lumen at constant flow controlled by a syringe pump. (C) Immunostaining of TY10 monolayers: endothelial cell marker PECAM-1/CD31, transporters Glut-1 and transferrin receptor (TfR), and tight junction proteins ZO-1, Occludin and Claudin-5. Staining for laminin provides evidence that TY10 cells deposit extracellular matrix while in culture. Scale bar, 100 mum. (D) Representative image of a chemically fixed sample of TY10 cells after they were grown in the brain microvessel-on-a-chip with medium flowing from left to right at 1 mµL/min for 7 days. Volumetric image was obtained using a spinning disk confocal microscope. Maximum z-projection is shown for a sample stained with DAPI (nuclei, blue) and phalloidin (actin, green). Scale bar, 100 mum. (D') Representative image of a chemically
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
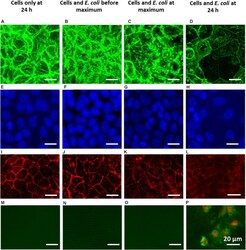
- FIGURE 5 Overlayer images of confocal stacks of Caco-2 BBe cell layers showing green-fluorescent cytoskeleton (A-D) and blue-fluorescent nuclei (E-H) , fluorescence images of red-fluorescent ZO-1 tight-junction proteins (I-L) and apoptosis of green-fluorescent membrane-damaged cells with red-fluorescent nuclei (M-P) . For comparison, a fluorescent image of apoptotic cells is shown in Supplementary Figure 3 . (A,E,I,M) 24 h Caco-2 BBe cell layers grown in the absence of an E. coli challenge. (B,F,J,N) Caco-2 BBe cell layers in the presence of an E. coli (10 4 mL - 1 ) challenge before the occurrence of the TEER maximum, i.e., at 4 h. (C,G,K,O) Caco-2 BBe cell layers grown in the presence of an E. coli (10 4 mL - 1 ) challenge at the TEER maximum, i.e., at 8 h. (D,H,L,P) Caco-2 BBe cell layers grown in the presence of an E. coli (10 4 mL - 1 ) challenge after the occurrence of the TEER maximum, i.e., after 24 h.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
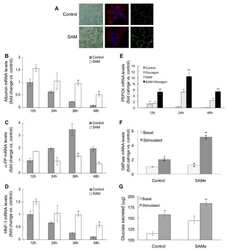
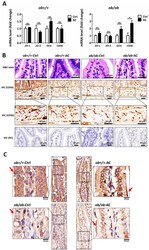
- Figure 5 The intestinal barrier is maintained by AC in ob/ob mice. ( A ) Relative mRNA expression of tight junction proteins and CD36 between ob+/+ -Ctrl and ob/ob -Ctrl mice; ( B ) The intestine was examined using hematoxylin and eosin staining and immunohistochemical staining of CD36. Magnification, 40x. Scale bars are 20 mum. The negative control for immunohistochemistry has been marked NC; ( C ) The intestine was examined using immunohistochemical staining for ZO-1. Magnification, 40x and 100x; scale bars are 50 mum and 10 mum, respectively. All data are expressed as mean +- SEM, * p < 0.05, ** p < 0.01, *** p < 0.001. Non-significant; ns. n = 4-8 mice in each group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 EEAC decreases intestinal permeability in the Caco-2 cell barrier model. ( A ) Cell viability assay of Caco-2 cells treated with 0-3000 muL/mL EEAC. ( B ) Effect of EEAC on intestinal permeability was measured using the trans-epithelial electrical resistance (TEER) assay. ( C ) Relative mRNA expression of tight junction proteins and PPARgamma were compared using controls (Ctrl). ( D ) Immunofluorescence staining of ZO-1 (green) of Caco-2 cells treated with or without EEAC. Nuclei were stained with DAPI (blue). The negative control is marked as NC. Scale bars, 20 mum. All data are expressed as mean +- SEM, * p < 0.05, ** p < 0.01, *** p < 0.001. Non-significant; ns. All experiments were repeated thrice ( n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
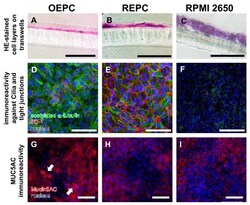
- Experimental details
- Figure 3 Characterisation of olfactory and respiratory primary cells in comparison to the standard cell line RPMI 2650. Monolayers are necessary to evaluate transport over the epithelial layer in the nasal mucosa: 14 um sections were made of olfactory epithelium primary cells (OEPC, A ), respiratory epithelial primary cells (REPC, B ) and RPMI 2650 ( C ) grown on a cell insert membrane for 21 days. Morphological features such as tight junctions and the formation of cilia are important influencing factors in investigations of drug permeation and clearance studies. Acetylated alpha-tubulin is a common marker for cilia [ 54 ]. IF double-staining of acetylated alpha-tubulin and the tight junction marker zonula occludens-1 (ZO-1) of ALI cultures of OEPC ( D ), REPC ( E ) and RPMI 2650 ( F ) after 21 days of incubation were made. An additional feature of mucosal cells is the ability to produce mucus. The marker protein mucin 5AC was used in this work, because the olfactory mucosa is to be simulated above all to investigate nose-to-brain transport. Again, IF was performed in OEPC ( G ), REPC ( H ) and RPMI 2650 ( I ). Scale bars: 100 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
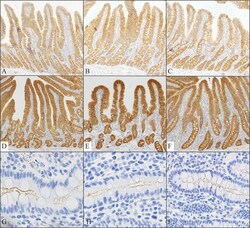
- Experimental details
- Fig 2 Representative immunohistochemistry images for Claudin 2 ( A-C ), Claudin 4 ( D-F ), and ZO-1 ( G-I ). Pigs were either PRRSV naive and fed ad libitum (Ad: A, D, and G ), PRRSV naive and pair-fed to PRRS+ pigs intake (PF: B, E, and H ), or challenged with PRRSV (PRRS+: C, F, and I ). Pigs were euthanized at days post inoculation (dpi) 17. Staining was limited to the epithelial cells, with higher stain density at cell membranes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Granulosa cell intercellular junctions are less abundant in Rspo2 Tg/Tg follicles. a Immunodetection analyses of FOXL2 and AMH (granulosa cells) at 21d in wild-type (WT) and Rspo2 Tg/Tg transplanted ovaries. In Rspo2 Tg/Tg ovaries, FOXL2 is still expressed in granulosa cells, but AMH expression is impaired compared to wild-type. b Immunodetection of LAMA1 (basal lamina), CDH2 (adherens junctions), and TJP1 (tight junctions) at 8d in WT and Rspo2 Tg/Tg transplanted ovaries. Follicles are surrounded by a continuous basal lamina in both genotypes as evidenced by LAMA1 immunostaining. CDH2 and TJP1 signals are decreased, notably at the interface between the oocyte and granulosa cells, indicating a deficit of cohesion in Rspo2 Tg/Tg follicles. Scale bar, 20 um. c This result is corroborated by quantitative analysis of CDH2 and TJP1 signal intensity on confocal sections. Data are presented as mean +- SEM. n = 5 WT and n = 9 Rspo2 Tg/Tg follicles from one transplanted ovary of each genotype. Student's t test, unpaired two sided (* p < 0.05; ** p < 0.01; *** p < 0.001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
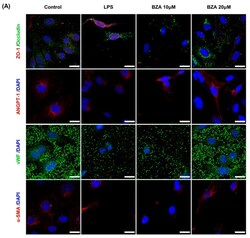
- Experimental details
- Figure 3 BZA increases angiogenesis and decreases BSCB destruction in vitro. ( A ) Representative immunofluorescent images (scale bar = 20 mum) of tight junction proteins such as ZO-1 and occludin and angiogenesis markers including ANGPT-1, vWF, and alpha-SMA examined in hCMEC/D3 cells. Quantitative fluorescence intensity for ( B ) ZO-1, ( C ) occludin, ( D ) ANGPT-1, ( E ) vWF, and ( F ) alpha-SMA. Data represent mean +- SEM ( n = 3 ( A , C ), n = 4 ( D ), n = 6 ( F ); performed in triplicate). ** p < 0.01, *** p < 0.001 (LPS-treated vs. control, DMSO), ## p < 0.01 (LPS-treated vs. LPS + BZA 10 muM), $ p < 0.05, $$ p < 0.01, $$$ p < 0.001 (LPS-treated vs. LPS + BZA 20 muM) , NS = Not significant, one-way ANOVA followed by the Bonferroni test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
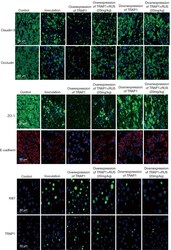
- Experimental details
- Figure 7 Ruscogenin attenuated the tight junction injury via regulating the expression of TRAP1. The expression of TRAP1, occludin, claudin-5, ZO-1, ki67, and E-cadherin expression in the liver cells stimulated by colorectal liver metastasis via regulating the expression of TRAP1. Representative images on TRAP1 (green), occludin (green), claudin-5 (green), ZO-1 (green), E-cadherin (red), and ki67 (green) in the liver tissues stimulated by colorectal liver metastasis were captured by immunofluorescence staining. Bar =50 um. TRAP1, tumor necrosis factor receptor associated protein 1; ZO-1, zonula occludens 1.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- The confocal imaging of bEnd.3 cells. Nuclei staining (blue). ZO1 staining (yellow). F-actin staining (red). Scale bar = 10 um. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
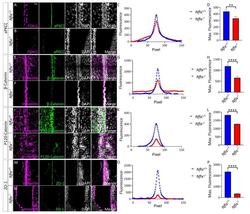
- Experimental details
- Figure 1 P5 Nfix -/- mice demonstrate abnormal localisation of key adhesion proteins to the cell junction. ( A , B , M , N ) Images of brain lateral ventricles from P5 Nfix +/+ ( A ) and Nfix -/- ( B ) mice marked with antibodies against ependymal marker FOXJ1 ( A , B ) or Vimentin ( M , N ) (purple), tight junction protein aPKCzeta ( A , B ) or ZO-1 ( M , N ) (green) and nuclear marker DAPI (grey) displayed separately and merged for each genotype. ( C , O ) Average fluorescence intensity of aPKCzeta ( C ) and ZO-1 ( O ) across the cell junctions of Nfix -/- (red) and Nfix +/+ (blue) ependymal cells. The peak fluorescence intensities of aPKCzeta and ZO-1 are lowered in Nfix -/- mice compared to controls. ( D , P ) Maximum fluorescent intensities of aPKCzeta ( D ) and ZO-1 ( P ) across the cell junctions of Nfix -/- (red) and Nfix +/+ (blue) ependymal cells. Maximum fluorescent intensity of aPKCzeta is significantly reduced in Nfix -/- mice compared to controls. ( E , F , I , J ) Images of brain lateral ventricles from P5 Nfix +/+ ( E , I ) and Nfix -/- ( F , J ) mice marked with antibodies against ependymal marker vimentin (purple), adherens junction protein beta-Catenin ( E , F ) or P120-Catenin ( I , J ) (green) and nuclear marker DAPI (grey) displayed separately and merged for each genotype. ( G , K ) Average fluorescent intensities of beta-Catenin ( G ) and P120-Catenin ( K ) across the cell junctions of Nfix -/- (red) and Nfix +/+ (blue) ependymal cells. The peak fluores
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
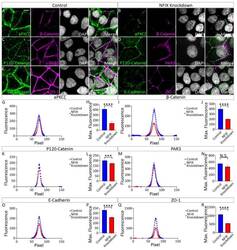
- Experimental details
- Figure 5 MCF7 cells treated with NFIX knockdown lentivirus demonstrate reduced localisation of adhesion proteins. ( A , B ) Images depicting MCF7 cells treated with scramble RNA lentivirus (scramble, A ) or NFIX shRNA lentivirus (NFIX KD, B ) and stained with antibodies against tight junction marker aPKCzeta (green), adherens junction marker beta-Catenin (purple) and nuclear marker DAPI (grey) displayed as separate panels or merged. ( C , D ) Images depicting NFIX KD ( C ) and scramble ( D ) cells stained with antibodies against adherens junction marker P120-Catenin (green), tight junction marker PAR3 (purple) and nuclear marker DAPI (grey) displayed as separate panels or merged ( E , F ) Images depicting NFIX KD ( E ) and scramble ( F ) cells stained with antibodies against adherens junction marker E-Cadherin (green), tight junction marker ZO-1 (purple) and nuclear marker DAPI (grey) displayed as separate panels or merged. ( G , I , K , M , O , Q ) Average fluorescent intensity across the cell junction was plotted for line scanning measurements of aPKCzeta ( G ), beta-Catenin ( I ), P120-Catenin ( K ), PAR3 ( M ), E-Cadherin ( O ) and ZO-1 ( Q ) for NFIX KD cells and controls. Tight junction proteins aPKCzeta and ZO-1 demonstrated reduced fluorescent intensity peaks in NFIX KD cells compared to controls. Tight junction protein PAR3 demonstrated no changes between treatment conditions. Adherens junction proteins beta-Catenin, P120-Catenin and E-Cadherin demonstrated reduced p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
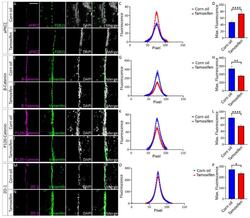
- Experimental details
- Tamoxifen treated Nfix iFOXJ1-GFP ependymal cells demonstrate reduced expression of adhesion proteins. ( A , B ) Lateral ventricle sections from Nfix iFOXJ1-GFP mice treated with corn oil ( A ) and tamoxifen ( B ) at 7 dpi labelled with aPKCzeta (purple), ependymal cell marker FOXJ1 (green) and nuclear marker DAPI (grey), displayed separately and merged. ( C ) Average aPKCzeta fluorescent intensity across the cell junction is displayed for tamoxifen treated Nfix iFOXJ1-GFP ependymal cells and controls. Average aPKCzeta fluorescent intensity across the cell junction was increased at the peak in tamoxifen treated Nfix iFOXJ1-GFP ependymal cells compared to controls. ( D ) Maximum aPKCzeta fluorescent intensity is displayed for tamoxifen treated Nfix iFOXJ1-GFP ependymal cells and controls. Maximum aPKCzeta fluorescent intensity is significantly increased in tamoxifen treated Nfix iFOXJ1-GFP ependymal cells compared to controls. ( E , F ) Lateral ventricle sections from Nfix iFOXJ1-GFP mice treated with corn oil ( E ) and tamoxifen ( F ) at 7 dpi labelled with beta-Catenin (purple), ependymal cell marker Vimentin (green) and nuclear marker DAPI (white), displayed separately and merged. ( G ) Average beta-Catenin fluorescent intensity across the cell junction is displayed for tamoxifen treated Nfix iFOXJ1-GFP ependymal cells and controls. Peak beta-Catenin fluorescent intensity is reduced in tamoxifen treated Nfix iFOXJ1-GFP mice compared to controls. ( H ) Maximum beta-Catenin f
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 Histological changes of hematoxylin-eosin staining assay in colonic and cecum tissue in HFD mice induced by P. vulgaris extract (500 mg/kg, PHAS 500) treatment. Micrographs are representative pictures with an original magnification of x20 (n = 4). (A) Colon tissue from the control group. (B) Colon tissue of HFD mice, showing severe goblet cell hyperplasia and mild epithelial hyperplasia with low-grade dysplasia. (C) Colon tissue of HFD + PHAS group, showing moderate goblet cell hyperplasia. (D) Cecum tissue from control mice. (E) Cecum tissue of HFD mice, showing severe goblet cell hyperplasia and mild epithelial hyperplasia with low-grade dysplasia. (F) Cecum tissue of HFD + PHAS group, showing moderate goblet cell hyperplasia. Protective effect of P. vulgaris extract (500 mg/kg, PHAS 500) treatment on colonic and cecum barrier integrity in HFD mice: in particular, immunoblot representatives of occludin expression in the colon (G) and cecum (I) and ZO-1 in the colon (H) and cecum (L) were showed. The levels are expressed as the density ratio of target to beta-actin. Data are expressed as means +- SEM ( n = 6). ** p < 0.01 vs. STD; # p < 0.05 and ## p < 0.01 vs. HFD.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
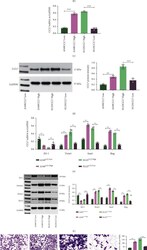
- Experimental details
- Figure 2 CCL7 knockdown suppresses EMT and mobility of LUAD cells. mRNA (a) and protein (b) levels of CCL7 in H1299, A549, H358 (low-invasive), and H125 (high-invasive) cells detected by qPCR and WB analyses; mRNA (c) and protein (d) levels of CCL7 in A549 and H125 cells after overexpression plasmid or shRNA transfection determined by qPCR and WB analyses; mRNA (e) and protein (f) levels of epithelial marker ZO-1 and mesenchymal markers Twist1, Snai1, and Slug in A549 and H125 cells examined by qPCR and WB analyses; (g) 24-h migration rate of A549 and H125 cells analyzed by the scratch test; (h) invasiveness of A549 and H125 cells analyzed by Transwell assay. Repetition = 3. ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
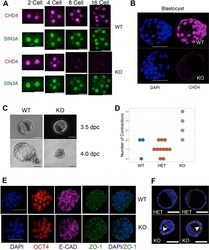
- Experimental details
- Fig. 1. CHD4 is required during the fourth day of development. (A) Representative composite spinning-disc images of anti-CHD4 (magenta) and SIN3A (green; used as a control) staining in wild-type and Chd4 -/- 2-, 4-, 8- and 16-cell embryos. Images are representative of eight 2-cell embryos, 12 4-cell embryos, 11 8-cell embryos (including both Chd4 -/- and Chd4 Delta / Delta embryos) and four 16-cell embryos. Scale bars: 33 um. (B) Composite confocal images of DAPI (blue) and CHD4 (magenta) staining in wild-type (WT) and Chd4 -/- (KO) 3.5 dpc embryos. Scale bars: 50 um. KO images are representative of >20 mutant blastocysts (including both Chd4 -/- and Chd4 Delta / Delta ). (C) Phase contrast images of wild-type (WT) and Chd4 -/- (KO) embryos flushed at different times on the fourth day post coitus (dpc). Scale bars: 50 um. KO images are representative of 16 Chd4 -/- embryos. (D) Average number of contractions per embryo genotype observed during live imaging (see supplementary material Movies 1-3 ). Each circle indicates the number of contractions for a single embryo of the indicated genotype. (E) Representative confocal 3D projection images of early 3.5 dpc embryos of the indicated genotypes stained for the indicated markers. KO images are representative of six embryos. Scale bars: 50 um. (F) Two representative images per genotype of late 3.5 dpc embryos stained for E-cadherin (white) and DAPI (blue). Each image is a composite of four to six stacks, wh
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
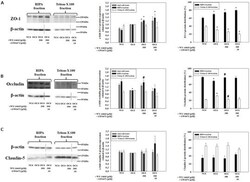
- Experimental details
- FIGURE 2 Influence of WY-14643 and PPARalpha antagonist GW6471 on effects of OC6 treatment on the protein expression of tight junction proteins ZO-1, occludin and claudin-5 of cerebEND cells. OC6 treatment of cerebENDs accords to 4 h OGD treatment with medium supernatants derived from 4 h OGD-treated C6 cells, whereas NC6 treatment means cerebEND cells incubated for 4 h under normoxic conditions with medium supernatants of 4 h normoxic treated C6 cells. Results for ZO-1 (A) , occludin (B) and claudin-5 (C) were displayed. Densitometric analyzed protein expression of total cell lysates were compared to RIPA and triton-X 100 fractions. Moreover, protein distributions [%] between RIPA and triton-X 100 fractions were presented. Values were related to beta-actin and according representative western blot images are depicted. Dotted lines indicated cuts of images of the same blot due to presentation reasons of selected bands. Statistical significance was labeled with * vs. NC6 and # vs. OC6 ( p < 0.05). Data are presented as means +- SEM ( n = 3-8).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
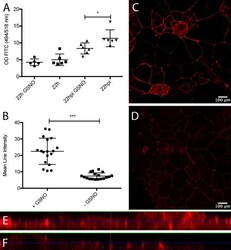
- Experimental details
- FIG 4 GSNO contributes to the maintenance of epithelial tightness of rotavirus-infected polarized Caco-2 cells and induces increased expression of ZO-1. (A) Polarized Caco-2 cells (>450 Omega/cm 2 ) were stimulated with GSNO for 24 h and infected with rotavirus. At 6 h p.i., the apical medium was replaced with medium containing 4-kDa FITC-dextran (2 mg/Transwell insert), and samples were obtained from the basolateral side at 22 h p.i. to measure passage of FITC-dextran. The controls were noninfected cells with and without GSNO and infected cells without GSNO. Basolateral samples were analyzed for FITC-dextran by spectrophotometry (494/518 nm), and values are presented as the optical density (OD) values and means with SD. * , P < 0.05, unpaired t test ( n = 5 or 6). (B) GSNO increases ZO-1 tight junction protein in Caco-2 cell monolayers. Caco-2 cell monolayers cultured on Lab-Tek II chamber slides to near confluence with and without 24-h exposure to GSNO were stained for ZO-1 expression (red; Alexa Fluor 594). Confocal images were captured and the mean intensities on single-cell circumference were measured using ImageJ. (C and D) Monolayers of GSNO-treated (C) and untreated (D) Caco-2 cells. Data are mean fluorescence intensities and means with SD. ***, P < 0.001, Mann-Whitney test ( n = 17). (E and F) Deconvolved images visualized in ortho-mode with XZ and YZ intensities of GSNO-treated (E) and untreated (F) Caco-2 cells. The XZ and YZ selections in each image are positioned
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Non-immunohistochemical staining with tissue clearing on formalin-fixed brain tissues. a Colour depth-coded image with maximum intensity projection of a piece of 1-mm-thick medulla block stained with cresyl violet and cleared with OPTIClear (z-stack depth = 228.83 um). Lower image showing a zoomed and tilted view of area outlined by the white box. b A piece of frontal cortical block immunostained for ZO-1 (green) and counterstained with DyLight 649-labelled Lycopersicon esculentum lectin (red). c DiI crystals were inserted into a cerebellar folium to trace the fibres up to the granular layer, followed by tissue clearing with OPTIClear for 6 h at 37 degC. Most of the fibres traced were mossy fibres. The inset shows the gross appearance of the sample with red crystals of DiI inserted within the OPTICleared sample. d Enlarged view of the white boxed region in a. with colour depth-coding (Z-depth 151.09 mum). e Z -stack images of dendritic spine-like projections visualised in areas outlined by white boxes. Scale bars, 1 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Effects of SA on small-intestinal injury in mice with MCD diet-induced steatohepatitis. ( A ) Histology was carried out using H&E staining (magnification, 100x and 200x), and representative images are shown. The height of five intact and well-oriented villi per sample was measured using ImageJ and the average value used as data. Results are the mean +- SEM for 4-6 mice per group. + p < 0.01. ( B ) Mucus production in the small intestine was detected using PAS staining (magnification, 100x and 200x), and representative images are shown. ( C ) Small-intestine sections were stained with ZO-1 (green) and DAPI (blue). Representative immunofluorescence images are shown. Magnified images represent high-magnification images of the boxed area.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Effect of miR-143 on the permeability of endothelial cells in vitro. (a) Methamphetamine increased the permeability of the HBMECs. (b) Effect of methamphetamine on the expression of claudin-5, occludin, and ZO-1 in HBMECs evaluated using western blot analysis. (c,d) Effect of miR-143 on the expression of tight junction proteins (c) and the permeability of HBMECs (d) . (e,f) Effect of anti-miR-143 on the expression of tight junction proteins (e) and the permeability of HBMECs (f) . All data are presented as the mean +- SD of three individual experiments. **p < 0.01 vs. the control group using Student's t-test. (g) Anti-miR-143 attenuated the methamphetamine-induced increase in the permeability of HBMECs. *p < 0.05 and **p < 0.01 vs. the anti-miR-con/control; ### p < 0.001 vs. the anti-miR-con/meth group using one-way ANOVA followed by the Holm-Sidak test. Meth, methamphetamine.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot