Antibody data
- Antibody Data
- Antigen structure
- References [52]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [28]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 47-0149-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD14 Monoclonal Antibody (61D3), APC-eFluor™ 780, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 61D3 monoclonal antibody reacts with human CD14, a 53-55 kDa GPI-linked glycoprotein. CD14 is expressed on monocytes, interfollicular macrophages and some dendritic cells. Complexes of LPS and LBP (LPS-Binding Protein) bind with high affinity to monocytes through the surface CD14.
- Antibody clone number
- 61D3
- Concentration
- 5 µL/Test
Submitted references Plasma and memory antibody responses to Gamma SARS-CoV-2 provide limited cross-protection to other variants.
Humoral immunity to SARS-CoV-2 elicited by combination COVID-19 vaccination regimens.
Boosting corrects a memory B cell defect in SARS-CoV-2 mRNA-vaccinated patients with inflammatory bowel disease.
Inactivated whole-virion vaccine BBV152/Covaxin elicits robust cellular immune memory to SARS-CoV-2 and variants of concern.
Antibody evolution to SARS-CoV-2 after single-dose Ad26.COV2.S vaccine in humans.
Broad and potent neutralizing human antibodies to tick-borne flaviviruses protect mice from disease.
Evolution of antibody immunity to SARS-CoV-2.
Naturally enhanced neutralizing breadth to SARS-CoV-2 after one year.
Naturally enhanced neutralizing breadth against SARS-CoV-2 one year after infection.
Changes in expression of PD-L1 on peripheral T cells in patients with melanoma and lung cancer treated with PD-1 inhibitors.
Evolution of Antibody Immunity to SARS-CoV-2.
mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants.
mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants.
A broadly neutralizing macaque monoclonal antibody against the HIV-1 V3-Glycan patch.
Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity.
Convergent Antibody Responses to SARS-CoV-2 Infection in Convalescent Individuals.
Isolation of single HIV-1 Envelope specific B cells and antibody cloning from immunized rhesus macaques.
Convergent antibody responses to SARS-CoV-2 in convalescent individuals.
Vascular occlusion by neutrophil extracellular traps in COVID-19.
Single-Cell Analyses Inform Mechanisms of Myeloid-Targeted Therapies in Colon Cancer.
Intrahepatic macrophage populations in the pathophysiology of primary sclerosing cholangitis.
Immunization expands B cells specific to HIV-1 V3 glycan in mice and macaques.
Characterizing the Role of Monocytes in T Cell Cancer Immunotherapy Using a 3D Microfluidic Model.
Modulation of B Cells and Homing Marker on NK Cells Through Extracorporeal Photopheresis in Patients With Steroid-Refractory/Resistant Graft-Vs.-Host Disease Without Hampering Anti-viral/Anti-leukemic Effects.
Butyrate upregulates the TLR4 expression and the phosphorylation of MAPKs and NK-κB in colon cancer cell in vitro.
Cellular metabolism constrains innate immune responses in early human ontogeny.
AURKA Suppresses Leukemic THP-1 Cell Differentiation through Inhibition of the KDM6B Pathway.
Comprehensive characterization of chorionic villi-derived mesenchymal stromal cells from human placenta.
The pro-inflammatory phenotype of the human non-classical monocyte subset is attributed to senescence.
Similarities and differences between helminth parasites and cancer cell lines in shaping human monocytes: Insights into parallel mechanisms of immune evasion.
Human umbilical cord mesenchymal stem cells improve the reserve function of perimenopausal ovary via a paracrine mechanism.
Enhancement of cytokine-driven NK cell IFN-γ production after vaccination of HCMV infected Africans.
A therapeutic T cell receptor mimic antibody targets tumor-associated PRAME peptide/HLA-I antigens.
Surface LAMP-2 Is an Endocytic Receptor That Diverts Antigen Internalized by Human Dendritic Cells into Highly Immunogenic Exosomes.
Platelet-Derived Ectosomes Reduce NK Cell Function.
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Thrombomodulin regulates monocye differentiation via PKCδ and ERK1/2 pathway in vitro and in atherosclerotic artery.
Investigating the causes for decreased levels of glutathione in individuals with type II diabetes.
Liposomal Glutathione Supplementation Restores TH1 Cytokine Response to Mycobacterium tuberculosis Infection in HIV-Infected Individuals.
A novel antibody-drug conjugate targeting SAIL for the treatment of hematologic malignancies.
Biologically active polymers from spontaneous carotenoid oxidation: a new frontier in carotenoid activity.
The strength of the antibody response to the nematode Ascaris lumbricoides inversely correlates with levels of B-Cell Activating Factor (BAFF).
KIR/HLA interactions negatively affect rituximab- but not GA101 (obinutuzumab)-induced antibody-dependent cellular cytotoxicity.
Human mesenchymal stem cells possess different biological characteristics but do not change their therapeutic potential when cultured in serum free medium.
Human female genital tract infection by the obligate intracellular bacterium Chlamydia trachomatis elicits robust Type 2 immunity.
Noncanonical dendritic cell differentiation and survival driven by a bacteremic pathogen.
Negative regulation of JAK2 by H3K9 methyltransferase G9a in leukemia.
Induction of autophagy is essential for monocyte-macrophage differentiation.
Plasmacytoid dendritic cells and C1q differentially regulate inflammatory gene induction by lupus immune complexes.
Upregulation of programmed death-1 on T cells and programmed death ligand-1 on monocytes in septic shock patients.
Ro60-associated single-stranded RNA links inflammation with fetal cardiac fibrosis via ligation of TLRs: a novel pathway to autoimmune-associated heart block.
B cells and monocytes from patients with active multiple sclerosis exhibit increased surface expression of both HERV-H Env and HERV-W Env, accompanied by increased seroreactivity.
Agudelo M, Muecksch F, Schaefer-Babajew D, Cho A, DaSilva J, Bednarski E, Ramos V, Oliveira TY, Cipolla M, Gazumyan A, Zong S, Rodrigues DAS, Lira GS, Conde L, Aguiar RS, Ferreira OC, Tanuri A, Affonso KC, Galliez RM, Castineiras TMPP, Echevarria-Lima J, Bozza MT, Vale AM, Bieniasz PD, Hatziioannou T, Nussenzweig MC
The Journal of experimental medicine 2022 Sep 5;219(9)
The Journal of experimental medicine 2022 Sep 5;219(9)
Humoral immunity to SARS-CoV-2 elicited by combination COVID-19 vaccination regimens.
Wang Z, Muecksch F, Muenn F, Cho A, Zong S, Raspe R, Ramos V, Johnson B, Ben Tanfous T, DaSilva J, Bednarski E, Guzman-Cardozo C, Turroja M, Millard KG, Tober-Lau P, Hillus D, Yao KH, Shimeliovich I, Dizon J, Kaczynska A, Jankovic M, Gazumyan A, Oliveira TY, Caskey M, Bieniasz PD, Hatziioannou T, Kurth F, Sander LE, Nussenzweig MC, Gaebler C
The Journal of experimental medicine 2022 Oct 3;219(10)
The Journal of experimental medicine 2022 Oct 3;219(10)
Boosting corrects a memory B cell defect in SARS-CoV-2 mRNA-vaccinated patients with inflammatory bowel disease.
Pape KA, Dileepan T, Matchett WE, Ellwood C, Stresemann S, Kabage AJ, Kozysa D, Evert C, Matson M, Lopez S, Krueger PD, Graiziger CT, Vaughn BP, Shmidt E, Rhein J, Schacker TW, Bold TD, Langlois RA, Khoruts A, Jenkins MK
JCI insight 2022 Jun 22;7(12)
JCI insight 2022 Jun 22;7(12)
Inactivated whole-virion vaccine BBV152/Covaxin elicits robust cellular immune memory to SARS-CoV-2 and variants of concern.
Vikkurthi R, Ansari A, Pai AR, Jha SN, Sachan S, Pandit S, Nikam B, Kalia A, Jit BP, Parray HA, Singh S, Kshetrapal P, Wadhwa N, Shrivastava T, Coshic P, Kumar S, Sharma P, Sharma N, Taneja J, Pandey AK, Sharma A, Thiruvengadam R, Grifoni A, Weiskopf D, Sette A, Bhatnagar S, Gupta N
Nature microbiology 2022 Jul;7(7):974-985
Nature microbiology 2022 Jul;7(7):974-985
Antibody evolution to SARS-CoV-2 after single-dose Ad26.COV2.S vaccine in humans.
Cho A, Muecksch F, Wang Z, Ben Tanfous T, DaSilva J, Raspe R, Johnson B, Bednarski E, Ramos V, Schaefer-Babajew D, Shimeliovich I, Dizon JP, Yao KH, Schmidt F, Millard KG, Turroja M, Jankovic M, Oliveira TY, Gazumyan A, Gaebler C, Caskey M, Hatziioannou T, Bieniasz PD, Nussenzweig MC
The Journal of experimental medicine 2022 Aug 1;219(8)
The Journal of experimental medicine 2022 Aug 1;219(8)
Broad and potent neutralizing human antibodies to tick-borne flaviviruses protect mice from disease.
Agudelo M, Palus M, Keeffe JR, Bianchini F, Svoboda P, Salát J, Peace A, Gazumyan A, Cipolla M, Kapoor T, Guidetti F, Yao KH, Elsterová J, Teislerová D, Chrdle A, Hönig V, Oliveira T, West AP, Lee YE, Rice CM, MacDonald MR, Bjorkman PJ, Růžek D, Robbiani DF, Nussenzweig MC
The Journal of experimental medicine 2021 May 3;218(5)
The Journal of experimental medicine 2021 May 3;218(5)
Evolution of antibody immunity to SARS-CoV-2.
Gaebler C, Wang Z, Lorenzi JCC, Muecksch F, Finkin S, Tokuyama M, Cho A, Jankovic M, Schaefer-Babajew D, Oliveira TY, Cipolla M, Viant C, Barnes CO, Bram Y, Breton G, Hägglöf T, Mendoza P, Hurley A, Turroja M, Gordon K, Millard KG, Ramos V, Schmidt F, Weisblum Y, Jha D, Tankelevich M, Martinez-Delgado G, Yee J, Patel R, Dizon J, Unson-O'Brien C, Shimeliovich I, Robbiani DF, Zhao Z, Gazumyan A, Schwartz RE, Hatziioannou T, Bjorkman PJ, Mehandru S, Bieniasz PD, Caskey M, Nussenzweig MC
Nature 2021 Mar;591(7851):639-644
Nature 2021 Mar;591(7851):639-644
Naturally enhanced neutralizing breadth to SARS-CoV-2 after one year.
Wang Z, Muecksch F, Schaefer-Babajew D, Finkin S, Viant C, Gaebler C, Hoffmann HH, Barnes CO, Cipolla M, Ramos V, Oliveira TY, Cho A, Schmidt F, da Silva J, Bednarski E, Aguado L, Yee J, Daga M, Turroja M, Millard KG, Jankovic M, Gazumyan A, Zhao Z, Rice CM, Bieniasz PD, Caskey M, Hatziioannou T, Nussenzweig MC
bioRxiv : the preprint server for biology 2021 Jun 2;
bioRxiv : the preprint server for biology 2021 Jun 2;
Naturally enhanced neutralizing breadth against SARS-CoV-2 one year after infection.
Wang Z, Muecksch F, Schaefer-Babajew D, Finkin S, Viant C, Gaebler C, Hoffmann HH, Barnes CO, Cipolla M, Ramos V, Oliveira TY, Cho A, Schmidt F, Da Silva J, Bednarski E, Aguado L, Yee J, Daga M, Turroja M, Millard KG, Jankovic M, Gazumyan A, Zhao Z, Rice CM, Bieniasz PD, Caskey M, Hatziioannou T, Nussenzweig MC
Nature 2021 Jul;595(7867):426-431
Nature 2021 Jul;595(7867):426-431
Changes in expression of PD-L1 on peripheral T cells in patients with melanoma and lung cancer treated with PD-1 inhibitors.
Dart SJ, Cook AM, Millward MJ, McDonnell AM, Chin WL, Hakeem MU, Meniawy TM, Bowyer SE
Scientific reports 2021 Jul 28;11(1):15312
Scientific reports 2021 Jul 28;11(1):15312
Evolution of Antibody Immunity to SARS-CoV-2.
Gaebler C, Wang Z, Lorenzi JCC, Muecksch F, Finkin S, Tokuyama M, Cho A, Jankovic M, Schaefer-Babajew D, Oliveira TY, Cipolla M, Viant C, Barnes CO, Hurley A, Turroja M, Gordon K, Millard KG, Ramos V, Schmidt F, Weisblum Y, Jha D, Tankelevich M, Yee J, Shimeliovich I, Robbiani DF, Zhao Z, Gazumyan A, Hatziioannou T, Bjorkman PJ, Mehandru S, Bieniasz PD, Caskey M, Nussenzweig MC, Hagglof T, Schwartz RE, Bram Y, Martinez-Delgado G, Mendoza P, Breton G, Dizon J, Unson-O'Brien C, Patel R
bioRxiv : the preprint server for biology 2021 Jan 4;
bioRxiv : the preprint server for biology 2021 Jan 4;
mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants.
Wang Z, Schmidt F, Weisblum Y, Muecksch F, Barnes CO, Finkin S, Schaefer-Babajew D, Cipolla M, Gaebler C, Lieberman JA, Oliveira TY, Yang Z, Abernathy ME, Huey-Tubman KE, Hurley A, Turroja M, West KA, Gordon K, Millard KG, Ramos V, Da Silva J, Xu J, Colbert RA, Patel R, Dizon J, Unson-O'Brien C, Shimeliovich I, Gazumyan A, Caskey M, Bjorkman PJ, Casellas R, Hatziioannou T, Bieniasz PD, Nussenzweig MC
bioRxiv : the preprint server for biology 2021 Jan 30;
bioRxiv : the preprint server for biology 2021 Jan 30;
mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants.
Wang Z, Schmidt F, Weisblum Y, Muecksch F, Barnes CO, Finkin S, Schaefer-Babajew D, Cipolla M, Gaebler C, Lieberman JA, Oliveira TY, Yang Z, Abernathy ME, Huey-Tubman KE, Hurley A, Turroja M, West KA, Gordon K, Millard KG, Ramos V, Da Silva J, Xu J, Colbert RA, Patel R, Dizon J, Unson-O'Brien C, Shimeliovich I, Gazumyan A, Caskey M, Bjorkman PJ, Casellas R, Hatziioannou T, Bieniasz PD, Nussenzweig MC
Nature 2021 Apr;592(7855):616-622
Nature 2021 Apr;592(7855):616-622
A broadly neutralizing macaque monoclonal antibody against the HIV-1 V3-Glycan patch.
Wang Z, Barnes CO, Gautam R, Cetrulo Lorenzi JC, Mayer CT, Oliveira TY, Ramos V, Cipolla M, Gordon KM, Gristick HB, West AP, Nishimura Y, Raina H, Seaman MS, Gazumyan A, Martin M, Bjorkman PJ, Nussenzweig MC, Escolano A
eLife 2020 Oct 21;9
eLife 2020 Oct 21;9
Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity.
Rydyznski Moderbacher C, Ramirez SI, Dan JM, Grifoni A, Hastie KM, Weiskopf D, Belanger S, Abbott RK, Kim C, Choi J, Kato Y, Crotty EG, Kim C, Rawlings SA, Mateus J, Tse LPV, Frazier A, Baric R, Peters B, Greenbaum J, Ollmann Saphire E, Smith DM, Sette A, Crotty S
Cell 2020 Nov 12;183(4):996-1012.e19
Cell 2020 Nov 12;183(4):996-1012.e19
Convergent Antibody Responses to SARS-CoV-2 Infection in Convalescent Individuals.
Robbiani DF, Gaebler C, Muecksch F, Lorenzi JCC, Wang Z, Cho A, Agudelo M, Barnes CO, Gazumyan A, Finkin S, Hagglof T, Oliveira TY, Viant C, Hurley A, Hoffmann HH, Millard KG, Kost RG, Cipolla M, Gordon K, Bianchini F, Chen ST, Ramos V, Patel R, Dizon J, Shimeliovich I, Mendoza P, Hartweger H, Nogueira L, Pack M, Horowitz J, Schmidt F, Weisblum Y, Michailidis E, Ashbrook AW, Waltari E, Pak JE, Huey-Tubman KE, Koranda N, Hoffman PR, West AP Jr, Rice CM, Hatziioannou T, Bjorkman PJ, Bieniasz PD, Caskey M, Nussenzweig MC
bioRxiv : the preprint server for biology 2020 May 22;
bioRxiv : the preprint server for biology 2020 May 22;
Isolation of single HIV-1 Envelope specific B cells and antibody cloning from immunized rhesus macaques.
Wang Z, Merkenschlager J, Chen ST, Oliveira TY, Ramos V, Gordon KM, Yao KH, Jankovic M, Nussenzweig M, Escolano A
Journal of immunological methods 2020 Mar;478:112734
Journal of immunological methods 2020 Mar;478:112734
Convergent antibody responses to SARS-CoV-2 in convalescent individuals.
Robbiani DF, Gaebler C, Muecksch F, Lorenzi JCC, Wang Z, Cho A, Agudelo M, Barnes CO, Gazumyan A, Finkin S, Hägglöf T, Oliveira TY, Viant C, Hurley A, Hoffmann HH, Millard KG, Kost RG, Cipolla M, Gordon K, Bianchini F, Chen ST, Ramos V, Patel R, Dizon J, Shimeliovich I, Mendoza P, Hartweger H, Nogueira L, Pack M, Horowitz J, Schmidt F, Weisblum Y, Michailidis E, Ashbrook AW, Waltari E, Pak JE, Huey-Tubman KE, Koranda N, Hoffman PR, West AP Jr, Rice CM, Hatziioannou T, Bjorkman PJ, Bieniasz PD, Caskey M, Nussenzweig MC
Nature 2020 Aug;584(7821):437-442
Nature 2020 Aug;584(7821):437-442
Vascular occlusion by neutrophil extracellular traps in COVID-19.
Leppkes M, Knopf J, Naschberger E, Lindemann A, Singh J, Herrmann I, Stürzl M, Staats L, Mahajan A, Schauer C, Kremer AN, Völkl S, Amann K, Evert K, Falkeis C, Wehrfritz A, Rieker RJ, Hartmann A, Kremer AE, Neurath MF, Muñoz LE, Schett G, Herrmann M
EBioMedicine 2020 Aug;58:102925
EBioMedicine 2020 Aug;58:102925
Single-Cell Analyses Inform Mechanisms of Myeloid-Targeted Therapies in Colon Cancer.
Zhang L, Li Z, Skrzypczynska KM, Fang Q, Zhang W, O'Brien SA, He Y, Wang L, Zhang Q, Kim A, Gao R, Orf J, Wang T, Sawant D, Kang J, Bhatt D, Lu D, Li CM, Rapaport AS, Perez K, Ye Y, Wang S, Hu X, Ren X, Ouyang W, Shen Z, Egen JG, Zhang Z, Yu X
Cell 2020 Apr 16;181(2):442-459.e29
Cell 2020 Apr 16;181(2):442-459.e29
Intrahepatic macrophage populations in the pathophysiology of primary sclerosing cholangitis.
Chen YY, Arndtz K, Webb G, Corrigan M, Akiror S, Liaskou E, Woodward P, Adams DH, Weston CJ, Hirschfield GM
JHEP reports : innovation in hepatology 2019 Nov;1(5):369-376
JHEP reports : innovation in hepatology 2019 Nov;1(5):369-376
Immunization expands B cells specific to HIV-1 V3 glycan in mice and macaques.
Escolano A, Gristick HB, Abernathy ME, Merkenschlager J, Gautam R, Oliveira TY, Pai J, West AP Jr, Barnes CO, Cohen AA, Wang H, Golijanin J, Yost D, Keeffe JR, Wang Z, Zhao P, Yao KH, Bauer J, Nogueira L, Gao H, Voll AV, Montefiori DC, Seaman MS, Gazumyan A, Silva M, McGuire AT, Stamatatos L, Irvine DJ, Wells L, Martin MA, Bjorkman PJ, Nussenzweig MC
Nature 2019 Jun;570(7762):468-473
Nature 2019 Jun;570(7762):468-473
Characterizing the Role of Monocytes in T Cell Cancer Immunotherapy Using a 3D Microfluidic Model.
Lee SWL, Adriani G, Ceccarello E, Pavesi A, Tan AT, Bertoletti A, Kamm RD, Wong SC
Frontiers in immunology 2018;9:416
Frontiers in immunology 2018;9:416
Modulation of B Cells and Homing Marker on NK Cells Through Extracorporeal Photopheresis in Patients With Steroid-Refractory/Resistant Graft-Vs.-Host Disease Without Hampering Anti-viral/Anti-leukemic Effects.
Wang L, Ni M, Hückelhoven-Krauss A, Sellner L, Hoffmann JM, Neuber B, Luft T, Hegenbart U, Schönland S, Kleist C, Sill M, Chen BA, Wuchter P, Eckstein V, Krüger W, Hilgendorf I, Yerushalmi R, Nagler A, Müller-Tidow C, Ho AD, Dreger P, Schmitt M, Schmitt A
Frontiers in immunology 2018;9:2207
Frontiers in immunology 2018;9:2207
Butyrate upregulates the TLR4 expression and the phosphorylation of MAPKs and NK-κB in colon cancer cell in vitro.
Xiao T, Wu S, Yan C, Zhao C, Jin H, Yan N, Xu J, Wu Y, Li C, Shao Q, Xia S
Oncology letters 2018 Oct;16(4):4439-4447
Oncology letters 2018 Oct;16(4):4439-4447
Cellular metabolism constrains innate immune responses in early human ontogeny.
Kan B, Michalski C, Fu H, Au HHT, Lee K, Marchant EA, Cheng MF, Anderson-Baucum E, Aharoni-Simon M, Tilley P, Mirmira RG, Ross CJ, Luciani DS, Jan E, Lavoie PM
Nature communications 2018 Nov 16;9(1):4822
Nature communications 2018 Nov 16;9(1):4822
AURKA Suppresses Leukemic THP-1 Cell Differentiation through Inhibition of the KDM6B Pathway.
Park JW, Cho H, Oh H, Kim JY, Seo SB
Molecules and cells 2018 May 31;41(5):444-453
Molecules and cells 2018 May 31;41(5):444-453
Comprehensive characterization of chorionic villi-derived mesenchymal stromal cells from human placenta.
Ventura Ferreira MS, Bienert M, Müller K, Rath B, Goecke T, Opländer C, Braunschweig T, Mela P, Brümmendorf TH, Beier F, Neuss S
Stem cell research & therapy 2018 Feb 5;9(1):28
Stem cell research & therapy 2018 Feb 5;9(1):28
The pro-inflammatory phenotype of the human non-classical monocyte subset is attributed to senescence.
Ong SM, Hadadi E, Dang TM, Yeap WH, Tan CT, Ng TP, Larbi A, Wong SC
Cell death & disease 2018 Feb 15;9(3):266
Cell death & disease 2018 Feb 15;9(3):266
Similarities and differences between helminth parasites and cancer cell lines in shaping human monocytes: Insights into parallel mechanisms of immune evasion.
Narasimhan PB, Akabas L, Tariq S, Huda N, Bennuru S, Sabzevari H, Hofmeister R, Nutman TB, Tolouei Semnani R
PLoS neglected tropical diseases 2018 Apr;12(4):e0006404
PLoS neglected tropical diseases 2018 Apr;12(4):e0006404
Human umbilical cord mesenchymal stem cells improve the reserve function of perimenopausal ovary via a paracrine mechanism.
Li J, Mao Q, He J, She H, Zhang Z, Yin C
Stem cell research & therapy 2017 Mar 9;8(1):55
Stem cell research & therapy 2017 Mar 9;8(1):55
Enhancement of cytokine-driven NK cell IFN-γ production after vaccination of HCMV infected Africans.
Darboe A, Danso E, Clarke E, Umesi A, Touray E, Wegmuller R, Moore SE, Riley EM, Goodier MR
European journal of immunology 2017 Jun;47(6):1040-1050
European journal of immunology 2017 Jun;47(6):1040-1050
A therapeutic T cell receptor mimic antibody targets tumor-associated PRAME peptide/HLA-I antigens.
Chang AY, Dao T, Gejman RS, Jarvis CA, Scott A, Dubrovsky L, Mathias MD, Korontsvit T, Zakhaleva V, Curcio M, Hendrickson RC, Liu C, Scheinberg DA
The Journal of clinical investigation 2017 Jun 30;127(7):2705-2718
The Journal of clinical investigation 2017 Jun 30;127(7):2705-2718
Surface LAMP-2 Is an Endocytic Receptor That Diverts Antigen Internalized by Human Dendritic Cells into Highly Immunogenic Exosomes.
Leone DA, Peschel A, Brown M, Schachner H, Ball MJ, Gyuraszova M, Salzer-Muhar U, Fukuda M, Vizzardelli C, Bohle B, Rees AJ, Kain R
Journal of immunology (Baltimore, Md. : 1950) 2017 Jul 15;199(2):531-546
Journal of immunology (Baltimore, Md. : 1950) 2017 Jul 15;199(2):531-546
Platelet-Derived Ectosomes Reduce NK Cell Function.
Sadallah S, Schmied L, Eken C, Charoudeh HN, Amicarella F, Schifferli JA
Journal of immunology (Baltimore, Md. : 1950) 2016 Sep 1;197(5):1663-71
Journal of immunology (Baltimore, Md. : 1950) 2016 Sep 1;197(5):1663-71
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Sakthivel P, Grunewald J, Eklund A, Bruder D, Wahlström J
Clinical and experimental immunology 2016 Feb;183(2):294-306
Clinical and experimental immunology 2016 Feb;183(2):294-306
Thrombomodulin regulates monocye differentiation via PKCδ and ERK1/2 pathway in vitro and in atherosclerotic artery.
Tsai CS, Lin YW, Huang CY, Shih CM, Tsai YT, Tsao NW, Lin CS, Shih CC, Jeng H, Lin FY
Scientific reports 2016 Dec 2;6:38421
Scientific reports 2016 Dec 2;6:38421
Investigating the causes for decreased levels of glutathione in individuals with type II diabetes.
Lagman M, Ly J, Saing T, Kaur Singh M, Vera Tudela E, Morris D, Chi PT, Ochoa C, Sathananthan A, Venketaraman V
PloS one 2015;10(3):e0118436
PloS one 2015;10(3):e0118436
Liposomal Glutathione Supplementation Restores TH1 Cytokine Response to Mycobacterium tuberculosis Infection in HIV-Infected Individuals.
Ly J, Lagman M, Saing T, Singh MK, Tudela EV, Morris D, Anderson J, Daliva J, Ochoa C, Patel N, Pearce D, Venketaraman V
Journal of interferon & cytokine research : the official journal of the International Society for Interferon and Cytokine Research 2015 Nov;35(11):875-87
Journal of interferon & cytokine research : the official journal of the International Society for Interferon and Cytokine Research 2015 Nov;35(11):875-87
A novel antibody-drug conjugate targeting SAIL for the treatment of hematologic malignancies.
Kim SY, Theunissen JW, Balibalos J, Liao-Chan S, Babcock MC, Wong T, Cairns B, Gonzalez D, van der Horst EH, Perez M, Levashova Z, Chinn L, D'Alessio JA, Flory M, Bermudez A, Jackson DY, Ha E, Monteon J, Bruhns MF, Chen G, Migone TS
Blood cancer journal 2015 May 29;5(5):e316
Blood cancer journal 2015 May 29;5(5):e316
Biologically active polymers from spontaneous carotenoid oxidation: a new frontier in carotenoid activity.
Johnston JB, Nickerson JG, Daroszewski J, Mogg TJ, Burton GW
PloS one 2014;9(10):e111346
PloS one 2014;9(10):e111346
The strength of the antibody response to the nematode Ascaris lumbricoides inversely correlates with levels of B-Cell Activating Factor (BAFF).
Bornacelly A, Mercado D, Acevedo N, Caraballo L
BMC immunology 2014 Jun 7;15:22
BMC immunology 2014 Jun 7;15:22
KIR/HLA interactions negatively affect rituximab- but not GA101 (obinutuzumab)-induced antibody-dependent cellular cytotoxicity.
Terszowski G, Klein C, Stern M
Journal of immunology (Baltimore, Md. : 1950) 2014 Jun 15;192(12):5618-24
Journal of immunology (Baltimore, Md. : 1950) 2014 Jun 15;192(12):5618-24
Human mesenchymal stem cells possess different biological characteristics but do not change their therapeutic potential when cultured in serum free medium.
Wang Y, Wu H, Yang Z, Chi Y, Meng L, Mao A, Yan S, Hu S, Zhang J, Zhang Y, Yu W, Ma Y, Li T, Cheng Y, Wang Y, Wang S, Liu J, Han J, Li C, Liu L, Xu J, Han ZB, Han ZC
Stem cell research & therapy 2014 Dec 4;5(6):132
Stem cell research & therapy 2014 Dec 4;5(6):132
Human female genital tract infection by the obligate intracellular bacterium Chlamydia trachomatis elicits robust Type 2 immunity.
Vicetti Miguel RD, Harvey SA, LaFramboise WA, Reighard SD, Matthews DB, Cherpes TL
PloS one 2013;8(3):e58565
PloS one 2013;8(3):e58565
Noncanonical dendritic cell differentiation and survival driven by a bacteremic pathogen.
Miles B, Scisci E, Carrion J, Sabino GJ, Genco CA, Cutler CW
Journal of leukocyte biology 2013 Aug;94(2):281-9
Journal of leukocyte biology 2013 Aug;94(2):281-9
Negative regulation of JAK2 by H3K9 methyltransferase G9a in leukemia.
Son HJ, Kim JY, Hahn Y, Seo SB
Molecular and cellular biology 2012 Sep;32(18):3681-94
Molecular and cellular biology 2012 Sep;32(18):3681-94
Induction of autophagy is essential for monocyte-macrophage differentiation.
Zhang Y, Morgan MJ, Chen K, Choksi S, Liu ZG
Blood 2012 Mar 22;119(12):2895-905
Blood 2012 Mar 22;119(12):2895-905
Plasmacytoid dendritic cells and C1q differentially regulate inflammatory gene induction by lupus immune complexes.
Santer DM, Wiedeman AE, Teal TH, Ghosh P, Elkon KB
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 15;188(2):902-15
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 15;188(2):902-15
Upregulation of programmed death-1 on T cells and programmed death ligand-1 on monocytes in septic shock patients.
Zhang Y, Li J, Lou J, Zhou Y, Bo L, Zhu J, Zhu K, Wan X, Cai Z, Deng X
Critical care (London, England) 2011;15(1):R70
Critical care (London, England) 2011;15(1):R70
Ro60-associated single-stranded RNA links inflammation with fetal cardiac fibrosis via ligation of TLRs: a novel pathway to autoimmune-associated heart block.
Clancy RM, Alvarez D, Komissarova E, Barrat FJ, Swartz J, Buyon JP
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 15;184(4):2148-55
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 15;184(4):2148-55
B cells and monocytes from patients with active multiple sclerosis exhibit increased surface expression of both HERV-H Env and HERV-W Env, accompanied by increased seroreactivity.
Brudek T, Christensen T, Aagaard L, Petersen T, Hansen HJ, Møller-Larsen A
Retrovirology 2009 Nov 16;6:104
Retrovirology 2009 Nov 16;6:104
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
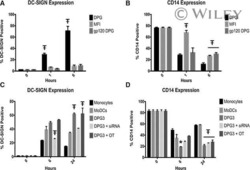
- Staining of normal human peripheral blood cells with Mouse IgG1 K Isotype Control APC-eFluor® 780 (Product # 47-4714-82) (blue histogram) or Anti-Human CD14 APC-eFluor® 780 (purple histogram). Cells in the monocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
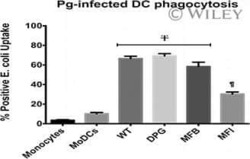
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
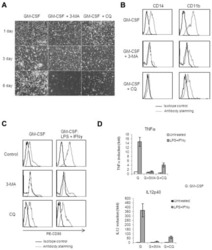
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
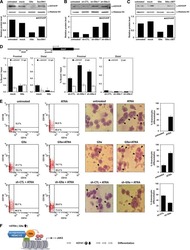
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
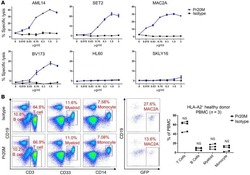
- Figure 2 Cell surface expression of SAIL in CLL, AML and MM patient samples and normal BMMC and PBMC controls. ( a ) Three CLL specimens analyzed by flow cytometry. CLL cells were identified as CD19/CD5 double-positive cells. The histograms present SAIL (filled) and isotype control (open) staining in the live-cell and the CLL population. ( b ) Flow cytometry analysis of three AML specimens. SAIL expression is assessed in live-cells, CD33-positive and CD34-positive cells. ( c ) Flow cytometry analysis of three MM specimens. CD38 high cells with CD56 expression were gated for MM cells. SAIL expression is assessed in the live-cell and the MM population. ( d and e ) Flow cytometry analysis of SAIL expression in BMMC ( d ) and PBMC ( e ) via co-staining with CD19, CD3, CD14, CD56, CD33, CD34 and a cocktail of lineage (LN) markers. Numbers in histograms are median-fluorescence-intensity fold-change values relative to the isotype control. Three and two representative examples are shown for the tumor and normal samples, respectively.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
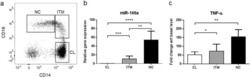
- Figure 2 PD-1 and PD-L1 were upregulated on T cells and monocytes in septic shock patients . Blood samples were obtained from 19 septic shock patients and 22 healthy controls and were stained for programmed death-1 (PD-1) and programmed death ligand-1 (PD-L1) gated on CD4 + T cells, CD8 + T cells, and CD14 + monocytes. (a) to (c) Percentage of PD-1 expression on (a) CD4 + T cells and (b) CD8 + T cells, and (c) percentage of PD-L1 expression on CD14 + monocytes. Each dot represents one individual. Data are mean +- standard error of the mean (SEM) of three independent experiments. ** P < 0.01 compared with healthy controls. (d) to (f) Mean fluorescence intensity (relative fluorescence units) of PD-1 expression on (d) CD4 + T cells, (e) PD-1 expression on CD8 + T cells, and (f) PD-L1 expression on CD14 + monocytes Each dot represents one individual. Data are mean +- SEM of three independent experiments. * P < 0.05 compared with healthy controls. (g) Representative PD-1 expression levels on CD4 + T cells and CD8 + T cells, and PD-L1 expression on CD14 + monocytes. Values in the upper-right quadrant indicate the percentage of cells that express PD-1 or PD-L1.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
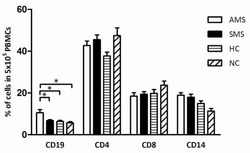
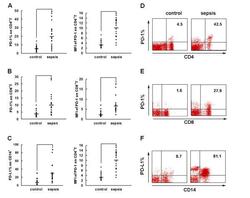
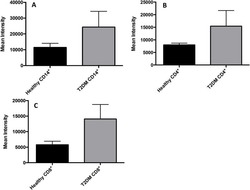
- Fig 7 Measurement of ROS in CD14 + cells, CD4 + T-cells, and CD8 + T-cells by cellROX stain mean intensity in T2DM patients compared to healthy. CD14 + cells were stained with cellROX green reagent, a marker of ROS, and a CD14 cell marker, CD14-PE. CD14 + -ROX + cells' mean intensity was analyzed by FLOW cytometry. There was an observable increase in ROX mean intensity in CD14 + cells isolated from individuals with T2DM compared to healthy volunteers (Fig. 7A). CD4 + cells were also stained with cellROX green reagent and a CD4 cell marker, CD4-Cy5. CD4 + -ROX + cells' mean intensity was analyzed by FLOW cytometry. There was an observable increase in ROX mean intensity in CD4 + T-cells isolated from individuals with T2DM compared to healthy volunteers (Fig. 7B). CD8 + cells were stained with cellROX green reagent and a CD8 cell marker, CD8-Cy5. CD8 + -ROX + cells' mean intensity was analyzed by FLOW cytometry. There was an observable increase in ROX mean intensity in CD8 + T-cells isolated from individuals with T2DM compared to healthy volunteers (Fig. 7C). Data represents mean +-SE from 5 healthy individuals and 5 individuals with T2DM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
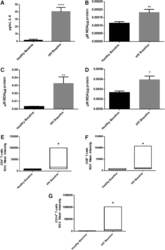
- FIG. 2. Baseline comparison of the interleukin-6 (IL-6) and reactive oxygen species (ROS) markers between healthy volunteers and HIV-positive individuals. We observed a significant increase in the levels of the proinflammatory cytokine, IL-6 in plasma samples collected from individuals with HIV infection compared to healthy individuals (A) . Data represent mean+-SE from comparing baseline levels of 10 healthy volunteers and 15 HIV-positive individuals, **** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
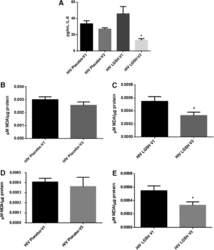
- FIG. 6. Difference in plasma IL-6 levels and ROS markers pre- and post-GSH supplementation. Sandwich ELISA was performed to compare the cytokine levels between pre-supplementation (V1) and post-supplementation (V3). Assay of cytokines showed a significant decrease in the levels of IL-6 in plasma samples collected from the lGSH-treatment group. There was no significant difference between the levels of IL-6 from the placebo group when comparing visit 1 and visit 3 (A) . Data represent mean+-SE, * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
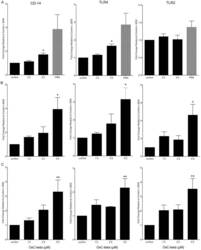
- Figure 1 Effect of OxC-beta on CD14, TLR-4, and TLR-2 levels in vitro . Human THP-1 monocytes (A), fibroblasts (B), and endothelial cells (C), were treated with the indicated concentrations of OxC-beta or vehicle control (DMSO) for 24 hours. Immune receptor content was measured 24 hours post-treatment by FACS analysis. OxC-beta-induced increase in receptor level was assessed relative to untreated control cells using a one-way analysis of variance with Tukey's post test for multiple comparisons. DMSO had no effect on receptor level (result not shown). Phorbol myristate acetate (PMA) was used as a positive control in experiments with THP-1 cells (hatched bars).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
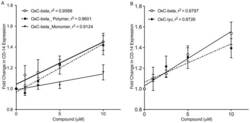
- Figure 5 Determination of activities relative to OxC-beta of (A) OxC-beta polymer and monomer fractions, and (B) oxidized lycopene (OxC-lyc), using a CD14 receptor expression assay. THP-1 cells were treated for 24 hours with the indicated concentrations of compounds. CD14 expression was quantified using FACS analysis. The effect of each compound is shown relative to untreated cells. Points represent the mean and standard error from three separate experiments. (A) Correlation analysis indicates a significant dose effect for each compound on CD14 expression with p-values of 0.0036 for OxC-beta, 0.0034 for the polymer, and 0.0113 for the monomer. Comparison of the relative activity of each compound indicates that the monomer is significantly less active than the polymer (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
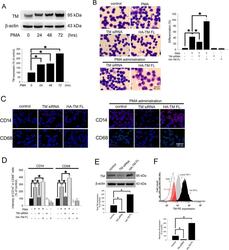
- Figure 1 PMA-induced TM expression mediates morphological changes and differentiation marker expression in THP-1 cells. ( A ) THP-1 cells were treated with 150 nM PMA for 24-72 hours. The total cell lysates were harvested, and the expression of TM was analyzed using western blot analysis. beta-actin was used as a loading control. Five independent experiments have been performed (n = 5) and representative images have been showed. The amount of proteins expression was quantified using densitometry and presented as bar graph. The data are presented as the mean +- SD (n = 5), and * p < 0.05 was considered significant. ( B ) THP-1 cells were transfected with TM siRNA or HA-TM FL plasmid for 24 h followed by PMA stimulation for 72 hours. The morphology of the cells was observed using light microscopy. The adherent differentiated macrophage-like cells are indicated by a white arrowhead. Five independent experiments have been performed (n = 5). The quantification is shown in the right graph. ( C ) The expression of the macrophage cell surface markers CD14 (red) and CD68 (green) was analyzed using immunofluorescence and microscopy. Hoechst staining was used to label the nuclei. The scale bar indicates 100 mum. Five independent experiments have been performed (n = 5), and showed representative images. ( D ) The expression of CD14 and CD68 was analyzed using flow cytometry. Data are expressed as a % of the control, are presented as the mean +- SD and represent the results of three indep
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
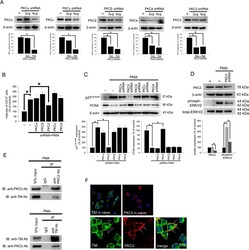
- Figure 4 TM regulates THP-1 cell differentiation via the PKCdelta-ERK1/2 signaling pathway. ( A ) THP-1 cells were transfected with 2 or 4 mug of PKCalpha, PKCbeta, PKCdelta, PKCepsilon, or PKCtheta shRNA for 24 h. Total cell lysates were purified, and knockdown efficiency was assayed using western blot analysis. ( B ) The THP-1 cells were knocked down by PKCalpha, PKCbeta, PKCdelta, PKCepsilon, and PKCtheta shRNAs for 24 h followed by PMA stimulation for 72 hours. The number of CD14 + cells was scored using flow cytometry. Data are expressed as a % of the control, are presented as the mean +- SD and represent the results of five independent experiments (n = 5, * p < 0.05 was considered significant). ( C ) Different sets of THP-1 cells were transfected with 4 mug of each shRNA for 24 h followed by PMA stimulation for 72 hours. The levels of p21 Cip1/WAF1 and PCNA were analyzed using western blot analysis. ( D ) The THP-1 cells were knocked down by PKCdelta shRNA for 24 h followed by PMA stimulation for 72 hours. The level of PKCdelta and total and phosphorylated ERK1/2 was analyzed using western blot analysis. In western blot analysis, beta-actin and total-ERK1/2 were used as loading controls. The density of each band was quantified using densitometry and related protein expression was presented as bar graph. The data are presented as the mean +- SD, and * p < 0.05 was considered significant (n = 5). ( E ) Lysates of THP-1 cells with PMA stimulation were extracted. Left, immu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
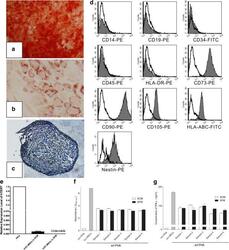
- Fig. 1 Flow cytometry analysis of phenotype characterization of hUCMSCs. Phenotype of CD73, CD90, CD105, CD14, CD34, CD45, CD79a and HLA-DR of hUCMSCs was detected by flow cytometry. Intensity >= 95% represented strong expression while
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2 Induced differentiation, flow cytometric and immunosuppressive ability analysis of human umbilical cord mesenchymal stem cells expanded in serum-free medium. After differentiation induction, (a) osteogenesis was confirmed by Alizarin Red (x40), (b) adipogenesis was stained by Oil Red O (x200) and (c) chondrogenesis was analyzed by Toluidine Blue (x100). (d) Serum-free medium (SFM)-expanded human umbilical cord mesenchymal stem cells (hUC-MSCs) at the 10th passage were labeled with antibodies against human antigens CD14-PE, CD19-PE, CD34-FITC, CD45-PE, CD73-PE, CD90-PE, CD105-PE, HLA-ABC-FITC, HLA-DR-PE and Nestin-PE. (e) Expression of hTERT in hUC-MSCs. Graph shows the level of hTERT transcripts of hUC-MSCs cultured in serum-containing medium (SCM) and SFM ( n = 5). Values presented as ratio of positive control (HeLa cells). Immunosuppressive ability of hUC-MSCs was evaluated by co-culturing with human peripheral blood mononuclear cells (hPBMCs). (f) Proliferation of hPBMCs was quantified based the measurement of BrdU incorporation during DNA synthesis. (g) Level of interferon gamma (IFN-gamma) in the supernatant was determined by ELISA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
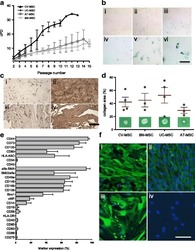
- Fig. 2 a Cumulative population-doubling (cPD) levels versus passage number for the four different sources of MSC. Black represents CV-MSC ( n = 7), dark gray UC-MSC ( n = 4), medium gray AT-MSC ( n = 5), and light gray BM-MSC ( n = 6). b IHC-based senescence-associated beta-galactosidase (SA-beta-gal) staining of CV-MSC in early (i, passage 4) and late (ii, passage 9) passages, AT-MSC in passage 6 (iii), BM-MSC in passage 6 (iv), and UC-MSC in passage 2 (v) and passage 4 (vi). Scale = 200 mum. c IHC of CV-MSC (i, ii) and BM-MSC (iii, iv) stained for osteopontin (i, iii) and fibronectin (ii, iv). Scale = 1 mm. d Collagen area (%) after collagen contraction assay for CV-MSC ( n = 4), BM-MSC ( n = 3), UC-MSC ( n = 4), and AT-MSC ( n = 3). Cells in passage 3 were used. Results expressed as mean +- SD, percentage of the total collagen area of the collagen gels without cells. e Surface marker expression of CV-MSC in early passages ( n = 5). Results expressed as mean +- SD (%). f Representative immunofluorescence of early passaged CV-MSC (i, iii) and BM-MSC (iii, iv) stained for SM22alpha (i, iii) and alpha-SMA (ii, iv). Scale = 50 mum. AT adipose tissue, BM bone marrow, CV chorionic villi, MSC mesenchymal stromal cells, UC umbilical cord
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
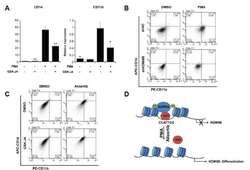
- Fig. 5 KDM6B promotes the differentiation of THP-1 cells (A) THP-1 cells were treated with 100 ng/ml PMA, 2 muM GSK-J4, or DMSO for 48 h. CD14 and CD11b expression levels were confirmed using qRT-PCR and normalized to GAPDH . Results are shown as mean +- SEM, n = 3; *p < 0.05, **p < 0.01. (B) We treated negative control (shNC)- and shKDM6B-transfected THP-1 cells with 100 ng/ml PMA for 48 h. The cells were stained with PE-CD11b and APC-CD14 antibodies. The percentage of cells in each quadrant is indicated in the figure. (C) We treated THP-1 cells with 2 muM GSK-J4 or 0.3 muM alisertib for 48 h. The cells were stained with PE-CD11b and APC-CD14 antibodies. The percentage of cells in each quadrant is indicated in the figure. (D) A model of AURKA regulating KDM6B expression in PMA-mediated THP-1 differentiation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
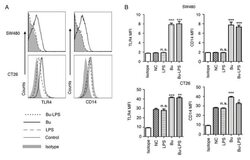
- Figure 4. Butyrate upregulates the levels of TLR4 and CD14 on colon cancer cells. The expression levels of TLR4 and CD14 on the membrane of SW480 cells and CT26 cells treated by butyrate and/or LPS were (A) analyzed using a flow cytometer, and (B) the MFI values of TLR4 and CD14 were quantitative analyzed. These experiments were repeated >=3 times, and representative graphs are presented. Compared with the NC group, *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
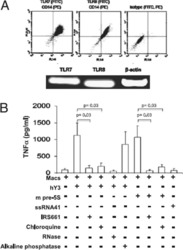
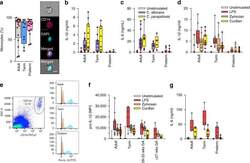
- Fig. 1 Responses to Candida spp . in neonatal immune cells. a Phagocytosis of Candida in monocytes (boxes and whiskers), including a representative flow microscopy diagram (white bar = ~10 um). Data pooled from multiple experiments over 14 months (9 to 17 subjects per age group; see Supplemental Data for clinical information on preterm subjects); b IL-1beta and c IL-6 response (blood mononuclear cells) to C. albicans or C. parapsilosis (24 h stimulation; 10 to 18 subjects per age group; boxes and whiskers); ( d ) IL-1beta (24 h; 11 to 21 subjects per age group; boxes and whiskers) and e representative gating for pro-IL-1beta (5 h LPS stimulation), gated on CD14-expressing cells (black = fluorescent-minus one control; orange = unstimulated; blue = LPS; representative preterm sample is from a 26 weeks' infant); f pro-IL-1beta (5 h) or g IL-6 (24 h) in response to LPS, zymosan or curdlan (mononuclear cells; 11 to 21 subjects per age group; boxes and whiskers); for b and c , data was pooled from multiple experiments assayed in four ELISA batches with similar distribution of samples per age group
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
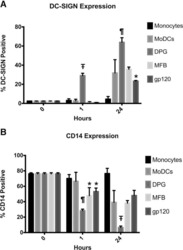
- Fig. 5 Gene expression and translation of dectin-1 signaling proteins. a Illustration of selected signaling molecules downstream of dectin-1; b Polysome profiles and c quantification of signalosome genes (qPCR) in monosome, disome, and light and heavy polysome fractions (monocytes). Data are from 4 subjects per age group (boxes and whiskers; RQ = relative quantification); d Quantification of signalosome genes (qPCR) in total RNA fractions (4 to 5 subjects/age group; mean +- SD); e Surface expression of dectin-1 (flow cytometry, mononuclear cells, gated on CD14-expressing cells; data pooled from 10 to 23 subjects per age group; boxes and whiskers); f Representative (cropped) Western blot of MALT1 and Bcl10 protein expression in monocytes after 0 to 60 min LPS stimulation. Representative blot is from a 29 weeks gestation sample. Images cropped from same blot probes with each antibody; cumulative quantification of 4 independent Western blot experiments for g MALT1 and h Bcl10 (mean +- SD)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
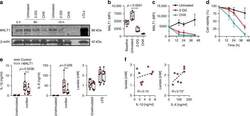
- Fig. 8 Inhibition glycolysis results in loss of MALT1 protein expression. Effect of blocking glycolysis (using 2-DG) or of blocking translation (using cycloheximide, as control) on MALT1 protein expression (monocytes). a MALT1 protein was detected by Western blot (left panel; representative of two experiments; cropped images from same blot probed with each antibody) at 8 h and 19 h. Lymphoblastoid cell line (LCL) lysate used as positive control for MALT1 protein expression; MALT1 protein detection ( b ) at 16 h and ( c ) over time (intracellular staining by flow cytometry, gated on CD14-expressing cells; MFI mean fluorescence intensity; dotted line: signal for fluorescence-minus-one staining control MFI level; boxes and whiskers with a paired 2-sided t -test in b ; mean +- SD in c and d ; d corresponding cell viability over time (mean +- SD); 6 subjects. e Effect of MALT1 inhibition on IL-1beta, IL-6, and lactate production at rest and following LPS (mononuclear cells; boxes and whiskers with 2-sided paired t -tests); f correlation between LPS-induced IL-1beta and IL-6, and lactate production (Spearman' r ; * p < 0.05; with dotted regression line); 8 subjects. All experiments were conducted in adult cells
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry