Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MHCD3817 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD38 Monoclonal Antibody (HIT2), PE-Texas Red
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- The R-phycoerythrin (PE)-Texas Red conjugate permits simultaneous multicolor labeling and detection of multiple targets with excitation by a single excitation source-the 488 nm line of the argon-ion laser.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- HIT2
- Vial size
- 500 µL
- Storage
- 4° C, store in dark
Submitted references Heterogeneous disease-propagating stem cells in juvenile myelomonocytic leukemia.
C/EBPα and GATA-2 Mutations Induce Bilineage Acute Erythroid Leukemia through Transformation of a Neomorphic Neutrophil-Erythroid Progenitor.
TARGET-Seq: A Protocol for High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
Unravelling Intratumoral Heterogeneity through High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
B Cell-Intrinsic Role for IRF5 in TLR9/BCR-Induced Human B Cell Activation, Proliferation, and Plasmablast Differentiation.
Checkpoints for Autoreactive B Cells in the Peripheral Blood of Lupus Patients Assessed by Flow Cytometry.
Junctional Adhesion Molecule-A Is Highly Expressed on Human Hematopoietic Repopulating Cells and Associates with the Key Hematopoietic Chemokine Receptor CXCR4.
Plasmacytoid dendritic cells promote HIV-1-induced group 3 innate lymphoid cell depletion.
Detailed characterization of tumor infiltrating lymphocytes in two distinct human solid malignancies show phenotypic similarities.
Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production.
CSK regulatory polymorphism is associated with systemic lupus erythematosus and influences B-cell signaling and activation.
Louka E, Povinelli B, Rodriguez-Meira A, Buck G, Wen WX, Wang G, Sousos N, Ashley N, Hamblin A, Booth CAG, Roy A, Elliott N, Iskander D, de la Fuente J, Fordham N, O'Byrne S, Inglott S, Norfo R, Salio M, Thongjuea S, Rao A, Roberts I, Mead AJ
The Journal of experimental medicine 2021 Feb 1;218(2)
The Journal of experimental medicine 2021 Feb 1;218(2)
C/EBPα and GATA-2 Mutations Induce Bilineage Acute Erythroid Leukemia through Transformation of a Neomorphic Neutrophil-Erythroid Progenitor.
Di Genua C, Valletta S, Buono M, Stoilova B, Sweeney C, Rodriguez-Meira A, Grover A, Drissen R, Meng Y, Beveridge R, Aboukhalil Z, Karamitros D, Belderbos ME, Bystrykh L, Thongjuea S, Vyas P, Nerlov C
Cancer cell 2020 May 11;37(5):690-704.e8
Cancer cell 2020 May 11;37(5):690-704.e8
TARGET-Seq: A Protocol for High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
Rodriguez-Meira A, O'Sullivan J, Rahman H, Mead AJ
STAR protocols 2020 Dec 18;1(3):100125
STAR protocols 2020 Dec 18;1(3):100125
Unravelling Intratumoral Heterogeneity through High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.
Rodriguez-Meira A, Buck G, Clark SA, Povinelli BJ, Alcolea V, Louka E, McGowan S, Hamblin A, Sousos N, Barkas N, Giustacchini A, Psaila B, Jacobsen SEW, Thongjuea S, Mead AJ
Molecular cell 2019 Mar 21;73(6):1292-1305.e8
Molecular cell 2019 Mar 21;73(6):1292-1305.e8
B Cell-Intrinsic Role for IRF5 in TLR9/BCR-Induced Human B Cell Activation, Proliferation, and Plasmablast Differentiation.
De S, Zhang B, Shih T, Singh S, Winkler A, Donnelly R, Barnes BJ
Frontiers in immunology 2017;8:1938
Frontiers in immunology 2017;8:1938
Checkpoints for Autoreactive B Cells in the Peripheral Blood of Lupus Patients Assessed by Flow Cytometry.
Malkiel S, Jeganathan V, Wolfson S, Manjarrez Orduño N, Marasco E, Aranow C, Mackay M, Gregersen PK, Diamond B
Arthritis & rheumatology (Hoboken, N.J.) 2016 Sep;68(9):2210-20
Arthritis & rheumatology (Hoboken, N.J.) 2016 Sep;68(9):2210-20
Junctional Adhesion Molecule-A Is Highly Expressed on Human Hematopoietic Repopulating Cells and Associates with the Key Hematopoietic Chemokine Receptor CXCR4.
Chang CH, Hale SJ, Cox CV, Blair A, Kronsteiner B, Grabowska R, Zhang Y, Cook D, Khoo CP, Schrader JB, Kabuga SB, Martin-Rendon E, Watt SM
Stem cells (Dayton, Ohio) 2016 Jun;34(6):1664-78
Stem cells (Dayton, Ohio) 2016 Jun;34(6):1664-78
Plasmacytoid dendritic cells promote HIV-1-induced group 3 innate lymphoid cell depletion.
Zhang Z, Cheng L, Zhao J, Li G, Zhang L, Chen W, Nie W, Reszka-Blanco NJ, Wang FS, Su L
The Journal of clinical investigation 2015 Sep;125(9):3692-703
The Journal of clinical investigation 2015 Sep;125(9):3692-703
Detailed characterization of tumor infiltrating lymphocytes in two distinct human solid malignancies show phenotypic similarities.
Kovacsovics-Bankowski M, Chisholm L, Vercellini J, Tucker CG, Montler R, Haley D, Newell P, Ma J, Tseng P, Wolf R, Vetto JT, Hammill C, Hansen P, Weinberg AD
Journal for immunotherapy of cancer 2014;2(1):38
Journal for immunotherapy of cancer 2014;2(1):38
Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production.
Perreau M, Savoye AL, De Crignis E, Corpataux JM, Cubas R, Haddad EK, De Leval L, Graziosi C, Pantaleo G
The Journal of experimental medicine 2013 Jan 14;210(1):143-56
The Journal of experimental medicine 2013 Jan 14;210(1):143-56
CSK regulatory polymorphism is associated with systemic lupus erythematosus and influences B-cell signaling and activation.
Manjarrez-Orduño N, Marasco E, Chung SA, Katz MS, Kiridly JF, Simpfendorfer KR, Freudenberg J, Ballard DH, Nashi E, Hopkins TJ, Cunninghame Graham DS, Lee AT, Coenen MJ, Franke B, Swinkels DW, Graham RR, Kimberly RP, Gaffney PM, Vyse TJ, Behrens TW, Criswell LA, Diamond B, Gregersen PK
Nature genetics 2012 Nov;44(11):1227-30
Nature genetics 2012 Nov;44(11):1227-30
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
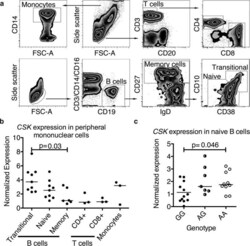
- Figure 2 CSK expression varies in lymphocyte subsets and is associated with CSK genotype. (a) Gating strategy employed to distinguish monocytes and T cells (upper panel) and B cell subpopulations (bottom panel). (b) CSK expression decreases in peripheral B cells as they mature from transitional to memory cells (p = 0.030, Kruskal-Wallis test). CSK expression in peripheral T cells is low (p = 0.0047, Kruskal-Wallis test between the five cell subpopulations). (c) In naive B cells, the rs34933034 risk allele (A) is associated with increased CSK expression (29 subjects analyzed, Kruskal-Wallis test). Cell subpopulations were isolated from peripheral mononuclear cells by cell sorting as shown in (a). Expression analysis was performed by qPCR with cDNA synthesized from RNA of isolated subpopulations from mononuclear cells of non-genotyped blood donors (b) or haplotype-matched GaP subjects (c), the horizontal bar marks the median of the values.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
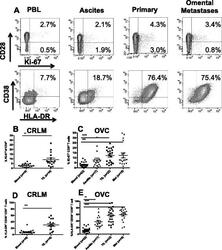
- Figure 5 CD8 + T cells phenotype in CRC and OVC patients. A . An example of flow cytometric analysis of CD3 + CD8 + T cells analyzed for CD28 and Ki-67, upper panels, and HLA-DR and CD38, lower panels. Percentage of Ki-67 + CD8 T cells in CRC samples (n = 16) B . and OVC samples (n = 22) C . Co-expression of CD38 and HLA-DR on CD8 + T cells in CRC samples (D) and OVC samples (E) .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
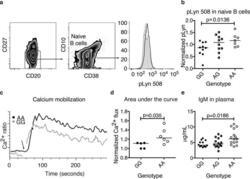
- Figure 3 The CSK risk allele is associated with increased phospho-Lyn 508 and enhanced activation of mature B cells. (a) Phosphorylated tyrosine 508 was measured by flow cytometry in naive B cells of haplotype-matched GaP donors homozygous for either allele of the rs34933034 variant; the profiles of a risk (bold) and non risk (shadowed) subject are shown, the thin line shows the isotype control; (b) pLyn 508 in carriers of the CSK risk allele. pLyn 508 was normalized to each day average (n = 27, p = 0.0136, Mann-Whitney test, see methods for correction after multiple comparison, and supplementary Figure 1 , Kruskal-Wallis test P = 0.0569). (c) A representative experiment of calcium mobilization. Basal calcium was read for 40 seconds, before activation with anti-IgM (Fab') 2 . After at least six minutes of recording, ionomycin was used as a positive control for calcium mobilization and showed no difference between genotypes (data not shown). (d) Data from eleven different ""B"" haplotype donors was normalized to one non-risk (GG) subject for comparison on each of four separate days of experiments. The area under the curve for the first 90 seconds (similar results at four minutes) is shown, (see methods for statistical analysis). (e) IgM plasma levels in 42 subjects homozygous for the B haplotype, Kruskal-Wallis test. In every graph, the horizontal line marks the median.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 The CSK risk allele is associated with expansion of transitional B cells in umbilical cord blood. (a) B cells were defined by a CD19 + gate (with CD3/CD14/CD16 exclusion); transitional cells were defined as CD38 hi , CD10 hi . The transitional cells were further divided into early and late stage by gain of IgD and decreased CD10 in late stage transitional B cells. The panel shows a representative plot of each homozygous genotype. (b) The percentage of transitional cells in the CD19 compartment is significantly higher in homozygous risk allele carriers compared with heterozygotes, with a similar trend in comparison with wild type homozygous individuals (n = 27, Kruskal-Wallis test). (c) Among transitional cells, there is an expansion of the late transitional compartment in individuals who carry the risk allele (Kruskal-Wallis test). The horizontal line marks the median.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry Other assay
Other assay