M801
antibody from Invitrogen Antibodies
Targeting: CXCL8
3-10C, AMCF-I, b-ENAP, GCP-1, GCP1, IL-8, IL8, K60, LECT, LUCT, LYNAP, MDNCF, MONAP, NAF, NAP-1, NAP1, SCYB8, TSG-1
Antibody data
- Antibody Data
- Antigen structure
- References [29]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- M801 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-8 (CXCL8) Monoclonal Antibody (3IL8-H10)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- M801 targets IL-8 in ELISA, and WB applications and shows reactivity with Human samples. The M801 immunogen is recombinant human IL-8. M801 detects IL-8 which has a predicted molecular weight of approximately 9 kDa. The M801 IL8 antibody (clone 3IL8-H10) has successfully been paired as the coating antibody in a sandwich ELISA with detection antibody M802B (biotinylated conjugate of clone I8-S2). Typical dilutions for sandwich ELISA include 1 µg/mL for coating and 0.125 - 0.25 µg/mL for detection. Antibody M801 (clone 3IL8-H10) and biotinylated antibody M802B (clone I8-S2) have successfully been used in combination with recombinant IL8 protein SIL8 in ELISA applications. This product has been tested for endotoxins by limulus amoebocyte lysate (LAL) assay and contains an endotoxin concentration of less than or equal to 10 endotoxin units per milligram (EU/mg).
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3IL8-H10
- Vial size
- 500 µg
- Concentration
- 1.0 mg/mL
- Storage
- -20°C
Submitted references Chronic effects of two rutile TiO(2) nanomaterials in human intestinal and hepatic cell lines.
Impact of Progerin Expression on Adipogenesis in Hutchinson-Gilford Progeria Skin-Derived Precursor Cells.
Neuropilin-1 is up-regulated by cancer-associated fibroblast-secreted IL-8 and associated with cell proliferation of gallbladder cancer.
The Antiseptic Octenidine Inhibits Langerhans Cell Activation and Modulates Cytokine Expression upon Superficial Wounding with Tape Stripping.
Extravascular gelation shrinkage-derived internal stress enables tumor starvation therapy with suppressed metastasis and recurrence.
Combined effects of okadaic acid and pectenotoxin-2, 13-desmethylspirolide C or yessotoxin in human intestinal Caco-2 cells.
Resolvin D2 decreases TLR4 expression to mediate resolution in human monocytes.
Microparticles engineered to highly express peroxisome proliferator-activated receptor-γ decreased inflammatory mediator production and increased adhesion of recipient monocytes.
The human salivary proteome is radiation responsive.
Attenuation of inflammatory mediator production by the NF-κB member RelB is mediated by microRNA-146a in lung fibroblasts.
Transforming growth factor-β impairs glucocorticoid activity in the A549 lung adenocarcinoma cell line.
Ocular fibroblast diversity: implications for inflammation and ocular wound healing.
Inhibitors of p38 suppress cytokine production in rheumatoid arthritis synovial membranes: does variable inhibition of interleukin-6 production limit effectiveness in vivo?
Intermittent haemodiafiltration in refractory congestive heart failure: BNP and balance of inflammatory cytokines.
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
Protein microarray platform for the multiplex analysis of biomarkers in human sera.
Expression of a Porphyromonas gingivalis lipid A palmitylacyltransferase in Escherichia coli yields a chimeric lipid A with altered ability to stimulate interleukin-8 secretion.
Regulation of myeloid cell function through the CD200 receptor.
Oxidized ATP (oATP) attenuates proinflammatory signaling via P2 receptor-independent mechanisms.
Monocyte chemoattractant protein-1 levels in bronchoalveolar lavage fluid of lung-transplanted patients treated with tacrolimus as rescue treatment for refractory acute rejection.
Monocyte chemoattractant protein-1 levels in bronchoalveolar lavage fluid of lung-transplanted patients treated with tacrolimus as rescue treatment for refractory acute rejection.
Development of protein microarray technology to monitor biomarkers of rheumatoid arthritis disease.
Development of protein microarray technology to monitor biomarkers of rheumatoid arthritis disease.
Regulation of interleukin 8 expression in human malignant melanoma cells.
Antineutrophil cytoplasmic antibodies induce monocyte IL-8 release. Role of surface proteinase-3, alpha1-antitrypsin, and Fcgamma receptors.
Secretion of proinflammatory cytokines by epithelial cells in response to Chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis.
Monocyte Fc gamma receptor cross-linking induces IL-8 production.
Rapamycin treatment depresses intragraft expression of KC/MIP-2, granzyme B, and IFN-gamma in rat recipients of cardiac allografts.
Jalili P, Krause BC, Lanceleur R, Burel A, Jungnickel H, Lampen A, Laux P, Luch A, Fessard V, Hogeveen K
Particle and fibre toxicology 2022 May 17;19(1):37
Particle and fibre toxicology 2022 May 17;19(1):37
Impact of Progerin Expression on Adipogenesis in Hutchinson-Gilford Progeria Skin-Derived Precursor Cells.
Najdi F, Krüger P, Djabali K
Cells 2021 Jun 25;10(7)
Cells 2021 Jun 25;10(7)
Neuropilin-1 is up-regulated by cancer-associated fibroblast-secreted IL-8 and associated with cell proliferation of gallbladder cancer.
Chen C, Zhang R, Ma L, Li Q, Zhao YL, Zhang GJ, Zhang D, Li WZ, Cao S, Wang L, Geng ZM
Journal of cellular and molecular medicine 2020 Nov;24(21):12608-12618
Journal of cellular and molecular medicine 2020 Nov;24(21):12608-12618
The Antiseptic Octenidine Inhibits Langerhans Cell Activation and Modulates Cytokine Expression upon Superficial Wounding with Tape Stripping.
Nikolić N, Kienzl P, Tajpara P, Vierhapper M, Matiasek J, Elbe-Bürger A
Journal of immunology research 2019;2019:5143635
Journal of immunology research 2019;2019:5143635
Extravascular gelation shrinkage-derived internal stress enables tumor starvation therapy with suppressed metastasis and recurrence.
Zhang K, Fang Y, He Y, Yin H, Guan X, Pu Y, Zhou B, Yue W, Ren W, Du D, Li H, Liu C, Sun L, Chen Y, Xu H
Nature communications 2019 Nov 26;10(1):5380
Nature communications 2019 Nov 26;10(1):5380
Combined effects of okadaic acid and pectenotoxin-2, 13-desmethylspirolide C or yessotoxin in human intestinal Caco-2 cells.
Alarcan J, Barbé S, Kopp B, Hessel-Pras S, Braeuning A, Lampen A, Le Hégarat L, Fessard V
Chemosphere 2019 Aug;228:139-148
Chemosphere 2019 Aug;228:139-148
Resolvin D2 decreases TLR4 expression to mediate resolution in human monocytes.
Croasdell A, Sime PJ, Phipps RP
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Sep;30(9):3181-93
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Sep;30(9):3181-93
Microparticles engineered to highly express peroxisome proliferator-activated receptor-γ decreased inflammatory mediator production and increased adhesion of recipient monocytes.
Sahler J, Woeller CF, Phipps RP
PloS one 2014;9(11):e113189
PloS one 2014;9(11):e113189
The human salivary proteome is radiation responsive.
Moore HD, Ivey RG, Voytovich UJ, Lin C, Stirewalt DL, Pogosova-Agadjanyan EL, Paulovich AG
Radiation research 2014 May;181(5):521-30
Radiation research 2014 May;181(5):521-30
Attenuation of inflammatory mediator production by the NF-κB member RelB is mediated by microRNA-146a in lung fibroblasts.
McMillan DH, Woeller CF, Thatcher TH, Spinelli SL, Maggirwar SB, Sime PJ, Phipps RP
American journal of physiology. Lung cellular and molecular physiology 2013 Jun 1;304(11):L774-81
American journal of physiology. Lung cellular and molecular physiology 2013 Jun 1;304(11):L774-81
Transforming growth factor-β impairs glucocorticoid activity in the A549 lung adenocarcinoma cell line.
Salem S, Harris T, Mok JS, Li MY, Keenan CR, Schuliga MJ, Stewart AG
British journal of pharmacology 2012 Aug;166(7):2036-48
British journal of pharmacology 2012 Aug;166(7):2036-48
Ocular fibroblast diversity: implications for inflammation and ocular wound healing.
Xi X, McMillan DH, Lehmann GM, Sime PJ, Libby RT, Huxlin KR, Feldon SE, Phipps RP
Investigative ophthalmology & visual science 2011 Jul 1;52(7):4859-65
Investigative ophthalmology & visual science 2011 Jul 1;52(7):4859-65
Inhibitors of p38 suppress cytokine production in rheumatoid arthritis synovial membranes: does variable inhibition of interleukin-6 production limit effectiveness in vivo?
Page TH, Brown A, Timms EM, Foxwell BM, Ray KP
Arthritis and rheumatism 2010 Nov;62(11):3221-31
Arthritis and rheumatism 2010 Nov;62(11):3221-31
Intermittent haemodiafiltration in refractory congestive heart failure: BNP and balance of inflammatory cytokines.
Libetta C, Sepe V, Zucchi M, Pisacco P, Cosmai L, Meloni F, Campana C, Rampino T, Monti C, Tavazzi L, Dal Canton A
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2007 Jul;22(7):2013-9
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2007 Jul;22(7):2013-9
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
Saffar AS, Alphonse MP, Shan L, Hayglass KT, Simons FE, Gounni AS
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
Saffar AS, Alphonse MP, Shan L, Hayglass KT, Simons FE, Gounni AS
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
Protein microarray platform for the multiplex analysis of biomarkers in human sera.
Urbanowska T, Mangialaio S, Zickler C, Cheevapruk S, Hasler P, Regenass S, Legay F
Journal of immunological methods 2006 Oct 20;316(1-2):1-7
Journal of immunological methods 2006 Oct 20;316(1-2):1-7
Expression of a Porphyromonas gingivalis lipid A palmitylacyltransferase in Escherichia coli yields a chimeric lipid A with altered ability to stimulate interleukin-8 secretion.
Bainbridge BW, Coats SR, Pham TT, Reife RA, Darveau RP
Cellular microbiology 2006 Jan;8(1):120-9
Cellular microbiology 2006 Jan;8(1):120-9
Regulation of myeloid cell function through the CD200 receptor.
Jenmalm MC, Cherwinski H, Bowman EP, Phillips JH, Sedgwick JD
Journal of immunology (Baltimore, Md. : 1950) 2006 Jan 1;176(1):191-9
Journal of immunology (Baltimore, Md. : 1950) 2006 Jan 1;176(1):191-9
Oxidized ATP (oATP) attenuates proinflammatory signaling via P2 receptor-independent mechanisms.
Beigi RD, Kertesy SB, Aquilina G, Dubyak GR
British journal of pharmacology 2003 Oct;140(3):507-19
British journal of pharmacology 2003 Oct;140(3):507-19
Monocyte chemoattractant protein-1 levels in bronchoalveolar lavage fluid of lung-transplanted patients treated with tacrolimus as rescue treatment for refractory acute rejection.
Meloni F, Cascina A, Paschetto E, Marone Bianco A, Morosini M, Pellegrini C, Fietta A, Vitulo P, Pozzi E, Viganò M
Transplantation proceedings 2003 Jun;35(4):1523-6
Transplantation proceedings 2003 Jun;35(4):1523-6
Monocyte chemoattractant protein-1 levels in bronchoalveolar lavage fluid of lung-transplanted patients treated with tacrolimus as rescue treatment for refractory acute rejection.
Meloni F, Cascina A, Paschetto E, Marone Bianco A, Morosini M, Pellegrini C, Fietta A, Vitulo P, Pozzi E, Viganò M
Transplantation proceedings 2003 Jun;35(4):1523-6
Transplantation proceedings 2003 Jun;35(4):1523-6
Development of protein microarray technology to monitor biomarkers of rheumatoid arthritis disease.
Urbanowska T, Mangialaio S, Hartmann C, Legay F
Cell biology and toxicology 2003 Jun;19(3):189-202
Cell biology and toxicology 2003 Jun;19(3):189-202
Development of protein microarray technology to monitor biomarkers of rheumatoid arthritis disease.
Urbanowska T, Mangialaio S, Hartmann C, Legay F
Cell biology and toxicology 2003 Jun;19(3):189-202
Cell biology and toxicology 2003 Jun;19(3):189-202
Regulation of interleukin 8 expression in human malignant melanoma cells.
Singh RK, Varney ML
Cancer research 1998 Apr 1;58(7):1532-7
Cancer research 1998 Apr 1;58(7):1532-7
Antineutrophil cytoplasmic antibodies induce monocyte IL-8 release. Role of surface proteinase-3, alpha1-antitrypsin, and Fcgamma receptors.
Ralston DR, Marsh CB, Lowe MP, Wewers MD
The Journal of clinical investigation 1997 Sep 15;100(6):1416-24
The Journal of clinical investigation 1997 Sep 15;100(6):1416-24
Secretion of proinflammatory cytokines by epithelial cells in response to Chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis.
Rasmussen SJ, Eckmann L, Quayle AJ, Shen L, Zhang YX, Anderson DJ, Fierer J, Stephens RS, Kagnoff MF
The Journal of clinical investigation 1997 Jan 1;99(1):77-87
The Journal of clinical investigation 1997 Jan 1;99(1):77-87
Monocyte Fc gamma receptor cross-linking induces IL-8 production.
Marsh CB, Gadek JE, Kindt GC, Moore SA, Wewers MD
Journal of immunology (Baltimore, Md. : 1950) 1995 Sep 15;155(6):3161-7
Journal of immunology (Baltimore, Md. : 1950) 1995 Sep 15;155(6):3161-7
Rapamycin treatment depresses intragraft expression of KC/MIP-2, granzyme B, and IFN-gamma in rat recipients of cardiac allografts.
Wieder KJ, Hancock WW, Schmidbauer G, Corpier CL, Wieder I, Kobzik L, Strom TB, Kupiec-Weglinski JW
Journal of immunology (Baltimore, Md. : 1950) 1993 Jul 15;151(2):1158-66
Journal of immunology (Baltimore, Md. : 1950) 1993 Jul 15;151(2):1158-66
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
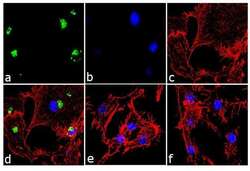
- Experimental details
- Immunofluorescence analysis of IL-8 was performed using 70% confluent log phase U-87 MG cells treated 1 uM thapsigargin for 24 hours. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with IL-8 (3IL8-H10) Mouse Monoclonal Antibody (Product # M801) at 2µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjµgate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e is untreated cell with no signal. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
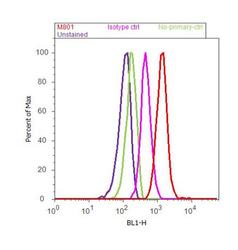
- Experimental details
- Flow cytometry analysis of IL-8 was done on U-87 MG cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with IL-8 Mouse Monoclonal Antibody (M801, red histogram) or with mouse isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control..
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
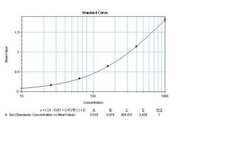
- Experimental details
- Sandwich ELISA analysis of human IL-8 was performed using a Human IL-8 Colorimetric ELISA kit (Product # EH2IL8) by loading 50 µL per well of Human IL-8 Recombinant Protein (Product # SIL8) in dodecuplicate at 1000, 400, 160, 64, 25.6, and 0 pg/mL across a 3 µg/mL mouse anti-Human IL-8 (Product # M801) pre-coated plate and incubating for 1 hour at room temperature. The plate was washed, and then incubated with 50 µL per well of a biotinylated mouse anti-human IL-8 monoclonal antibody (Product # M802B) in duplicate at 0.25 µg/mL for 1 hour at room temperature. The plate was washed and incubated with 100 µL per well of Streptavidin-HRP (Product # N504) in all test wells at a 1:4,000 dilution for 30 minutes at room temperature. Detection was performed using 1-Step Ultra TMB substrate (Product # 34028) for 30 minutes at room temperature in the dark. The plate was then stopped with 0.16M sulfuric acid. Absorbances were read on a spectrophotometer at 450-550 nm.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
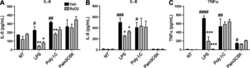
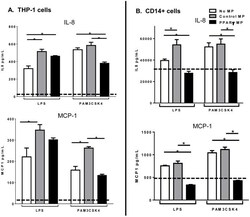
- Figure 4 Microparticle composition influences inflammatory cytokine production from activated monocytes. THP-1 cells ( A ) or CD14 sorted blood monocytes ( B ) were treated with control microparticles (grey bars) or PPARgamma-overexpressing microparticles (black bars) or no microparticles (white bars) for 4 hours before activation with LPS or PAM3CSK4 for 24 hours. Supernatants were collected and pro-inflammatory cytokines IL-8 (top), MCP-1 (bottom) were measured by ELISA. Dotted line represents baseline cytokine production from unactivated cells with no microparticle exposure. Mean values with standard errors represent one of at least 3 experiments. Two-way ANOVA with Tukey's multiple comparison post test was performed to determine statistical significance. * indicates (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
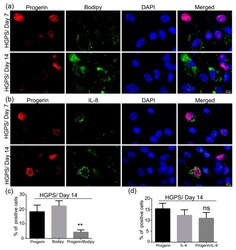
- Figure 9 Co-expression of progerin with Bodipy and IL-8 in HGPS differentiated SKPs. ( a ) Immunofluorescence staining for progerin and Bodipy at days 7 and 14 of adipogenesis in HGPS (HGADFN127). ( b ) Immunofluorescence staining for progerin and IL-8 at days 7 and 14 of adipogenesis in HGPS. Cells were counterstained with DAPI. ( c ) Percentage of positive nuclei expressing either progerin, Bodipy, or both (progerin/Bodipy 493/503). ( d ) Percentage of progerin-positive nuclei, IL-8-positive or progerin/IL-8-positive cells. Values are presented as mean +- SD ( n = 3), not significant (ns), ** p < 0.01, ( c , d ) unpaired t -test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
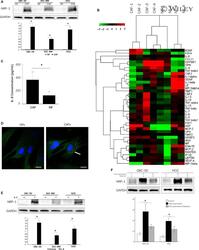
- 2 FIGURE CAFs up-regulated NRP-1 expression by IL-8 in gallbladder cancer (GBC). (A) CAFs up-regulated NRP-1 expression in different GBC cell lines (GBC-SD, SGC-996 and NOZ). (B) Cytokine array cluster analysis of the exocrine cytokines in CAFs and NFs. (C) The IL-8 expression in CAFs and NFs were determined by ELISA Kit. (D) IL-8 in NFs and CAFs cells. Green: IL-8 (marked with arrow); Blue: nuclei (stained with DAPI). Scale bar: 25 mum. (E) IL-8 up-regulated NRP-1 expression in different GBC cell lines (GBC-SD, SGC-996 and NOZ). (F) IL-8 receptors inhibitor Reparixin reduced the expression of NRP-1 in GBC cell lines which up-regulated by CAFs supernatants (GBC-SD, NOZ). * P < .05 by ANOVA
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry