Antibody data
- Antibody Data
- Antigen structure
- References [2]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-37518 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IFN alpha Monoclonal Antibody (278CT1)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 278CT1
- Vial size
- 400 µL
- Concentration
- 0.5 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Defining the Innate Immune Responses for SARS-CoV-2-Human Macrophage Interactions.
Defining the Immune Responses for SARS-CoV-2-Human Macrophage Interactions.
Abdelmoaty MM, Yeapuri P, Machhi J, Olson KE, Shahjin F, Kumar V, Zhou Y, Liang J, Pandey K, Acharya A, Byrareddy SN, Mosley RL, Gendelman HE
Frontiers in immunology 2021;12:741502
Frontiers in immunology 2021;12:741502
Defining the Immune Responses for SARS-CoV-2-Human Macrophage Interactions.
Abdelmoaty M, Yeapuri P, Machhi J, Olson K, Shahjin F, Zhou Y, Jingjing L, Pandey K, Acharya A, Byrareddy S, Mosley L, Gendelman H
bioRxiv : the preprint server for biology 2021 Jul 15;
bioRxiv : the preprint server for biology 2021 Jul 15;
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western Blot analysis of IFN alpha using the IFN alpha Monoclonal Antibody (Product # MA5-37518) in IFN cell line lysates (35 µg/lane).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
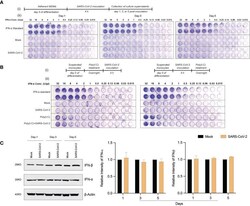
- Experimental details
- Figure 3 SARS-CoV-2 does not induce IFN activity in monocytes-macrophages. (A) MDMs were infected with SARS-CoV-2 (MOI=0.01) and the culture supernatants were collected at days 1, 3, and 5 post-infection. The collected culture supernatants were used to assess IFN activity in VSV-challenged bovine MDBK cells. Recombinant human IFN-alpha was used as standard (64 U/mul). 2-fold dilution was used for different conditions. ( A. i) Experimental timeline. ( A. ii) Representative images of IFN activity assay plates. (B) No additive or synergistic responses in poly(I:C)-induced IFN activity between control and SARS-CoV-2-challenged monocytes. Suspended monocytes were treated with 100 mug/ml poly(I:C) overnight before or after the infection with SARS-CoV-2 (MOI=0.01) for 4 h. ( B. i) Experimental timeline. (B .ii ) Representative images of IFN activity assay plates. (C) Western blot analysis was performed to determine expression of IFN-alpha and IFN-beta in cell lysates at different times after viral exposure. Representative immunoblot and densitometric quantification are shown. All experiments were done at least twice, and one representative image is shown. (C) Data represent mean +- SEM (n=3 donors). Statistical significance between the groups was determined with unpaired Student's t-test and p < 0.05 was considered significant.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot