Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Western blot [3]
- Immunohistochemistry [2]
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-78968 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- DC-SIGN (CD209) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Reconstitute with 0.2 mL of distilled water to yield a concentration of 500 µg/mL.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 500 µg/mL
- Storage
- -20°C
Submitted references The use of patient-derived breast tissue explants to study macrophage polarization and the effects of environmental chemical exposure.
Gregory KJ, Morin SM, Kubosiak A, Ser-Dolansky J, Schalet BJ, Jerry DJ, Schneider SS
Immunology and cell biology 2020 Nov;98(10):883-896
Immunology and cell biology 2020 Nov;98(10):883-896
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
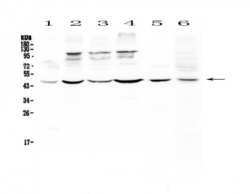
- Experimental details
- Western blot analysis of DC-SIGN (CD209) in Lane 1: human HeLa whole cell lysate, Lane 2: human MCF-7 whole cell lysate, Lane 3: human HepG2 whole cell lysate, Lane 4: human A549 whole cell lysate, Lane 5: rat spleen tissue lysate, Lane 6: mouse thymus tissue lysate using 50 µg (reducing conditions) per well. Electrophoresis was performed on 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours and protein was transferred to a nitrocellulose membrane at 150mA for 50-90 minutes. Sample was blocked with 5% Non-fat Milk/TBS for 1.5 hours at room temperature, incubated with DC-SIGN (CD209) polyclonal antibody (Product # PA5-78968) at a dilution of 0.5 µg/mL (overnight at 4°C), followed by goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:10,000. Signal development was performed using a chemiluminescence (ECL) kit.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
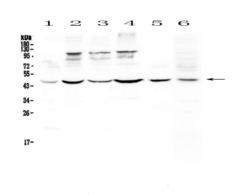
- Experimental details
- Western blot analysis of DC-SIGN (CD209) in Lane 1: human HeLa whole cell lysate, Lane 2: human MCF-7 whole cell lysate, Lane 3: human HepG2 whole cell lysate, Lane 4: human A549 whole cell lysate, Lane 5: rat spleen tissue lysate, Lane 6: mouse thymus tissue lysate using 50 µg (reducing conditions) per well. Electrophoresis was performed on 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours and protein was transferred to a nitrocellulose membrane at 150mA for 50-90 minutes. Sample was blocked with 5% Non-fat Milk/TBS for 1.5 hours at room temperature, incubated with DC-SIGN (CD209) polyclonal antibody (Product # PA5-78968) at a dilution of 0.5 µg/mL (overnight at 4°C), followed by goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:10,000. Signal development was performed using a chemiluminescence (ECL) kit.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
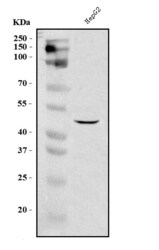
- Experimental details
- Western blot analysis of DC-SIGN in, Lane 1: human HepG2 whole cell lysates. Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 µg of sample under reducing conditions. After Electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. The membrane was blocked with 5% non-fat milk/TBS for 1. 5 hour at RT. The membrane was incubated with DC-SIGN (CD209) Polyclonal Antibody (Product # PA5-78968) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0. 1% Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5,000 for 1. 5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit. A specific band was detected for DC-SIGN at approximately 46 kDa. The expected band size for DC-SIGN is at 46 kDa.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
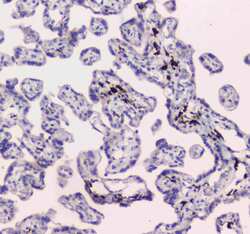
- Experimental details
- Immunohistochemistry analysis of DC-SIGN (CD209) on paraffin-embedded human placenta tissue. Antigen retrieval was performed using citrate buffer (pH6, epitope retrieval solution) for 20 mins. Sample was blocked using 10% goat serum, incubated with DC-SIGN (CD209) polyclonal antibody (Product# PA5-78968) with a dilution of 1 µg/mL (overnight at 4°C), and followed by biotinylated goat anti-rabbit IgG (30 minutes at 37°C). Development was performed using Streptavidin-Biotin-Complex (SABC) with DAB chromogen method.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
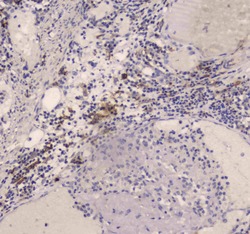
- Experimental details
- Immunohistochemistry analysis of DC-SIGN (CD209) on paraffin-embedded human intestinal cancer tissue. Antigen retrieval was performed using citrate buffer (pH6, epitope retrieval solution) for 20 mins. Sample was blocked using 10% goat serum, incubated with DC-SIGN (CD209) polyclonal antibody (Product# PA5-78968) with a dilution of 1 µg/mL (overnight at 4°C), and followed by biotinylated goat anti-rabbit IgG (30 minutes at 37°C). Development was performed using Streptavidin-Biotin-Complex (SABC) with DAB chromogen method.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
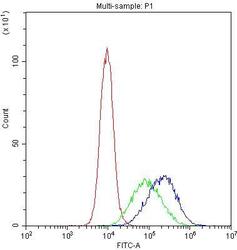
- Experimental details
- Flow Cytometry of DC-SIGN (CD209) in THP-1 cells (blue line), isotype control rabbit IgG (green line) and unlabeled (red line). Samples were blocked with 10% goat serum, incubated with DC-SIGN (CD209) Polyclonal Antibody (Product # PA5-78968) at a dilution of 1 μg (per 1x10^6 cells), followed by DyLight®488 conjugated goat anti-rabbit IgG (for 30 minutes at 20°C) using 5-10 μg (per 1x10^6 cells) dilution.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
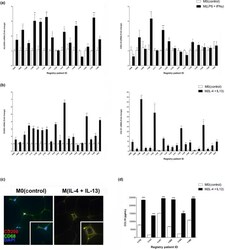
- Experimental details
- Figure 1 Normal breast PDEs can be polarized toward M(IFNgamma + LPS) or M(IL-4 + IL-13). RNA was harvested from cytokine-exposed PDEs and mRNA levels of (a) HLA-DRA and CXCL10 (15 patients) or (b) CD209 and CCL18 (20 patients) were analyzed by real-time PCR. All real-time PCR results are from two separate experiments (performed technical duplicate) and results were normalized to amplification of CD68 (macrophage marker). (c) PDE sections were subjected to fluorescent immunohistochemical analysis, dual stained for CD68 and CD209 and merged images were captured at 4000x. Representative pictures are displayed for tissues from each treatment group. (d) Supernatant was collected from PDEs (six patients) treated with IL-4 + IL-13 cytokines and CCL18 protein secretion was measured by ELISA performed in biological triplicate and technical duplicate M0(control) PDEs. Data within each bar represent triplicate samples isolated from individual patients, are presented as mean +- s.e.m. and are expressed as fold change with respect to M0(control) PDEs. * P < 0.05, ** P < 0.01, *** P < 0.001 (significantly different from indicated data set using a Student's t -test). IFN, interferon; IL, interleukin; LPS, lipopolysaccharide; mRNA, messenger RNA; PDE, patient-derived explant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
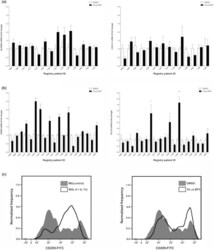
- Experimental details
- Figure 2 The xenoestrogen, BP3, can increase the expression of M2a markers in a subset of PDEs. RNA was harvested from PDEs treated with either vehicle (DMSO) or 30 mu m BP3 and mRNA levels of (a) HLA-DRA and CXL10 (14 patients) or (b) CD209 and CCL18 (19 patients) were analyzed by real-time PCR. All real-time PCR results are from two separate experiments (performed in technical duplicate) and results were normalized to amplification of CD68 (macrophage marker). Data within each bar represent triplicate samples isolated from individual patients, are presented as mean +- s.e.m. and are expressed as fold change with respect to DMSO treated M0(control) PDEs. * P < 0.05, ** P < 0.01, *** P < 0.001 (significantly different from indicated data set using a Student's t -test). (c) Cell surface protein expression of CD209 in treated primary human macrophages was measured by flow cytometry. BP3, benzophenone-3; DMSO, dimethyl sulfoxide; FITC, fluorescein isothiocyanate; mRNA, messenger RNA; PDE, patient-derived explant.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot