Antibody data
- Antibody Data
- Antigen structure
- References [40]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Immunohistochemistry [7]
- Other assay [14]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 53-9003-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor™ 488, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibodies AE1 and AE3 recognize many of the acidic and basic cytokeratin family members. Cytokeratins are intermediate filament proteins comprising one component of the cytoskeleton. There are two large families of cytokeratins, acidic and basic, but all contain the same basic domains (i.e. an alpha-helical core with an N- and C-terminal domain). The proteins are expressed in epithelial cells, but are developmentally regulated. Many tumors also express these proteins and their expression can help identify the origin of a neoplasm.
- Conjugate
- Green dye
- Antibody clone number
- AE1/AE3
- Concentration
- 0.5 mg/mL
Submitted references miR-126-3p-loaded small extracellular vesicles secreted by urine-derived stem cells released from a phototriggered imine crosslink hydrogel could enhance vaginal epithelization after vaginoplasty.
Analysis of the glyco-code in pancreatic ductal adenocarcinoma identifies glycan-mediated immune regulatory circuits.
TALPID3/KIAA0586 Regulates Multiple Aspects of Neuromuscular Patterning During Gastrointestinal Development in Animal Models and Human.
Functional cooperation between co-amplified genes promotes aggressive phenotypes of HER2-positive breast cancer.
Dexamethasone Creates a Suppressive Microenvironment and Promotes Aspergillus fumigatus Invasion in a Human 3D Epithelial/Immune Respiratory Model.
The Extracellular Small Leucine-Rich Proteoglycan Biglycan Is a Key Player in Gastric Cancer Aggressiveness.
CD74 and CD44 Expression on CTCs in Cancer Patients with Brain Metastasis.
Clinical Significance of TWIST-Positive Circulating Tumor Cells in Patients with Esophageal Squamous Cell Carcinoma.
Prognostic impact of CD73 expression and its relationship to PD-L1 in patients with radically treated pancreatic cancer.
Cysteine-Rich Angiogenic Inducer 61: Pro-Survival Function and Role as a Biomarker for Disseminating Breast Cancer Cells.
Differential pre-malignant programs and microenvironment chart distinct paths to malignancy in human colorectal polyps.
Detection of EGFR Mutations in cfDNA and CTCs, and Comparison to Tumor Tissue in Non-Small-Cell-Lung-Cancer (NSCLC) Patients.
PD-L1 Expression on Circulating Tumour Cells May Be Predictive of Response to Regorafenib in Patients Diagnosed with Chemorefractory Metastatic Colorectal Cancer.
Polymorphonuclear MDSCs are enriched in the stroma and expanded in metastases of prostate cancer.
Direct differentiation of cord blood derived mesenchymal stem cells into keratinocytes without feeder layers and cAMP inducers.
Evaluation of Microfluidic Ceiling Designs for the Capture of Circulating Tumor Cells on a Microarray Platform.
The Repertoire of Serous Ovarian Cancer Non-genetic Heterogeneity Revealed by Single-Cell Sequencing of Normal Fallopian Tube Epithelial Cells.
A single cell atlas of the human liver tumor microenvironment.
Temporal and spatial heterogeneity of host response to SARS-CoV-2 pulmonary infection.
Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome.
Temporal and Spatial Heterogeneity of Host Response to SARS-CoV-2 Pulmonary Infection.
Loss of the Krüppel-like factor 4 tumor suppressor is associated with epithelial-mesenchymal transition in colorectal cancer.
Highly multiplexed immunofluorescence images and single-cell data of immune markers in tonsil and lung cancer.
Screening Circulating Tumor Cells as a Noninvasive Cancer Test in 3388 Individuals from High-Risk Groups (ICELLATE2).
Cell signaling heterogeneity is modulated by both cell-intrinsic and -extrinsic mechanisms: An integrated approach to understanding targeted therapy.
Betamethasone prevents human rhinovirus- and cigarette smoke- induced loss of respiratory epithelial barrier function.
The VAR2CSA malaria protein efficiently retrieves circulating tumor cells in an EpCAM-independent manner.
Workflow optimization of whole genome amplification and targeted panel sequencing for CTC mutation detection.
Neither epithelial nor mesenchymal circulating tumor cells isolated from breast cancer patients are tumorigenic in NOD-scid Il2rg(null) mice.
Modified Leukocyte Filter Removes Tumor Cells from the Salvaged Blood.
Mutagenic, surviving and tumorigenic effects of follicular fluid in the context of p53 loss: initiation of fimbria carcinogenesis.
Whole-body tissue stabilization and selective extractions via tissue-hydrogel hybrids for high-resolution intact circuit mapping and phenotyping.
A breast cancer stem cell niche supported by juxtacrine signalling from monocytes and macrophages.
In vitro validation of an ultra-sensitive scanning fluorescence microscope for analysis of circulating tumor cells.
Tyrosine phosphoproteomics identifies both codrivers and cotargeting strategies for T790M-related EGFR-TKI resistance in non-small cell lung cancer.
Size-based isolation of circulating tumor cells in lung cancer patients using a microcavity array system.
Isolation of rare tumor cells from blood cells with buoyant immuno-microbubbles.
Flow cytometric determination of stem/progenitor content in epithelial tissues: an example from nonsmall lung cancer and normal lung.
Binding and isolation of tumor cells in biological media with perfluorocarbon microbubbles.
KIT (CD117) expression in a subset of non-small cell lung carcinoma (NSCLC) patients.
Xu Y, Qiu Y, Lin Q, Huang C, Li J, Chen L, Xue Z, Wu Q, Wang Y
Stem cell research & therapy 2022 Jul 23;13(1):331
Stem cell research & therapy 2022 Jul 23;13(1):331
Analysis of the glyco-code in pancreatic ductal adenocarcinoma identifies glycan-mediated immune regulatory circuits.
Rodriguez E, Boelaars K, Brown K, Madunić K, van Ee T, Dijk F, Verheij J, Li RJE, Schetters STT, Meijer LL, Le Large TYS, Driehuis E, Clevers H, Bruijns SCM, O'Toole T, van Vliet SJ, Bijlsma MF, Wuhrer M, Kazemier G, Giovannetti E, Garcia-Vallejo JJ, van Kooyk Y
Communications biology 2022 Jan 11;5(1):41
Communications biology 2022 Jan 11;5(1):41
TALPID3/KIAA0586 Regulates Multiple Aspects of Neuromuscular Patterning During Gastrointestinal Development in Animal Models and Human.
Delalande JM, Nagy N, McCann CJ, Natarajan D, Cooper JE, Carreno G, Dora D, Campbell A, Laurent N, Kemos P, Thomas S, Alby C, Attié-Bitach T, Lyonnet S, Logan MP, Goldstein AM, Davey MG, Hofstra RMW, Thapar N, Burns AJ
Frontiers in molecular neuroscience 2021;14:757646
Frontiers in molecular neuroscience 2021;14:757646
Functional cooperation between co-amplified genes promotes aggressive phenotypes of HER2-positive breast cancer.
Yang Y, Leonard M, Luo Z, Yeo S, Bick G, Hao M, Cai C, Charif M, Wang J, Guan JL, Lower EE, Zhang X
Cell reports 2021 Mar 9;34(10):108822
Cell reports 2021 Mar 9;34(10):108822
Dexamethasone Creates a Suppressive Microenvironment and Promotes Aspergillus fumigatus Invasion in a Human 3D Epithelial/Immune Respiratory Model.
Luvanda MK, Posch W, Noureen A, Lafon E, Zaderer V, Lass-Flörl C, Wilflingseder D
Journal of fungi (Basel, Switzerland) 2021 Mar 18;7(3)
Journal of fungi (Basel, Switzerland) 2021 Mar 18;7(3)
The Extracellular Small Leucine-Rich Proteoglycan Biglycan Is a Key Player in Gastric Cancer Aggressiveness.
Pinto F, Santos-Ferreira L, Pinto MT, Gomes C, Reis CA
Cancers 2021 Mar 16;13(6)
Cancers 2021 Mar 16;13(6)
CD74 and CD44 Expression on CTCs in Cancer Patients with Brain Metastasis.
Loreth D, Schuette M, Zinke J, Mohme M, Piffko A, Schneegans S, Stadler J, Janning M, Loges S, Joosse SA, Lamszus K, Westphal M, Müller V, Glatzel M, Matschke J, Gebhardt C, Schneider SW, Belczacka I, Volkmer B, Greinert R, Yaspo ML, Harter PN, Pantel K, Wikman H
International journal of molecular sciences 2021 Jun 29;22(13)
International journal of molecular sciences 2021 Jun 29;22(13)
Clinical Significance of TWIST-Positive Circulating Tumor Cells in Patients with Esophageal Squamous Cell Carcinoma.
Lee HJ, Kim GH, Park SJ, Kwon CH, Lee MW, Lee BE, Baek DH, I H
Gut and liver 2021 Jul 15;15(4):553-561
Gut and liver 2021 Jul 15;15(4):553-561
Prognostic impact of CD73 expression and its relationship to PD-L1 in patients with radically treated pancreatic cancer.
Tahkola K, Ahtiainen M, Kellokumpu I, Mecklin JP, Laukkarinen J, Laakkonen J, Kenessey I, Jalkanen S, Salmi M, Böhm J
Virchows Archiv : an international journal of pathology 2021 Feb;478(2):209-217
Virchows Archiv : an international journal of pathology 2021 Feb;478(2):209-217
Cysteine-Rich Angiogenic Inducer 61: Pro-Survival Function and Role as a Biomarker for Disseminating Breast Cancer Cells.
Bartkowiak K, Heidrich I, Kwiatkowski M, Gorges TM, Andreas A, Geffken M, Verpoort K, Müller V, Schlüter H, Pantel K
Cancers 2021 Feb 2;13(3)
Cancers 2021 Feb 2;13(3)
Differential pre-malignant programs and microenvironment chart distinct paths to malignancy in human colorectal polyps.
Chen B, Scurrah CR, McKinley ET, Simmons AJ, Ramirez-Solano MA, Zhu X, Markham NO, Heiser CN, Vega PN, Rolong A, Kim H, Sheng Q, Drewes JL, Zhou Y, Southard-Smith AN, Xu Y, Ro J, Jones AL, Revetta F, Berry LD, Niitsu H, Islam M, Pelka K, Hofree M, Chen JH, Sarkizova S, Ng K, Giannakis M, Boland GM, Aguirre AJ, Anderson AC, Rozenblatt-Rosen O, Regev A, Hacohen N, Kawasaki K, Sato T, Goettel JA, Grady WM, Zheng W, Washington MK, Cai Q, Sears CL, Goldenring JR, Franklin JL, Su T, Huh WJ, Vandekar S, Roland JT, Liu Q, Coffey RJ, Shrubsole MJ, Lau KS
Cell 2021 Dec 22;184(26):6262-6280.e26
Cell 2021 Dec 22;184(26):6262-6280.e26
Detection of EGFR Mutations in cfDNA and CTCs, and Comparison to Tumor Tissue in Non-Small-Cell-Lung-Cancer (NSCLC) Patients.
Liu HE, Vuppalapaty M, Wilkerson C, Renier C, Chiu M, Lemaire C, Che J, Matsumoto M, Carroll J, Crouse S, Hanft VR, Jeffrey SS, Di Carlo D, Garon EB, Goldman J, Sollier E
Frontiers in oncology 2020;10:572895
Frontiers in oncology 2020;10:572895
PD-L1 Expression on Circulating Tumour Cells May Be Predictive of Response to Regorafenib in Patients Diagnosed with Chemorefractory Metastatic Colorectal Cancer.
Raimondi L, Raimondi FM, Di Benedetto L, Cimino G, Spinelli GP
International journal of molecular sciences 2020 Sep 20;21(18)
International journal of molecular sciences 2020 Sep 20;21(18)
Polymorphonuclear MDSCs are enriched in the stroma and expanded in metastases of prostate cancer.
Wen J, Huang G, Liu S, Wan J, Wang X, Zhu Y, Kaliney W, Zhang C, Cheng L, Wen X, Lu X
The journal of pathology. Clinical research 2020 Jul;6(3):171-177
The journal of pathology. Clinical research 2020 Jul;6(3):171-177
Direct differentiation of cord blood derived mesenchymal stem cells into keratinocytes without feeder layers and cAMP inducers.
Ghauri AK, Wahid M, Mirza T, Uddin JAA
Pakistan journal of medical sciences 2020 Jul-Aug;36(5):946-951
Pakistan journal of medical sciences 2020 Jul-Aug;36(5):946-951
Evaluation of Microfluidic Ceiling Designs for the Capture of Circulating Tumor Cells on a Microarray Platform.
Liu HY, Koch C, Haller A, Joosse SA, Kumar R, Vellekoop MJ, Horst LJ, Keller L, Babayan A, Failla AV, Jensen J, Peine S, Keplinger F, Fuchs H, Pantel K, Hirtz M
Advanced biosystems 2020 Feb;4(2):e1900162
Advanced biosystems 2020 Feb;4(2):e1900162
The Repertoire of Serous Ovarian Cancer Non-genetic Heterogeneity Revealed by Single-Cell Sequencing of Normal Fallopian Tube Epithelial Cells.
Hu Z, Artibani M, Alsaadi A, Wietek N, Morotti M, Shi T, Zhong Z, Santana Gonzalez L, El-Sahhar S, Carrami EM, Mallett G, Feng Y, Masuda K, Zheng Y, Chong K, Damato S, Dhar S, Campo L, Garruto Campanile R, Soleymani Majd H, Rai V, Maldonado-Perez D, Jones S, Cerundolo V, Sauka-Spengler T, Yau C, Ahmed AA
Cancer cell 2020 Feb 10;37(2):226-242.e7
Cancer cell 2020 Feb 10;37(2):226-242.e7
A single cell atlas of the human liver tumor microenvironment.
Massalha H, Bahar Halpern K, Abu-Gazala S, Jana T, Massasa EE, Moor AE, Buchauer L, Rozenberg M, Pikarsky E, Amit I, Zamir G, Itzkovitz S
Molecular systems biology 2020 Dec;16(12):e9682
Molecular systems biology 2020 Dec;16(12):e9682
Temporal and spatial heterogeneity of host response to SARS-CoV-2 pulmonary infection.
Desai N, Neyaz A, Szabolcs A, Shih AR, Chen JH, Thapar V, Nieman LT, Solovyov A, Mehta A, Lieb DJ, Kulkarni AS, Jaicks C, Xu KH, Raabe MJ, Pinto CJ, Juric D, Chebib I, Colvin RB, Kim AY, Monroe R, Warren SE, Danaher P, Reeves JW, Gong J, Rueckert EH, Greenbaum BD, Hacohen N, Lagana SM, Rivera MN, Sholl LM, Stone JR, Ting DT, Deshpande V
Nature communications 2020 Dec 9;11(1):6319
Nature communications 2020 Dec 9;11(1):6319
Epithelial/Mesenchymal Characteristics and PD-L1 Co-Expression in CTCs of Metastatic Breast Cancer Patients Treated with Eribulin: Correlation with Clinical Outcome.
Polioudaki H, Mala A, Gkimprixi E, Papadaki MA, Chantziou A, Tzardi M, Mavroudis D, Agelaki S, Theodoropoulos PA
Cancers 2020 Dec 11;12(12)
Cancers 2020 Dec 11;12(12)
Temporal and Spatial Heterogeneity of Host Response to SARS-CoV-2 Pulmonary Infection.
Desai N, Neyaz A, Szabolcs A, Shih AR, Chen JH, Thapar V, Nieman LT, Solovyov A, Mehta A, Lieb DJ, Kulkarni AS, Jaicks C, Pinto CJ, Juric D, Chebib I, Colvin RB, Kim AY, Monroe R, Warren SE, Danaher P, Reeves JW, Gong J, Rueckert EH, Greenbaum BD, Hacohen N, Lagana SM, Rivera MN, Sholl LM, Stone JR, Ting DT, Deshpande V
medRxiv : the preprint server for health sciences 2020 Aug 2;
medRxiv : the preprint server for health sciences 2020 Aug 2;
Loss of the Krüppel-like factor 4 tumor suppressor is associated with epithelial-mesenchymal transition in colorectal cancer.
Agbo KC, Huang JZ, Ghaleb AM, Williams JL, Shroyer KR, Bialkowska AB, Yang VW
Journal of cancer metastasis and treatment 2019;5
Journal of cancer metastasis and treatment 2019;5
Highly multiplexed immunofluorescence images and single-cell data of immune markers in tonsil and lung cancer.
Rashid R, Gaglia G, Chen YA, Lin JR, Du Z, Maliga Z, Schapiro D, Yapp C, Muhlich J, Sokolov A, Sorger P, Santagata S
Scientific data 2019 Dec 17;6(1):323
Scientific data 2019 Dec 17;6(1):323
Screening Circulating Tumor Cells as a Noninvasive Cancer Test in 3388 Individuals from High-Risk Groups (ICELLATE2).
Castro J, Sanchez L, Nuñez MT, Lu M, Castro T, Sharifi HR, Ericsson C
Disease markers 2018;2018:4653109
Disease markers 2018;2018:4653109
Cell signaling heterogeneity is modulated by both cell-intrinsic and -extrinsic mechanisms: An integrated approach to understanding targeted therapy.
Kim E, Kim JY, Smith MA, Haura EB, Anderson ARA
PLoS biology 2018 Mar;16(3):e2002930
PLoS biology 2018 Mar;16(3):e2002930
Betamethasone prevents human rhinovirus- and cigarette smoke- induced loss of respiratory epithelial barrier function.
Waltl EE, Selb R, Eckl-Dorna J, Mueller CA, Cabauatan CR, Eiwegger T, Resch-Marat Y, Niespodziana K, Vrtala S, Valenta R, Niederberger V
Scientific reports 2018 Jun 26;8(1):9688
Scientific reports 2018 Jun 26;8(1):9688
The VAR2CSA malaria protein efficiently retrieves circulating tumor cells in an EpCAM-independent manner.
Agerbæk MØ, Bang-Christensen SR, Yang MH, Clausen TM, Pereira MA, Sharma S, Ditlev SB, Nielsen MA, Choudhary S, Gustavsson T, Sorensen PH, Meyer T, Propper D, Shamash J, Theander TG, Aicher A, Daugaard M, Heeschen C, Salanti A
Nature communications 2018 Aug 16;9(1):3279
Nature communications 2018 Aug 16;9(1):3279
Workflow optimization of whole genome amplification and targeted panel sequencing for CTC mutation detection.
Liu HE, Triboulet M, Zia A, Vuppalapaty M, Kidess-Sigal E, Coller J, Natu VS, Shokoohi V, Che J, Renier C, Chan NH, Hanft VR, Jeffrey SS, Sollier-Christen E
NPJ genomic medicine 2017;2:34
NPJ genomic medicine 2017;2:34
Neither epithelial nor mesenchymal circulating tumor cells isolated from breast cancer patients are tumorigenic in NOD-scid Il2rg(null) mice.
Donnenberg VS, Huber A, Basse P, Rubin JP, Donnenberg AD
NPJ breast cancer 2016;2:16004
NPJ breast cancer 2016;2:16004
Modified Leukocyte Filter Removes Tumor Cells from the Salvaged Blood.
Mei K, Du L, Yan M, Zhang Z, Zhang F, Gong L, Sun K, Zhang J, Tang Y, Jiang C, Liu J
PloS one 2015;10(6):e0130864
PloS one 2015;10(6):e0130864
Mutagenic, surviving and tumorigenic effects of follicular fluid in the context of p53 loss: initiation of fimbria carcinogenesis.
Huang HS, Chu SC, Hsu CF, Chen PC, Ding DC, Chang MY, Chu TY
Carcinogenesis 2015 Nov;36(11):1419-28
Carcinogenesis 2015 Nov;36(11):1419-28
Whole-body tissue stabilization and selective extractions via tissue-hydrogel hybrids for high-resolution intact circuit mapping and phenotyping.
Treweek JB, Chan KY, Flytzanis NC, Yang B, Deverman BE, Greenbaum A, Lignell A, Xiao C, Cai L, Ladinsky MS, Bjorkman PJ, Fowlkes CC, Gradinaru V
Nature protocols 2015 Nov;10(11):1860-1896
Nature protocols 2015 Nov;10(11):1860-1896
A breast cancer stem cell niche supported by juxtacrine signalling from monocytes and macrophages.
Lu H, Clauser KR, Tam WL, Fröse J, Ye X, Eaton EN, Reinhardt F, Donnenberg VS, Bhargava R, Carr SA, Weinberg RA
Nature cell biology 2014 Nov;16(11):1105-17
Nature cell biology 2014 Nov;16(11):1105-17
In vitro validation of an ultra-sensitive scanning fluorescence microscope for analysis of circulating tumor cells.
Hillig T, Nygaard AB, Nekiunaite L, Klingelhöfer J, Sölétormos G
APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 2014 Jun;122(6):545-51
APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 2014 Jun;122(6):545-51
Tyrosine phosphoproteomics identifies both codrivers and cotargeting strategies for T790M-related EGFR-TKI resistance in non-small cell lung cancer.
Yoshida T, Zhang G, Smith MA, Lopez AS, Bai Y, Li J, Fang B, Koomen J, Rawal B, Fisher KJ, Chen YA, Kitano M, Morita Y, Yamaguchi H, Shibata K, Okabe T, Okamoto I, Nakagawa K, Haura EB
Clinical cancer research : an official journal of the American Association for Cancer Research 2014 Aug 1;20(15):4059-4074
Clinical cancer research : an official journal of the American Association for Cancer Research 2014 Aug 1;20(15):4059-4074
Size-based isolation of circulating tumor cells in lung cancer patients using a microcavity array system.
Hosokawa M, Kenmotsu H, Koh Y, Yoshino T, Yoshikawa T, Naito T, Takahashi T, Murakami H, Nakamura Y, Tsuya A, Shukuya T, Ono A, Akamatsu H, Watanabe R, Ono S, Mori K, Kanbara H, Yamaguchi K, Tanaka T, Matsunaga T, Yamamoto N
PloS one 2013;8(6):e67466
PloS one 2013;8(6):e67466
Isolation of rare tumor cells from blood cells with buoyant immuno-microbubbles.
Shi G, Cui W, Benchimol M, Liu YT, Mattrey RF, Mukthavaram R, Kesari S, Esener SC, Simberg D
PloS one 2013;8(3):e58017
PloS one 2013;8(3):e58017
Flow cytometric determination of stem/progenitor content in epithelial tissues: an example from nonsmall lung cancer and normal lung.
Donnenberg VS, Landreneau RJ, Pfeifer ME, Donnenberg AD
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2013 Jan;83(1):141-9
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2013 Jan;83(1):141-9
Binding and isolation of tumor cells in biological media with perfluorocarbon microbubbles.
Shi G, Cui W, Mukthavaram R, Liu YT, Simberg D
Methods (San Diego, Calif.) 2013 Dec 1;64(2):102-7
Methods (San Diego, Calif.) 2013 Dec 1;64(2):102-7
KIT (CD117) expression in a subset of non-small cell lung carcinoma (NSCLC) patients.
Donnenberg AD, Zimmerlin L, Landreneau RJ, Luketich JD, Donnenberg VS
PloS one 2012;7(12):e52885
PloS one 2012;7(12):e52885
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
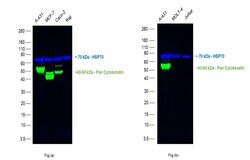
- Multiplexed fluorescent western blot was performed using pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82). Whole cell extracts of A-431 (Lane 1), MCF-7 (Lane 2), Caco-2 (Lane 3), Raji (Lane 4) as seen in Fig (a), A-431 (Lane 1), MOLT-4 (Lane 2) and Jurkat (Lane 3) as seen in Fig (b) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP03222BOX). Resolved proteins were transferred onto a Low-Fluorescence PVDF Transfer Membrane (Product # 22860) using iBlot® 2 Dry Blotting System (Product # IB21001) and blocked with Blocker™ FL Fluorescent Blocking Buffer (10X) (Product # 37565). The blots were probed with Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488 (Product # 53-9003-82, 1 µg/mL) and HSP70 Polyclonal Antibody (Product # PA5-28003, 1:4000 dilution). Secondary antibody Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ Plus 800 (Product # A32808, 1:10000 dilution) was used for detection of HSP70. Fluorescent detection was performed using iBright FL1500 (Product # A44115).
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
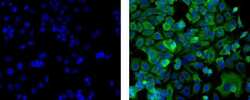
- Immunocytochemistry of fixed and permeabilized MCF-7 cells using 1 µg/mL of Mouse IgG1 K Isotype Control Alexa Fluor® 488 (left) or 1 µg/mL of Anti-Pan Cytokeratin (AE1/AE3) Alexa Fluor® 488 (right). Nuclei are stained with DAPI.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
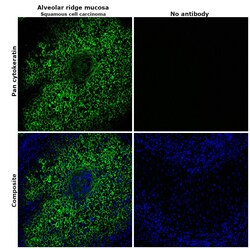
- Immunohistochemical analysis of pan cytokeratin was performed using formalin-fixed paraffin-embedded alveolar ridge mucosa (squamous cell carcinoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
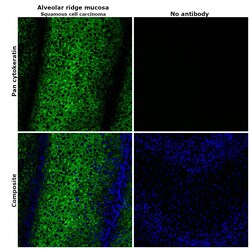
- Immunohistochemical analysis of pan cytokeratin was performed using formalin-fixed paraffin-embedded alveolar ridge mucosa (squamous cell carcinoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
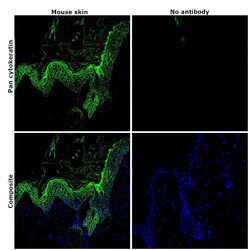
- Experimental details
- Immunohistochemical analysis of pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave oven at 110 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
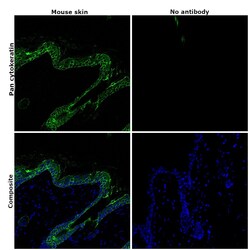
- Experimental details
- Immunohistochemical analysis of pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave oven at 110 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
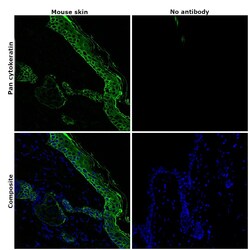
- Experimental details
- Immunohistochemical analysis of pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave oven at 110 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave oven at 110 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
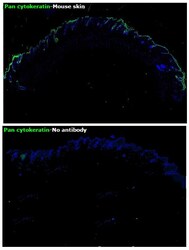
- Experimental details
- Immunohistochemical analysis of pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a microwave oven at 110 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), Alexa Fluor 488, eBioscience™ (Product # 53-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
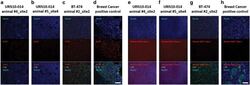
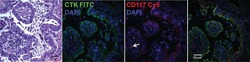
- Experimental details
- Figure 2 Immunofluorescent staining for human-specific Ki-67, human-specific cytokeratin, and murine major histocompatibility complex (MHC) Class I. Nuclei were stained with DAPI. Observed neoplasms in the URN10-014 group were negative for human Ki-67+ and human-specific cytokeratin (columns a , b ), but stained positively for murine-specific MHC Class I ( e , f ). A BT474 xenograft ( c , g ) and a human metastatic breast cancer control ( d , h ) were positive for human cytokeratin and Ki-67, but negative for murine MHC Class I antigen. Scale bar (white)=100 mum.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
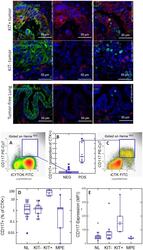
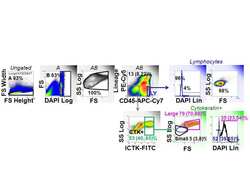
- Experimental details
- Figure 1 CD117 expression in normal lung (NL) and NSCLC. Photomicrographs: Expression of CD117 and Ki-67 in NSC lung cancer and normal lung. The left columns show sections stained with CD117 (red), cytokeratin (green) and DAPI (blue). Sections in the center column show CD117 (red) and DAPI (blue) only, in order to reveal CD117 staining obscured by bright cytokeratin expression. Sections in the right column shows CD117 (red), the proliferation marker Ki67 (green) and DAPI (blue). Tumors were classified as KIT+ or negative on the basis of CD117 immunofluorescent staining of FFPE. In KIT+ tumors (top photomicrographs) CD117 (red stain, center and right panels) was expressed in virtually all cytokeratin+ tumor cells (green stain, left panels). Ki-67+ proliferating cells (green stain, right panels) were frequently seen among CD117+ tumor cells. In KIT negative tumors (center row of photomicrographs), only solitary CD117+ mast cells were detected (red stain, center and right panels). Proliferating Ki-67+ cells were frequent among cytokeratin+ CD117 negative tumor cells. Normal tumor-adjacent lung also appeared to lack CD117 expression among cytokeratin+ airway cells (bottom photomicrographs). Proliferating Ki-67+ cells were infrequent and confined to the basal layer of airway epithelium. When all NSCLC tumors are considered together, flow cytometry revealed bimodal CD117 expression (center panels A-C). Cells were gated on hematopoietic lineage negative singlet events with DNA conte
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
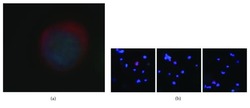
- Figure 1 (a) A circulating tumor cell prepared from a 7.5 ml blood sample from a 79-year-old male with no previous history of cancer. The cell is stained for cytokeratin (red) and for the cell nucleus (blue), typical of epithelial cells. Epithelial cells should not normally be present in the blood. The cell was negative for CD45, that is, not an immune cell. The cell nucleus has a large size typical of transcriptionally active cells, such as cancer cells, and the rounded shape of a cell in suspension, rather than the angular shape and cell sheath context of a normal solid tissue epithelium cell. (b) Three additional examples of circulating tumor cells stained for cytokeratin (red) and for the cell nucleus (blue). The lower magnification also shows the residual leucocytes surrounding the circulating tumor cells (blue nuclei, with no cytokeratin (red). The samples were enriched about 7500-fold for CTCs, with about 10,000 DAPI and CD45-positive leucocytes left in the sample after enrichment.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
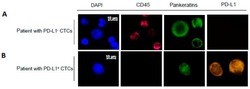
- Figure 2 Representative images of CTCs detected and subjected to immunostaining with DAPI, CD45, Pankeratins and PD-L1. Example images of CTCs from a patient with PD-L1 - CTCs ( A ) and PD-L1 + CTCs ( B ) are shown. The scale bar of 10 mum was applied to all pictures. ( C ) Number of CTCs and PD-L1 status isolated from blood samples from 38 patients (P1 through P38) with detectable PD-L1 status. ""P1"" stands for patient 1. Red bar represents the number of CK(+)/PD-L1(+) CTCs. Blue bar represents the number of CK(+)/PD-L1(-) CTCs.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
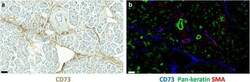
- Fig. 1 CD73 expression in normal pancreas. a Immunoperoxidase staining of normal pancreas for CD73 (brown). b Multicolour immunofluorescence staining of a consecutive section of normal pancreas for CD73 (blue), pan-cytokeratin (green) and alpha-smooth muscle actin (red). Bars, 50 mum
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
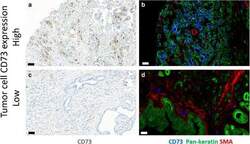
- Fig. 2 CD73 expression in adenocarcinoma of pancreas. Representative immunoperoxidase stainings (a and c) for CD73 (brown) and multicolour immunofluorescence stainings (b, d) for CD73 (blue), pan-cytokeratin (green) and alpha-smooth muscle actin (red). Bars, 50 mum
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
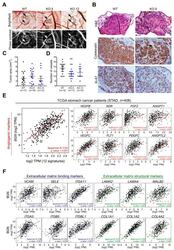
- Figure 6 Effect of biglycan in in vivo angiogenesis. ( A ) Representative images of tumors formed in the in vivo CAM by MKN74 cell models (WT and biglycan KO clones-KO.5 and KO.12). Phase contrast was used to better visualize tumor foci and vessels. MKN74 form multiple tumor foci (arrows/circles). Magnification at 20x. ( B ) Histological images of the formed tumors, cytokeratin staining confirming the presence of human epithelial tumors in the CAM. Ki-67 expression analysis was used to assess tumor aggressiveness. Histologically, tumors formed by KO cells present a less cohesive-like tumor mass with increased extracellular matrix stiffness. Hematoxilin & Eosin (H&E) staining images at 100X magnification, cytokeration and Ki-67 images at 200x magnification. ( C ) Quantification of the tumor area (mm 2 ) with no significant differences in the tumors being derived from the different cell lines. ( D ) Number of new vessels (less than 20 mum in diameter) formed towards the inoculation site. biglycan KO inoculated cells showed less capacity to form new vessels when compared to WT biglycan-positive tumors. ( E ) In silico gene analysis in GC tissues samples (TCGA, n = 408) showing that BGN was strongly positively correlated with angiogenic markers ( VEGFB, VEGFC, KDR, FLT1, FGF2, PDGFC, ANGPT1, ANGPT2, ANGPTL2, ANGPTL1, ANGPTL4, and ANGPTL7 ) . ( F ) In silico analysis demonstrating that BGN is positively correlated with ECM binding ( VCAM1, SELE, ITGA11, ITGA5, ITGB5, ITGBL1 ) and
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
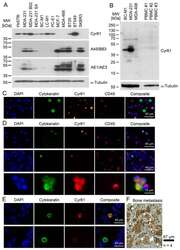
- Figure 5 Cyr61 detection in breast cancer cells. ( A ) Comparison of the cytoplasmic Cyr61 levels with cytokeratin levels, as analysed by the pan-cytokeratin antibody cocktails A45/BB3 and AE1/AE3 by Western blot analysis. ( B ) A comparison of the Cyr61 levels in the peripheral blood mononuclear cells (PBMC) of healthy women with the Cyr61 levels in breast cancer cell lines. ( C ) Cyr61 detection in BC-M1 and MDA-MB-468 spiked into blood samples from healthy women by immunocytochemical double staining. ( D ) Cyr61 detection in CTC from the peripheral blood of breast cancer patients (details: Table 1 ). ( E ) Detection of Cyr61 in the DTC from the bone marrow of breast cancer patients. The upper row shows a Cyr61-positive DTC, and the bottom row shows a Cyr61-negative DTC. ( F ) An immunohistochemical Cyr61 detection in the bone metastases of breast cancer patients. ( C - E ) The composite images are overlays of the Cytokeratin, Cyr61, Dapi and CD45 (if applied) signals, n biol : 3 ( A , C , E ), 4 ( F ).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
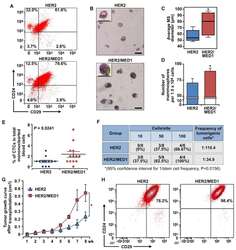
- Figure 4. MED1 overexpression promotes MMTV-HER2 CSC formation (A) FACS analyses of MMTV-HER2 and MMTV-HER2/MMTV-MED1 CSCs using antibodies against cell surface markers Lin, CD24, and CD29. (B) Mammosphere assays using FACS-sorted tumor cells in (A). Scale bar: 100 mum. (C and D) Average diameters (C) and numbers (D) of mammospheres formed in (B). (E) Statistics of flow cytometry analysis of CD45 - CK18 + EpCAM hi circulating tumor cells (CTCs) in mononuclear blood cells from MMTV-HER2 and MMTV-HER2/MMTV-MED1 tumor-bearing mice (n = 13). (F) Limiting dilution analyses of tumor-initiating cells in MMTV-HER2 and MMTV-HER2/MMTV-MED1 bulk tumors. (G) Growth curves of orthotopic MMTV-HER2 and MMTV-HER2/MMTV-MED1 tumor xenografts (n = 6). (H) FACS analyses of the grafted tumors using cell surface markers Lin (CD31, CD45, and Ter119), CD24, and CD29. The values are obtained from three independent experiments and shown as mean +- SD. *p < 0.05.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
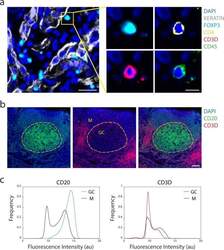
- Fig. 3 Antibody staining quality. ( a ) Immunofluorescence image from LUNG-3-PR showing epithelial tumor cells marked by Keratin (white) and a regulatory T cell marked by FOXP3 (cyan), CD4 (yellow), CD3D (red), and CD45 (green) (scale bar: 25 um; inset scale bar: 10 um). ( b ) A region of TONSIL-1 showing CD20 (green) and CD3D (red) expression. Area inside yellow dashed circle denotes germinal center (GC), and area outside denotes the mantle (M) region (scale bar: 100 um). ( c ) Probability density function of fluorescence signal intensity of every pixel in the germinal center (n = 1,446,450 pixels) and mantle (n = 4,369,358 pixels) for CD20 and CD3D within the region shown in ( b ). X-axis is fluorescence intensity (log2 au) and y-axis is frequency of pixels.
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot