H00023435-M01
antibody from Novus Biologicals
Targeting: TARDBP
ALS10, TDP-43
 Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Blocking/Neutralizing
Blocking/Neutralizing Functional assay
Functional assayAntibody data
- Antibody Data
- Antigen structure
- References [171]
- Comments [0]
- Validations
- Western blot [3]
- ELISA [1]
- Immunocytochemistry [1]
- Immunoprecipitation [1]
- Immunohistochemistry [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- H00023435-M01 - Provider product page

- Provider
- Novus Biologicals
- Proper citation
- Novus Cat#H00023435-M01, RRID:AB_548546
- Product name
- Mouse Monoclonal TDP-43/TARDBP Antibody
- Antibody type
- Monoclonal
- Description
- IgG purified. TARDBP - TAR DNA binding protein
- Reactivity
- Human, Mouse, Rat, Bacteria, Simian
- Host
- Mouse
- Isotype
- IgG
- Vial size
- 0.1 mg
- Storage
- Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles.
Submitted references Biomarkers for diseases with TDP-43 pathology.
Caspase-4 mediates cytoplasmic accumulation of TDP-43 in the primate brains.
Impaired Nucleoporins Are Present in Sporadic Amyotrophic Lateral Sclerosis Motor Neurons that Exhibit Mislocalization of the 43-kDa TAR DNA-Binding Protein.
TDP-43 enhances translation of specific mRNAs linked to neurodegenerative disease.
Presence of tau astrogliopathy in frontotemporal dementia caused by a novel Grn nonsense (Trp2*) mutation.
Biallelic expansion of an intronic repeat in RFC1 is a common cause of late-onset ataxia.
The First Historically Reported Italian Family with FTD/ALS Teaches a Lesson on C9orf72 RE: Clinical Heterogeneity and Oligogenic Inheritance.
mTh1 driven expression of hTDP-43 results in typical ALS/FTLD neuropathological symptoms.
Towards a TDP-43-Based Biomarker for ALS and FTLD.
TAR DNA-Binding Protein 43 and Disrupted in Schizophrenia 1 Coaggregation Disrupts Dendritic Local Translation and Mental Function in Frontotemporal Lobar Degeneration.
A feedback loop between dipeptide-repeat protein, TDP-43 and karyopherin-α mediates C9orf72-related neurodegeneration.
Age-related deregulation of TDP-43 after stroke enhances NF-κB-mediated inflammation and neuronal damage.
TDP-43 regulates the alternative splicing of hnRNP A1 to yield an aggregation-prone variant in amyotrophic lateral sclerosis.
Chronic Administration of Pimozide Fails to Attenuate Motor and Pathological Deficits in Two Mouse Models of Amyotrophic Lateral Sclerosis.
Gene Expression Profile in Frontal Cortex in Sporadic Frontotemporal Lobar Degeneration-TDP.
Alzheimer neuropathology without frontotemporal lobar degeneration hallmarks (TAR DNA-binding protein 43 inclusions) in missense progranulin mutation Cys139Arg.
Co-occurrence of mixed proteinopathies in late-stage Huntington's disease.
Cre-dependent AAV vectors for highly targeted expression of disease-related proteins and neurodegeneration in the substantia nigra.
Systematic Screening of Ubiquitin/p62 Aggregates in Cerebellar Cortex Expands the Neuropathological Phenotype of the C9orf72 Expansion Mutation.
The clinical, neuroanatomical, and neuropathologic phenotype of TBK1-associated frontotemporal dementia: A longitudinal case report.
Frontotemporal Dementia Caused by the P301L Mutation in the MAPT Gene: Clinicopathological Features of 13 Cases from the Same Geographical Origin in Barcelona, Spain.
Drosophila lines with mutant and wild type human TDP-43 replacing the endogenous gene reveals phosphorylation and ubiquitination in mutant lines in the absence of viability or lifespan defects.
The unexpected co-occurrence of GRN and MAPT p.A152T in Basque families: Clinical and pathological characteristics.
Clusterin protects neurons against intracellular proteotoxicity.
Dioxins and related environmental contaminants increase TDP-43 levels.
TBK1 Mutation Spectrum in an Extended European Patient Cohort with Frontotemporal Dementia and Amyotrophic Lateral Sclerosis.
Motor-Coordinative and Cognitive Dysfunction Caused by Mutant TDP-43 Could Be Reversed by Inhibiting Its Mitochondrial Localization.
Monocytes of patients with amyotrophic lateral sclerosis linked to gene mutations display altered TDP-43 subcellular distribution.
Incidental neuronal intermediate filament inclusion pathology: unexpected biopsy findings in a 37-year-old woman with epilepsy.
TDP-43 Promotes Neurodegeneration by Impairing Chromatin Remodeling.
Withania somnifera Reverses Transactive Response DNA Binding Protein 43 Proteinopathy in a Mouse Model of Amyotrophic Lateral Sclerosis/Frontotemporal Lobar Degeneration.
Neuropathological criteria of anti-IgLON5-related tauopathy.
Hereditary leukoencephalopathy with axonal spheroids: a spectrum of phenotypes from CNS vasculitis to parkinsonism in an adult onset leukodystrophy series.
Two mutations G335D and Q343R within the amyloidogenic core region of TDP-43 influence its aggregation and inclusion formation.
Increased cytoplasmic TARDBP mRNA in affected spinal motor neurons in ALS caused by abnormal autoregulation of TDP-43.
Developmentally Regulated RNA-binding Protein 1 (Drb1)/RNA-binding Motif Protein 45 (RBM45), a Nuclear-Cytoplasmic Trafficking Protein, Forms TAR DNA-binding Protein 43 (TDP-43)-mediated Cytoplasmic Aggregates.
Pisa syndrome in a patient with pathologically confirmed Parkinson's disease.
Altered Mitochondria, Protein Synthesis Machinery, and Purine Metabolism Are Molecular Contributors to the Pathogenesis of Creutzfeldt-Jakob Disease.
The inhibition of TDP-43 mitochondrial localization blocks its neuronal toxicity.
CCNF mutations in amyotrophic lateral sclerosis and frontotemporal dementia.
Early diagnosis of cardiac involvement in idiopathic inflammatory myopathy by cardiac magnetic resonance tomography.
Cytoplasmic mislocalization of RNA splicing factors and aberrant neuronal gene splicing in TDP-43 transgenic pig brain.
Low molecular weight species of TDP-43 generated by abnormal splicing form inclusions in amyotrophic lateral sclerosis and result in motor neuron death.
Neuropathological assessments of the pathology in frontotemporal lobar degeneration with TDP43-positive inclusions: an inter-laboratory study by the BrainNet Europe consortium.
TDP-43 as a possible biomarker for frontotemporal lobar degeneration: a systematic review of existing antibodies.
Casein kinase II induced polymerization of soluble TDP-43 into filaments is inhibited by heat shock proteins.
A 6.4 Mb duplication of the α-synuclein locus causing frontotemporal dementia and Parkinsonism: phenotype-genotype correlations.
Parkin-mediated reduction of nuclear and soluble TDP-43 reverses behavioral decline in symptomatic mice.
Abnormal serine phosphorylation of insulin receptor substrate 1 is associated with tau pathology in Alzheimer's disease and tauopathies.
TDP-43 causes differential pathology in neuronal versus glial cells in the mouse brain.
Plasma phosphorylated TDP-43 levels are elevated in patients with frontotemporal dementia carrying a C9orf72 repeat expansion or a GRN mutation.
A pathogenic progranulin mutation and C9orf72 repeat expansion in a family with frontotemporal dementia.
Widespread RNA metabolism impairment in sporadic inclusion body myositis TDP43-proteinopathy.
Multiple organ involvement by alpha-synuclein pathology in Lewy body disorders.
UBE2E ubiquitin-conjugating enzymes and ubiquitin isopeptidase Y regulate TDP-43 protein ubiquitination.
Accumulation of C-terminal fragments of transactive response DNA-binding protein 43 leads to synaptic loss and cognitive deficits in human TDP-43 transgenic mice.
Cell injury and premature neurodegeneration in focal malformations of cortical development.
The novel MAPT mutation K298E: mechanisms of mutant tau toxicity, brain pathology and tau expression in induced fibroblast-derived neurons.
Disease-associated mutations of TDP-43 promote turnover of the protein through the proteasomal pathway.
No interaction between tau and TDP-43 pathologies in either frontotemporal lobar degeneration or motor neurone disease.
TDP-1, the Caenorhabditis elegans ortholog of TDP-43, limits the accumulation of double-stranded RNA.
Clinical and neuropathological variability in clinically isolated central nervous system Whipple's disease.
Reduced cellular Ca(2+) availability enhances TDP-43 cleavage by apoptotic caspases.
Parkin reverses TDP-43-induced cell death and failure of amino acid homeostasis.
Interaction of transactive response DNA binding protein 43 with nuclear factor κB in mild cognitive impairment with episodic memory deficits.
The pathological phenotypes of human TDP-43 transgenic mouse models are independent of downregulation of mouse Tdp-43.
ALS-associated TDP-43 induces endoplasmic reticulum stress, which drives cytoplasmic TDP-43 accumulation and stress granule formation.
Reduced cholinergic olfactory centrifugal inputs in patients with neurodegenerative disorders and MPTP-treated monkeys.
Neurodegenerative disease status and post-mortem pathology in idiopathic rapid-eye-movement sleep behaviour disorder: an observational cohort study.
MAPT H1 haplotype is associated with enhanced α-synuclein deposition in dementia with Lewy bodies.
Characterization of thorn-shaped astrocytes in white matter of temporal lobe in Alzheimer's disease brains.
Reactive astrocytes secrete lcn2 to promote neuron death.
Primary progressive aphasia with parkinsonism.
Asymptomatic hyper-creatine-kinase-emia as sole manifestation of inclusion body myositis.
Selective forelimb impairment in rats expressing a pathological TDP-43 25 kDa C-terminal fragment to mimic amyotrophic lateral sclerosis.
Globular glial-like inclusions in a patient with advanced Alzheimer's disease.
The long non-coding RNA nuclear-enriched abundant transcript 1_2 induces paraspeckle formation in the motor neuron during the early phase of amyotrophic lateral sclerosis.
Expression of ALS-linked TDP-43 mutant in astrocytes causes non-cell-autonomous motor neuron death in rats.
Parkin ubiquitinates Tar-DNA binding protein-43 (TDP-43) and promotes its cytosolic accumulation via interaction with histone deacetylase 6 (HDAC6).
Accelerated disease onset with stabilized familial amyotrophic lateral sclerosis (ALS)-linked mutant TDP-43 proteins.
Prominent psychiatric symptoms in patients with Parkinson's disease and concomitant argyrophilic grain disease.
The involvement of the cerebellum in amyotrophic lateral sclerosis.
Atypical neuropathological sCJD-MM phenotype with abundant white matter Kuru-type plaques sparing the cerebellar cortex.
Co-occurrence of different pathologies in dementia: implications for dementia diagnosis.
Breakpoint sequence analysis of an AβPP locus duplication associated with autosomal dominant Alzheimer's disease and severe cerebral amyloid angiopathy.
TDP-43 plasma levels are higher in amyotrophic lateral sclerosis.
An MND/ALS phenotype associated with C9orf72 repeat expansion: abundant p62-positive, TDP-43-negative inclusions in cerebral cortex, hippocampus and cerebellum but without associated cognitive decline.
Matrix metalloproteinase-9 expression in the nuclear compartment of neurons and glial cells in aging and stroke.
XBP1 depletion precedes ubiquitin aggregation and Golgi fragmentation in TDP-43 transgenic rats.
Wild type TDP-43 induces neuro-inflammation and alters APP metabolism in lentiviral gene transfer models.
TDP-43 regulates global translational yield by splicing of exon junction complex component SKAR.
Identification of c-myc-dependent proteins in the medulloblastoma cell line D425Med.
Mutant TDP-43 in motor neurons promotes the onset and progression of ALS in rats.
Epitope mapping of antibodies against TDP-43 and detection of protease-resistant fragments of pathological TDP-43 in amyotrophic lateral sclerosis and frontotemporal lobar degeneration.
Unusual clinical presentation and neuropathology in two subjects with fused-in sarcoma (FUS) positive inclusions.
Frontotemporal lobar degeneration-related proteins induce only subtle memory-related deficits when bilaterally overexpressed in the dorsal hippocampus.
Pick's pathology in Parkinson's disease with dementia.
Abnormal regenerative responses and impaired axonal outgrowth after nerve crush in TDP-43 transgenic mouse models of amyotrophic lateral sclerosis.
Hirano body-rich subtypes of Creutzfeldt-Jakob disease.
Pathological hallmarks of amyotrophic lateral sclerosis/frontotemporal lobar degeneration in transgenic mice produced with TDP-43 genomic fragments.
Neurofibrillary tangle pathology and Braak staging in chronic epilepsy in relation to traumatic brain injury and hippocampal sclerosis: a post-mortem study.
The ALS-associated proteins FUS and TDP-43 function together to affect Drosophila locomotion and life span.
Cytoplasmic accumulation of TDP-43 in circulating lymphomonocytes of ALS patients with and without TARDBP mutations.
TAR-DNA binding protein-43 and alterations in the hippocampus.
FTLD-TDP with motor neuron disease, visuospatial impairment and a progressive supranuclear palsy-like syndrome: broadening the clinical phenotype of TDP-43 proteinopathies. A report of three cases.
Neocortical layer formation of human developing brains and lissencephalies: consideration of layer-specific marker expression.
Elevated CSF TDP-43 levels in amyotrophic lateral sclerosis: specificity, sensitivity, and a possible prognostic value.
TDP-43 potentiates alpha-synuclein toxicity to dopaminergic neurons in transgenic mice.
TDP-43 potentiates alpha-synuclein toxicity to dopaminergic neurons in transgenic mice.
Aggregation of the 35-kDa fragment of TDP-43 causes formation of cytoplasmic inclusions and alteration of RNA processing.
Increased dopaminergic cells and protein aggregates in the olfactory bulb of patients with neurodegenerative disorders.
Distribution and pattern of pathology in subjects with familial or sporadic late-onset cerebellar ataxia as assessed by p62/sequestosome immunohistochemistry.
Molecular mechanisms of MLC1 and GLIALCAM mutations in megalencephalic leukoencephalopathy with subcortical cysts.
TDP-43 pathology may occur in the BRI2 gene-related dementias.
β-amyloid triggers ALS-associated TDP-43 pathology in AD models.
The first case of protease-sensitive prionopathy (PSPr) in The Netherlands: a patient with an unusual GSS-like clinical phenotype.
Altered distributions of Gemini of coiled bodies and mitochondria in motor neurons of TDP-43 transgenic mice.
Tubulin polymerization promoting protein (TPPP/p25) as a marker for oligodendroglial changes in multiple sclerosis.
Depletion of oxidative and endoplasmic reticulum stress regulators in Pick disease.
Mutations of optineurin in amyotrophic lateral sclerosis.
Amyotrophic lateral sclerosis: dash-like accumulation of phosphorylated TDP-43 in somatodendritic and axonal compartments of somatomotor neurons of the lower brainstem and spinal cord.
ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS.
Knockdown of transactive response DNA-binding protein (TDP-43) downregulates histone deacetylase 6.
Characterization of alternative isoforms and inclusion body of the TAR DNA-binding protein-43.
TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration.
TDP-43 physically interacts with amyotrophic lateral sclerosis-linked mutant CuZn superoxide dismutase.
Expansive gene transfer in the rat CNS rapidly produces amyotrophic lateral sclerosis relevant sequelae when TDP-43 is overexpressed.
Neurotoxic effects of TDP-43 overexpression in C. elegans.
Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice.
Alteration of biochemical and pathological properties of TDP-43 protein by a lipid mediator, 15-deoxy-Delta(12,14)-prostaglandin J(2).
Plasma phosphorylated-TDP-43 protein levels correlate with brain pathology in frontotemporal lobar degeneration.
Cytosolic TDP-43 expression following axotomy is associated with caspase 3 activation in NFL-/- mice: support for a role for TDP-43 in the physiological response to neuronal injury.
The complement factor C5a receptor is upregulated in NFL-/- mouse motor neurons.
VCP mutations causing frontotemporal lobar degeneration disrupt localization of TDP-43 and induce cell death.
TAR DNA-Binding protein 43 accumulation in protein aggregate myopathies.
Expression of TDP-43 C-terminal Fragments in Vitro Recapitulates Pathological Features of TDP-43 Proteinopathies.
Early-onset familial lewy body dementia with extensive tauopathy: a clinical, genetic, and neuropathological study.
Increased TDP-43 protein in cerebrospinal fluid of patients with amyotrophic lateral sclerosis.
Sporadic amyotrophic lateral sclerosis of long duration is associated with relatively mild TDP-43 pathology.
Expression of collagen XVII and ubiquitin-binding protein p62 in motor neuron disease.
Divergent patterns of cytosolic TDP-43 and neuronal progranulin expression following axotomy: implications for TDP-43 in the physiological response to neuronal injury.
Frontotemporal dementia in a large Swedish family is caused by a progranulin null mutation.
Abnormal hippocampal distribution of TDP-43 in patients with-late onset psychosis.
Ubiquitin associated protein 1 is a risk factor for frontotemporal lobar degeneration.
Frontotemporal dementia presenting as schizophrenia-like psychosis in young people: clinicopathological series and review of cases.
TDP-43 accumulation in inclusion body myopathy muscle suggests a common pathogenic mechanism with frontotemporal dementia.
White matter tauopathy with globular glial inclusions: a distinct sporadic frontotemporal lobar degeneration.
TDP-43 in cerebrospinal fluid of patients with frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation.
A distinct clinical, neuropsychological and radiological phenotype is associated with progranulin gene mutations in a large UK series.
Epitope mapping of 2E2-D3, a monoclonal antibody directed against human TDP-43.
Molecular pathogenesis of frontotemporal lobar degeneration: basic science seminar in neurology.
Enrichment of C-terminal fragments in TAR DNA-binding protein-43 cytoplasmic inclusions in brain but not in spinal cord of frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
MAPT S305I mutation: implications for argyrophilic grain disease.
TDP-43-immunoreactive neuronal and glial inclusions in the neostriatum in amyotrophic lateral sclerosis with and without dementia.
Missense mutations in the progranulin gene linked to frontotemporal lobar degeneration with ubiquitin-immunoreactive inclusions reduce progranulin production and secretion.
Colocalization of transactivation-responsive DNA-binding protein 43 and huntingtin in inclusions of Huntington disease.
Enduring involvement of tau, beta-amyloid, alpha-synuclein, ubiquitin and TDP-43 pathology in the amyotrophic lateral sclerosis/parkinsonism-dementia complex of Guam (ALS/PDC).
Ultrastructural localization of TDP-43 in filamentous neuronal inclusions in various neurodegenerative diseases.
Maturation process of TDP-43-positive neuronal cytoplasmic inclusions in amyotrophic lateral sclerosis with and without dementia.
TDP-43 protein in plasma may index TDP-43 brain pathology in Alzheimer's disease and frontotemporal lobar degeneration.
A yeast TDP-43 proteinopathy model: Exploring the molecular determinants of TDP-43 aggregation and cellular toxicity.
DEAD-box RNA helicase subunits of the Drosha complex are required for processing of rRNA and a subset of microRNAs.
TDP-43 immunoreactivity in hippocampal sclerosis and Alzheimer's disease.
Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations.
TDP-43 is deposited in the Guam parkinsonism-dementia complex brains.
TDP-43 immunoreactivity in neuronal inclusions in familial amyotrophic lateral sclerosis with or without SOD1 gene mutation.
TDP-43-positive white matter pathology in frontotemporal lobar degeneration with ubiquitin-positive inclusions.
TDP-43 in the ubiquitin pathology of frontotemporal dementia with VCP gene mutations.
Evidence that TDP-43 is not the major ubiquitinated target within the pathological inclusions of amyotrophic lateral sclerosis.
TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
Steinacker P, Barschke P, Otto M
Molecular and cellular neurosciences 2019 Jun;97:43-59
Molecular and cellular neurosciences 2019 Jun;97:43-59
Caspase-4 mediates cytoplasmic accumulation of TDP-43 in the primate brains.
Yin P, Guo X, Yang W, Yan S, Yang S, Zhao T, Sun Q, Liu Y, Li S, Li XJ
Acta neuropathologica 2019 Jun;137(6):919-937
Acta neuropathologica 2019 Jun;137(6):919-937
Impaired Nucleoporins Are Present in Sporadic Amyotrophic Lateral Sclerosis Motor Neurons that Exhibit Mislocalization of the 43-kDa TAR DNA-Binding Protein.
Aizawa H, Yamashita T, Kato H, Kimura T, Kwak S
Journal of clinical neurology (Seoul, Korea) 2019 Jan;15(1):62-67
Journal of clinical neurology (Seoul, Korea) 2019 Jan;15(1):62-67
TDP-43 enhances translation of specific mRNAs linked to neurodegenerative disease.
Neelagandan N, Gonnella G, Dang S, Janiesch PC, Miller KK, Küchler K, Marques RF, Indenbirken D, Alawi M, Grundhoff A, Kurtz S, Duncan KE
Nucleic acids research 2019 Jan 10;47(1):341-361
Nucleic acids research 2019 Jan 10;47(1):341-361
Presence of tau astrogliopathy in frontotemporal dementia caused by a novel Grn nonsense (Trp2*) mutation.
Gómez-Tortosa E, Baradaran-Heravi Y, González Alvarez V, Sainz MJ, Prieto-Jurczynska C, Guerrero-López R, Agüero Rabes P, Van Broeckhoven C, van der Zee J, Rábano Gutiérrez A, EU EOD Consortium.
Neurobiology of aging 2019 Apr;76:214.e11-214.e15
Neurobiology of aging 2019 Apr;76:214.e11-214.e15
Biallelic expansion of an intronic repeat in RFC1 is a common cause of late-onset ataxia.
Cortese A, Simone R, Sullivan R, Vandrovcova J, Tariq H, Yau WY, Humphrey J, Jaunmuktane Z, Sivakumar P, Polke J, Ilyas M, Tribollet E, Tomaselli PJ, Devigili G, Callegari I, Versino M, Salpietro V, Efthymiou S, Kaski D, Wood NW, Andrade NS, Buglo E, Rebelo A, Rossor AM, Bronstein A, Fratta P, Marques WJ, Züchner S, Reilly MM, Houlden H
Nature genetics 2019 Apr;51(4):649-658
Nature genetics 2019 Apr;51(4):649-658
The First Historically Reported Italian Family with FTD/ALS Teaches a Lesson on C9orf72 RE: Clinical Heterogeneity and Oligogenic Inheritance.
Giannoccaro MP, Bartoletti-Stella A, Piras S, Casalena A, Oppi F, Ambrosetto G, Montagna P, Liguori R, Parchi P, Capellari S
Journal of Alzheimer's disease : JAD 2018;62(2):687-697
Journal of Alzheimer's disease : JAD 2018;62(2):687-697
mTh1 driven expression of hTDP-43 results in typical ALS/FTLD neuropathological symptoms.
Scherz B, Rabl R, Flunkert S, Rohler S, Neddens J, Taub N, Temmel M, Panzenboeck U, Niederkofler V, Zimmermann R, Hutter-Paier B
PloS one 2018;13(5):e0197674
PloS one 2018;13(5):e0197674
Towards a TDP-43-Based Biomarker for ALS and FTLD.
Feneberg E, Gray E, Ansorge O, Talbot K, Turner MR
Molecular neurobiology 2018 Oct;55(10):7789-7801
Molecular neurobiology 2018 Oct;55(10):7789-7801
TAR DNA-Binding Protein 43 and Disrupted in Schizophrenia 1 Coaggregation Disrupts Dendritic Local Translation and Mental Function in Frontotemporal Lobar Degeneration.
Endo R, Takashima N, Nekooki-Machida Y, Komi Y, Hui KK, Takao M, Akatsu H, Murayama S, Sawa A, Tanaka M
Biological psychiatry 2018 Oct 1;84(7):509-521
Biological psychiatry 2018 Oct 1;84(7):509-521
A feedback loop between dipeptide-repeat protein, TDP-43 and karyopherin-α mediates C9orf72-related neurodegeneration.
Solomon DA, Stepto A, Au WH, Adachi Y, Diaper DC, Hall R, Rekhi A, Boudi A, Tziortzouda P, Lee YB, Smith B, Bridi JC, Spinelli G, Dearlove J, Humphrey DM, Gallo JM, Troakes C, Fanto M, Soller M, Rogelj B, Parsons RB, Shaw CE, Hortobágyi T, Hirth F
Brain : a journal of neurology 2018 Oct 1;141(10):2908-2924
Brain : a journal of neurology 2018 Oct 1;141(10):2908-2924
Age-related deregulation of TDP-43 after stroke enhances NF-κB-mediated inflammation and neuronal damage.
Thammisetty SS, Pedragosa J, Weng YC, Calon F, Planas A, Kriz J
Journal of neuroinflammation 2018 Nov 9;15(1):312
Journal of neuroinflammation 2018 Nov 9;15(1):312
TDP-43 regulates the alternative splicing of hnRNP A1 to yield an aggregation-prone variant in amyotrophic lateral sclerosis.
Deshaies JE, Shkreta L, Moszczynski AJ, Sidibé H, Semmler S, Fouillen A, Bennett ER, Bekenstein U, Destroismaisons L, Toutant J, Delmotte Q, Volkening K, Stabile S, Aulas A, Khalfallah Y, Soreq H, Nanci A, Strong MJ, Chabot B, Vande Velde C
Brain : a journal of neurology 2018 May 1;141(5):1320-1333
Brain : a journal of neurology 2018 May 1;141(5):1320-1333
Chronic Administration of Pimozide Fails to Attenuate Motor and Pathological Deficits in Two Mouse Models of Amyotrophic Lateral Sclerosis.
Pozzi S, Thammisetty SS, Julien JP
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2018 Jul;15(3):715-727
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2018 Jul;15(3):715-727
Gene Expression Profile in Frontal Cortex in Sporadic Frontotemporal Lobar Degeneration-TDP.
Andrés-Benito P, Gelpi E, Povedano M, Santpere G, Ferrer I
Journal of neuropathology and experimental neurology 2018 Jul 1;77(7):608-627
Journal of neuropathology and experimental neurology 2018 Jul 1;77(7):608-627
Alzheimer neuropathology without frontotemporal lobar degeneration hallmarks (TAR DNA-binding protein 43 inclusions) in missense progranulin mutation Cys139Arg.
Redaelli V, Rossi G, Maderna E, Kovacs GG, Piccoli E, Caroppo P, Cacciatore F, Spinello S, Grisoli M, Sozzi G, Salmaggi A, Tagliavini F, Giaccone G
Brain pathology (Zurich, Switzerland) 2018 Jan;28(1):72-76
Brain pathology (Zurich, Switzerland) 2018 Jan;28(1):72-76
Co-occurrence of mixed proteinopathies in late-stage Huntington's disease.
St-Amour I, Turgeon A, Goupil C, Planel E, Hébert SS
Acta neuropathologica 2018 Feb;135(2):249-265
Acta neuropathologica 2018 Feb;135(2):249-265
Cre-dependent AAV vectors for highly targeted expression of disease-related proteins and neurodegeneration in the substantia nigra.
Grames MS, Dayton RD, Jackson KL, Richard AD, Lu X, Klein RL
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2018 Aug;32(8):4420-4427
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2018 Aug;32(8):4420-4427
Systematic Screening of Ubiquitin/p62 Aggregates in Cerebellar Cortex Expands the Neuropathological Phenotype of the C9orf72 Expansion Mutation.
Ramos-Campoy O, Ávila-Polo R, Grau-Rivera O, Antonell A, Clarimón J, Rojas-García R, Charif S, Santiago-Valera V, Hernandez I, Aguilar M, Almenar C, Lopez-Villegas D, Bajo L, Pastor P, Van der Zee J, Lladó A, Sanchez-Valle R, Gelpi E
Journal of neuropathology and experimental neurology 2018 Aug 1;77(8):703-709
Journal of neuropathology and experimental neurology 2018 Aug 1;77(8):703-709
The clinical, neuroanatomical, and neuropathologic phenotype of TBK1-associated frontotemporal dementia: A longitudinal case report.
Koriath CA, Bocchetta M, Brotherhood E, Woollacott IO, Norsworthy P, Simón-Sánchez J, Blauwendraat C, Dick KM, Gordon E, Harding SR, Fox NC, Crutch S, Warren JD, Revesz T, Lashley T, Mead S, Rohrer JD
Alzheimer's & dementia (Amsterdam, Netherlands) 2017;6:75-81
Alzheimer's & dementia (Amsterdam, Netherlands) 2017;6:75-81
Frontotemporal Dementia Caused by the P301L Mutation in the MAPT Gene: Clinicopathological Features of 13 Cases from the Same Geographical Origin in Barcelona, Spain.
Borrego-Écija S, Morgado J, Palencia-Madrid L, Grau-Rivera O, Reñé R, Hernández I, Almenar C, Balasa M, Antonell A, Molinuevo JL, Lladó A, Martínez de Pancorbo M, Gelpi E, Sánchez-Valle R
Dementia and geriatric cognitive disorders 2017;44(3-4):213-221
Dementia and geriatric cognitive disorders 2017;44(3-4):213-221
Drosophila lines with mutant and wild type human TDP-43 replacing the endogenous gene reveals phosphorylation and ubiquitination in mutant lines in the absence of viability or lifespan defects.
Chang JC, Morton DB
PloS one 2017;12(7):e0180828
PloS one 2017;12(7):e0180828
The unexpected co-occurrence of GRN and MAPT p.A152T in Basque families: Clinical and pathological characteristics.
Moreno F, Indakoetxea B, Barandiaran M, Caballero MC, Gorostidi A, Calafell F, Gabilondo A, Tainta M, Zulaica M, Martí Massó JF, López de Munain A, Sánchez-Juan P, Lee SE
PloS one 2017;12(6):e0178093
PloS one 2017;12(6):e0178093
Clusterin protects neurons against intracellular proteotoxicity.
Gregory JM, Whiten DR, Brown RA, Barros TP, Kumita JR, Yerbury JJ, Satapathy S, McDade K, Smith C, Luheshi LM, Dobson CM, Wilson MR
Acta neuropathologica communications 2017 Nov 7;5(1):81
Acta neuropathologica communications 2017 Nov 7;5(1):81
Dioxins and related environmental contaminants increase TDP-43 levels.
Ash PEA, Stanford EA, Al Abdulatif A, Ramirez-Cardenas A, Ballance HI, Boudeau S, Jeh A, Murithi JM, Tripodis Y, Murphy GJ, Sherr DH, Wolozin B
Molecular neurodegeneration 2017 May 5;12(1):35
Molecular neurodegeneration 2017 May 5;12(1):35
TBK1 Mutation Spectrum in an Extended European Patient Cohort with Frontotemporal Dementia and Amyotrophic Lateral Sclerosis.
van der Zee J, Gijselinck I, Van Mossevelde S, Perrone F, Dillen L, Heeman B, Bäumer V, Engelborghs S, De Bleecker J, Baets J, Gelpi E, Rojas-García R, Clarimón J, Lleó A, Diehl-Schmid J, Alexopoulos P, Perneczky R, Synofzik M, Just J, Schöls L, Graff C, Thonberg H, Borroni B, Padovani A, Jordanova A, Sarafov S, Tournev I, de Mendonça A, Miltenberger-Miltényi G, Simões do Couto F, Ramirez A, Jessen F, Heneka MT, Gómez-Tortosa E, Danek A, Cras P, Vandenberghe R, De Jonghe P, De Deyn PP, Sleegers K, Cruts M, Van Broeckhoven C, Goeman J, Nuytten D, Smets K, Robberecht W, Damme PV, Bleecker J, Santens P, Dermaut B, Versijpt J, Michotte A, Ivanoiu A, Deryck O, Bergmans B, Delbeck J, Bruyland M, Willems C, Salmon E, Pastor P, Ortega-Cubero S, Benussi L, Ghidoni R, Binetti G, Hernández I, Boada M, Ruiz A, Sorbi S, Nacmias B, Bagnoli S, Sorbi S, Sanchez-Valle R, Llado A, Santana I, Rosário Almeida M, Frisoni GB, Maetzler W, Matej R, Fraidakis MJ, Kovacs GG, Fabrizi GM, Testi S
Human mutation 2017 Mar;38(3):297-309
Human mutation 2017 Mar;38(3):297-309
Motor-Coordinative and Cognitive Dysfunction Caused by Mutant TDP-43 Could Be Reversed by Inhibiting Its Mitochondrial Localization.
Wang W, Arakawa H, Wang L, Okolo O, Siedlak SL, Jiang Y, Gao J, Xie F, Petersen RB, Wang X
Molecular therapy : the journal of the American Society of Gene Therapy 2017 Jan 4;25(1):127-139
Molecular therapy : the journal of the American Society of Gene Therapy 2017 Jan 4;25(1):127-139
Monocytes of patients with amyotrophic lateral sclerosis linked to gene mutations display altered TDP-43 subcellular distribution.
De Marco G, Lomartire A, Calvo A, Risso A, De Luca E, Mostert M, Mandrioli J, Caponnetto C, Borghero G, Manera U, Canosa A, Moglia C, Restagno G, Fini N, Tarella C, Giordana MT, Rinaudo MT, Chiò A
Neuropathology and applied neurobiology 2017 Feb;43(2):133-153
Neuropathology and applied neurobiology 2017 Feb;43(2):133-153
Incidental neuronal intermediate filament inclusion pathology: unexpected biopsy findings in a 37-year-old woman with epilepsy.
Gelpi E, Carrato C, Grau-López L, Becerra JL, Garcia-Armengol R, Massuet A, Cervera-Carles L, Clarimon J, Beyer K, Álvarez R
Neuropathology and applied neurobiology 2017 Dec;43(7):636-640
Neuropathology and applied neurobiology 2017 Dec;43(7):636-640
TDP-43 Promotes Neurodegeneration by Impairing Chromatin Remodeling.
Berson A, Sartoris A, Nativio R, Van Deerlin V, Toledo JB, Porta S, Liu S, Chung CY, Garcia BA, Lee VM, Trojanowski JQ, Johnson FB, Berger SL, Bonini NM
Current biology : CB 2017 Dec 4;27(23):3579-3590.e6
Current biology : CB 2017 Dec 4;27(23):3579-3590.e6
Withania somnifera Reverses Transactive Response DNA Binding Protein 43 Proteinopathy in a Mouse Model of Amyotrophic Lateral Sclerosis/Frontotemporal Lobar Degeneration.
Dutta K, Patel P, Rahimian R, Phaneuf D, Julien JP
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2017 Apr;14(2):447-462
Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics 2017 Apr;14(2):447-462
Neuropathological criteria of anti-IgLON5-related tauopathy.
Gelpi E, Höftberger R, Graus F, Ling H, Holton JL, Dawson T, Popovic M, Pretnar-Oblak J, Högl B, Schmutzhard E, Poewe W, Ricken G, Santamaria J, Dalmau J, Budka H, Revesz T, Kovacs GG
Acta neuropathologica 2016 Oct;132(4):531-43
Acta neuropathologica 2016 Oct;132(4):531-43
Hereditary leukoencephalopathy with axonal spheroids: a spectrum of phenotypes from CNS vasculitis to parkinsonism in an adult onset leukodystrophy series.
Lynch DS, Jaunmuktane Z, Sheerin UM, Phadke R, Brandner S, Milonas I, Dean A, Bajaj N, McNicholas N, Costello D, Cronin S, McGuigan C, Rossor M, Fox N, Murphy E, Chataway J, Houlden H
Journal of neurology, neurosurgery, and psychiatry 2016 May;87(5):512-9
Journal of neurology, neurosurgery, and psychiatry 2016 May;87(5):512-9
Two mutations G335D and Q343R within the amyloidogenic core region of TDP-43 influence its aggregation and inclusion formation.
Jiang LL, Zhao J, Yin XF, He WT, Yang H, Che MX, Hu HY
Scientific reports 2016 Mar 31;6:23928
Scientific reports 2016 Mar 31;6:23928
Increased cytoplasmic TARDBP mRNA in affected spinal motor neurons in ALS caused by abnormal autoregulation of TDP-43.
Koyama A, Sugai A, Kato T, Ishihara T, Shiga A, Toyoshima Y, Koyama M, Konno T, Hirokawa S, Yokoseki A, Nishizawa M, Kakita A, Takahashi H, Onodera O
Nucleic acids research 2016 Jul 8;44(12):5820-36
Nucleic acids research 2016 Jul 8;44(12):5820-36
Developmentally Regulated RNA-binding Protein 1 (Drb1)/RNA-binding Motif Protein 45 (RBM45), a Nuclear-Cytoplasmic Trafficking Protein, Forms TAR DNA-binding Protein 43 (TDP-43)-mediated Cytoplasmic Aggregates.
Mashiko T, Sakashita E, Kasashima K, Tominaga K, Kuroiwa K, Nozaki Y, Matsuura T, Hamamoto T, Endo H
The Journal of biological chemistry 2016 Jul 15;291(29):14996-5007
The Journal of biological chemistry 2016 Jul 15;291(29):14996-5007
Pisa syndrome in a patient with pathologically confirmed Parkinson's disease.
Solla P, Grau-Rivera O, Gelpi E, Marrosu F, Martí MJ
Neuropathology and applied neurobiology 2016 Dec;42(7):654-658
Neuropathology and applied neurobiology 2016 Dec;42(7):654-658
Altered Mitochondria, Protein Synthesis Machinery, and Purine Metabolism Are Molecular Contributors to the Pathogenesis of Creutzfeldt-Jakob Disease.
Ansoleaga B, Garcia-Esparcia P, Llorens F, Hernández-Ortega K, Carmona Tech M, Antonio Del Rio J, Zerr I, Ferrer I
Journal of neuropathology and experimental neurology 2016 Aug;75(8):755-769
Journal of neuropathology and experimental neurology 2016 Aug;75(8):755-769
The inhibition of TDP-43 mitochondrial localization blocks its neuronal toxicity.
Wang W, Wang L, Lu J, Siedlak SL, Fujioka H, Liang J, Jiang S, Ma X, Jiang Z, da Rocha EL, Sheng M, Choi H, Lerou PH, Li H, Wang X
Nature medicine 2016 Aug;22(8):869-78
Nature medicine 2016 Aug;22(8):869-78
CCNF mutations in amyotrophic lateral sclerosis and frontotemporal dementia.
Williams KL, Topp S, Yang S, Smith B, Fifita JA, Warraich ST, Zhang KY, Farrawell N, Vance C, Hu X, Chesi A, Leblond CS, Lee A, Rayner SL, Sundaramoorthy V, Dobson-Stone C, Molloy MP, van Blitterswijk M, Dickson DW, Petersen RC, Graff-Radford NR, Boeve BF, Murray ME, Pottier C, Don E, Winnick C, McCann EP, Hogan A, Daoud H, Levert A, Dion PA, Mitsui J, Ishiura H, Takahashi Y, Goto J, Kost J, Gellera C, Gkazi AS, Miller J, Stockton J, Brooks WS, Boundy K, Polak M, Muñoz-Blanco JL, Esteban-Pérez J, Rábano A, Hardiman O, Morrison KE, Ticozzi N, Silani V, de Belleroche J, Glass JD, Kwok JB, Guillemin GJ, Chung RS, Tsuji S, Brown RH Jr, García-Redondo A, Rademakers R, Landers JE, Gitler AD, Rouleau GA, Cole NJ, Yerbury JJ, Atkin JD, Shaw CE, Nicholson GA, Blair IP
Nature communications 2016 Apr 15;7:11253
Nature communications 2016 Apr 15;7:11253
Early diagnosis of cardiac involvement in idiopathic inflammatory myopathy by cardiac magnetic resonance tomography.
Rosenbohm A, Buckert D, Gerischer N, Walcher T, Kassubek J, Rottbauer W, Ludolph AC, Bernhardt P
Journal of neurology 2015;262(4):949-56
Journal of neurology 2015;262(4):949-56
Cytoplasmic mislocalization of RNA splicing factors and aberrant neuronal gene splicing in TDP-43 transgenic pig brain.
Wang G, Yang H, Yan S, Wang CE, Liu X, Zhao B, Ouyang Z, Yin P, Liu Z, Zhao Y, Liu T, Fan N, Guo L, Li S, Li XJ, Lai L
Molecular neurodegeneration 2015 Sep 3;10:42
Molecular neurodegeneration 2015 Sep 3;10:42
Low molecular weight species of TDP-43 generated by abnormal splicing form inclusions in amyotrophic lateral sclerosis and result in motor neuron death.
Xiao S, Sanelli T, Chiang H, Sun Y, Chakrabartty A, Keith J, Rogaeva E, Zinman L, Robertson J
Acta neuropathologica 2015 Jul;130(1):49-61
Acta neuropathologica 2015 Jul;130(1):49-61
Neuropathological assessments of the pathology in frontotemporal lobar degeneration with TDP43-positive inclusions: an inter-laboratory study by the BrainNet Europe consortium.
Alafuzoff I, Pikkarainen M, Neumann M, Arzberger T, Al-Sarraj S, Bodi I, Bogdanovic N, Bugiani O, Ferrer I, Gelpi E, Gentleman S, Giaccone G, Graeber MB, Hortobagyi T, Ince PG, Ironside JW, Kavantzas N, King A, Korkolopoulou P, Kovács GG, Meyronet D, Monoranu C, Nilsson T, Parchi P, Patsouris E, Revesz T, Roggendorf W, Rozemuller A, Seilhean D, Streichenberger N, Thal DR, Wharton SB, Kretzschmar H
Journal of neural transmission (Vienna, Austria : 1996) 2015 Jul;122(7):957-72
Journal of neural transmission (Vienna, Austria : 1996) 2015 Jul;122(7):957-72
TDP-43 as a possible biomarker for frontotemporal lobar degeneration: a systematic review of existing antibodies.
Goossens J, Vanmechelen E, Trojanowski JQ, Lee VM, Van Broeckhoven C, van der Zee J, Engelborghs S
Acta neuropathologica communications 2015 Apr 1;3:15
Acta neuropathologica communications 2015 Apr 1;3:15
Casein kinase II induced polymerization of soluble TDP-43 into filaments is inhibited by heat shock proteins.
Carlomagno Y, Zhang Y, Davis M, Lin WL, Cook C, Dunmore J, Tay W, Menkosky K, Cao X, Petrucelli L, Deture M
PloS one 2014;9(3):e90452
PloS one 2014;9(3):e90452
A 6.4 Mb duplication of the α-synuclein locus causing frontotemporal dementia and Parkinsonism: phenotype-genotype correlations.
Kara E, Kiely AP, Proukakis C, Giffin N, Love S, Hehir J, Rantell K, Pandraud A, Hernandez DG, Nacheva E, Pittman AM, Nalls MA, Singleton AB, Revesz T, Bhatia KP, Quinn N, Hardy J, Holton JL, Houlden H
JAMA neurology 2014 Sep;71(9):1162-71
JAMA neurology 2014 Sep;71(9):1162-71
Parkin-mediated reduction of nuclear and soluble TDP-43 reverses behavioral decline in symptomatic mice.
Wenqiang C, Lonskaya I, Hebron ML, Ibrahim Z, Olszewski RT, Neale JH, Moussa CE
Human molecular genetics 2014 Sep 15;23(18):4960-9
Human molecular genetics 2014 Sep 15;23(18):4960-9
Abnormal serine phosphorylation of insulin receptor substrate 1 is associated with tau pathology in Alzheimer's disease and tauopathies.
Yarchoan M, Toledo JB, Lee EB, Arvanitakis Z, Kazi H, Han LY, Louneva N, Lee VM, Kim SF, Trojanowski JQ, Arnold SE
Acta neuropathologica 2014 Nov;128(5):679-89
Acta neuropathologica 2014 Nov;128(5):679-89
TDP-43 causes differential pathology in neuronal versus glial cells in the mouse brain.
Yan S, Wang CE, Wei W, Gaertig MA, Lai L, Li S, Li XJ
Human molecular genetics 2014 May 15;23(10):2678-93
Human molecular genetics 2014 May 15;23(10):2678-93
Plasma phosphorylated TDP-43 levels are elevated in patients with frontotemporal dementia carrying a C9orf72 repeat expansion or a GRN mutation.
Suárez-Calvet M, Dols-Icardo O, Lladó A, Sánchez-Valle R, Hernández I, Amer G, Antón-Aguirre S, Alcolea D, Fortea J, Ferrer I, van der Zee J, Dillen L, Van Broeckhoven C, Molinuevo JL, Blesa R, Clarimón J, Lleó A
Journal of neurology, neurosurgery, and psychiatry 2014 Jun;85(6):684-91
Journal of neurology, neurosurgery, and psychiatry 2014 Jun;85(6):684-91
A pathogenic progranulin mutation and C9orf72 repeat expansion in a family with frontotemporal dementia.
Lashley T, Rohrer JD, Mahoney C, Gordon E, Beck J, Mead S, Warren J, Rossor M, Revesz T
Neuropathology and applied neurobiology 2014 Jun;40(4):502-13
Neuropathology and applied neurobiology 2014 Jun;40(4):502-13
Widespread RNA metabolism impairment in sporadic inclusion body myositis TDP43-proteinopathy.
Cortese A, Plagnol V, Brady S, Simone R, Lashley T, Acevedo-Arozena A, de Silva R, Greensmith L, Holton J, Hanna MG, Fisher EM, Fratta P
Neurobiology of aging 2014 Jun;35(6):1491-8
Neurobiology of aging 2014 Jun;35(6):1491-8
Multiple organ involvement by alpha-synuclein pathology in Lewy body disorders.
Gelpi E, Navarro-Otano J, Tolosa E, Gaig C, Compta Y, Rey MJ, Martí MJ, Hernández I, Valldeoriola F, Reñé R, Ribalta T
Movement disorders : official journal of the Movement Disorder Society 2014 Jul;29(8):1010-8
Movement disorders : official journal of the Movement Disorder Society 2014 Jul;29(8):1010-8
UBE2E ubiquitin-conjugating enzymes and ubiquitin isopeptidase Y regulate TDP-43 protein ubiquitination.
Hans F, Fiesel FC, Strong JC, Jäckel S, Rasse TM, Geisler S, Springer W, Schulz JB, Voigt A, Kahle PJ
The Journal of biological chemistry 2014 Jul 4;289(27):19164-79
The Journal of biological chemistry 2014 Jul 4;289(27):19164-79
Accumulation of C-terminal fragments of transactive response DNA-binding protein 43 leads to synaptic loss and cognitive deficits in human TDP-43 transgenic mice.
Medina DX, Orr ME, Oddo S
Neurobiology of aging 2014 Jan;35(1):79-87
Neurobiology of aging 2014 Jan;35(1):79-87
Cell injury and premature neurodegeneration in focal malformations of cortical development.
Iyer A, Prabowo A, Anink J, Spliet WG, van Rijen PC, Aronica E
Brain pathology (Zurich, Switzerland) 2014 Jan;24(1):1-17
Brain pathology (Zurich, Switzerland) 2014 Jan;24(1):1-17
The novel MAPT mutation K298E: mechanisms of mutant tau toxicity, brain pathology and tau expression in induced fibroblast-derived neurons.
Iovino M, Pfisterer U, Holton JL, Lashley T, Swingler RJ, Calo L, Treacy R, Revesz T, Parmar M, Goedert M, Muqit MM, Spillantini MG
Acta neuropathologica 2014 Feb;127(2):283-95
Acta neuropathologica 2014 Feb;127(2):283-95
Disease-associated mutations of TDP-43 promote turnover of the protein through the proteasomal pathway.
Araki W, Minegishi S, Motoki K, Kume H, Hohjoh H, Araki YM, Tamaoka A
Molecular neurobiology 2014 Dec;50(3):1049-58
Molecular neurobiology 2014 Dec;50(3):1049-58
No interaction between tau and TDP-43 pathologies in either frontotemporal lobar degeneration or motor neurone disease.
Robinson AC, Thompson JC, Weedon L, Rollinson S, Pickering-Brown S, Snowden JS, Davidson YS, Mann DM
Neuropathology and applied neurobiology 2014 Dec;40(7):844-54
Neuropathology and applied neurobiology 2014 Dec;40(7):844-54
TDP-1, the Caenorhabditis elegans ortholog of TDP-43, limits the accumulation of double-stranded RNA.
Saldi TK, Ash PE, Wilson G, Gonzales P, Garrido-Lecca A, Roberts CM, Dostal V, Gendron TF, Stein LD, Blumenthal T, Petrucelli L, Link CD
The EMBO journal 2014 Dec 17;33(24):2947-66
The EMBO journal 2014 Dec 17;33(24):2947-66
Clinical and neuropathological variability in clinically isolated central nervous system Whipple's disease.
Balasa M, Gelpi E, Rey MJ, Vila J, Ramió-Torrentà L, Quiles Granado AM, Molina Latorre R, Lepidi H, Raoult D, Saiz A
Brain pathology (Zurich, Switzerland) 2014 Apr;24(3):230-8
Brain pathology (Zurich, Switzerland) 2014 Apr;24(3):230-8
Reduced cellular Ca(2+) availability enhances TDP-43 cleavage by apoptotic caspases.
De Marco G, Lomartire A, Mandili G, Lupino E, Buccinnà B, Ramondetti C, Moglia C, Novelli F, Piccinini M, Mostert M, Rinaudo MT, Chiò A, Calvo A
Biochimica et biophysica acta 2014 Apr;1843(4):725-34
Biochimica et biophysica acta 2014 Apr;1843(4):725-34
Parkin reverses TDP-43-induced cell death and failure of amino acid homeostasis.
Hebron M, Chen W, Miessau MJ, Lonskaya I, Moussa CE
Journal of neurochemistry 2014 Apr;129(2):350-61
Journal of neurochemistry 2014 Apr;129(2):350-61
Interaction of transactive response DNA binding protein 43 with nuclear factor κB in mild cognitive impairment with episodic memory deficits.
Ohta Y, Tremblay C, Schneider JA, Bennett DA, Calon F, Julien JP
Acta neuropathologica communications 2014 Apr 1;2:37
Acta neuropathologica communications 2014 Apr 1;2:37
The pathological phenotypes of human TDP-43 transgenic mouse models are independent of downregulation of mouse Tdp-43.
Xu YF, Prudencio M, Hubbard JM, Tong J, Whitelaw EC, Jansen-West K, Stetler C, Cao X, Song J, Zhang YJ
PloS one 2013;8(7):e69864
PloS one 2013;8(7):e69864
ALS-associated TDP-43 induces endoplasmic reticulum stress, which drives cytoplasmic TDP-43 accumulation and stress granule formation.
Walker AK, Soo KY, Sundaramoorthy V, Parakh S, Ma Y, Farg MA, Wallace RH, Crouch PJ, Turner BJ, Horne MK, Atkin JD
PloS one 2013;8(11):e81170
PloS one 2013;8(11):e81170
Reduced cholinergic olfactory centrifugal inputs in patients with neurodegenerative disorders and MPTP-treated monkeys.
Mundiñano IC, Hernandez M, Dicaudo C, Ordoñez C, Marcilla I, Tuñon MT, Luquin MR
Acta neuropathologica 2013 Sep;126(3):411-25
Acta neuropathologica 2013 Sep;126(3):411-25
Neurodegenerative disease status and post-mortem pathology in idiopathic rapid-eye-movement sleep behaviour disorder: an observational cohort study.
Iranzo A, Tolosa E, Gelpi E, Molinuevo JL, Valldeoriola F, Serradell M, Sanchez-Valle R, Vilaseca I, Lomeña F, Vilas D, Lladó A, Gaig C, Santamaria J
The Lancet. Neurology 2013 May;12(5):443-53
The Lancet. Neurology 2013 May;12(5):443-53
MAPT H1 haplotype is associated with enhanced α-synuclein deposition in dementia with Lewy bodies.
Colom-Cadena M, Gelpi E, Martí MJ, Charif S, Dols-Icardo O, Blesa R, Clarimón J, Lleó A
Neurobiology of aging 2013 Mar;34(3):936-42
Neurobiology of aging 2013 Mar;34(3):936-42
Characterization of thorn-shaped astrocytes in white matter of temporal lobe in Alzheimer's disease brains.
López-González I, Carmona M, Blanco R, Luna-Muñoz J, Martínez-Mandonado A, Mena R, Ferrer I
Brain pathology (Zurich, Switzerland) 2013 Mar;23(2):144-53
Brain pathology (Zurich, Switzerland) 2013 Mar;23(2):144-53
Reactive astrocytes secrete lcn2 to promote neuron death.
Bi F, Huang C, Tong J, Qiu G, Huang B, Wu Q, Li F, Xu Z, Bowser R, Xia XG, Zhou H
Proceedings of the National Academy of Sciences of the United States of America 2013 Mar 5;110(10):4069-74
Proceedings of the National Academy of Sciences of the United States of America 2013 Mar 5;110(10):4069-74
Primary progressive aphasia with parkinsonism.
Doherty KM, Rohrer JD, Lees AJ, Holton JL, Warren J
Movement disorders : official journal of the Movement Disorder Society 2013 Jun;28(6):741-6
Movement disorders : official journal of the Movement Disorder Society 2013 Jun;28(6):741-6
Asymptomatic hyper-creatine-kinase-emia as sole manifestation of inclusion body myositis.
Finsterer J, Stöllberger C, Kovacs GG
Neurology international 2013 Jun 25;5(2):34-6
Neurology international 2013 Jun 25;5(2):34-6
Selective forelimb impairment in rats expressing a pathological TDP-43 25 kDa C-terminal fragment to mimic amyotrophic lateral sclerosis.
Dayton RD, Gitcho MA, Orchard EA, Wilson JD, Wang DB, Cain CD, Johnson JA, Zhang YJ, Petrucelli L, Mathis JM, Klein RL
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Jul;21(7):1324-34
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Jul;21(7):1324-34
Globular glial-like inclusions in a patient with advanced Alzheimer's disease.
Gelpi E, Cullel F, Navarro-Otano J, Lladó A
Acta neuropathologica 2013 Jul;126(1):155-7
Acta neuropathologica 2013 Jul;126(1):155-7
The long non-coding RNA nuclear-enriched abundant transcript 1_2 induces paraspeckle formation in the motor neuron during the early phase of amyotrophic lateral sclerosis.
Nishimoto Y, Nakagawa S, Hirose T, Okano HJ, Takao M, Shibata S, Suyama S, Kuwako K, Imai T, Murayama S, Suzuki N, Okano H
Molecular brain 2013 Jul 8;6:31
Molecular brain 2013 Jul 8;6:31
Expression of ALS-linked TDP-43 mutant in astrocytes causes non-cell-autonomous motor neuron death in rats.
Tong J, Huang C, Bi F, Wu Q, Huang B, Liu X, Li F, Zhou H, Xia XG
The EMBO journal 2013 Jul 3;32(13):1917-26
The EMBO journal 2013 Jul 3;32(13):1917-26
Parkin ubiquitinates Tar-DNA binding protein-43 (TDP-43) and promotes its cytosolic accumulation via interaction with histone deacetylase 6 (HDAC6).
Hebron ML, Lonskaya I, Sharpe K, Weerasinghe PP, Algarzae NK, Shekoyan AR, Moussa CE
The Journal of biological chemistry 2013 Feb 8;288(6):4103-15
The Journal of biological chemistry 2013 Feb 8;288(6):4103-15
Accelerated disease onset with stabilized familial amyotrophic lateral sclerosis (ALS)-linked mutant TDP-43 proteins.
Watanabe S, Kaneko K, Yamanaka K
The Journal of biological chemistry 2013 Feb 1;288(5):3641-54
The Journal of biological chemistry 2013 Feb 1;288(5):3641-54
Prominent psychiatric symptoms in patients with Parkinson's disease and concomitant argyrophilic grain disease.
Grau-Rivera O, Gelpi E, Rey MJ, Valldeoriola F, Tolosa E, Compta Y, Martí MJ
Journal of neurology 2013 Dec;260(12):3002-9
Journal of neurology 2013 Dec;260(12):3002-9
The involvement of the cerebellum in amyotrophic lateral sclerosis.
Prell T, Grosskreutz J
Amyotrophic lateral sclerosis & frontotemporal degeneration 2013 Dec;14(7-8):507-15
Amyotrophic lateral sclerosis & frontotemporal degeneration 2013 Dec;14(7-8):507-15
Atypical neuropathological sCJD-MM phenotype with abundant white matter Kuru-type plaques sparing the cerebellar cortex.
Gelpi E, Soler Insa JM, Parchi P, Saverioni D, Yagüe J, Nos C, Martínez-Saez E, Ribalta T, Ferrer I, Sanchez-Valle R
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Apr;33(2):204-8
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Apr;33(2):204-8
Co-occurrence of different pathologies in dementia: implications for dementia diagnosis.
Echávarri C, Burgmans S, Caballero MC, García-Bragado F, Verhey FR, Uylings HB
Journal of Alzheimer's disease : JAD 2012;30(4):909-17
Journal of Alzheimer's disease : JAD 2012;30(4):909-17
Breakpoint sequence analysis of an AβPP locus duplication associated with autosomal dominant Alzheimer's disease and severe cerebral amyloid angiopathy.
Antonell A, Gelpi E, Sánchez-Valle R, Martínez R, Molinuevo JL, Lladó A
Journal of Alzheimer's disease : JAD 2012;28(2):303-8
Journal of Alzheimer's disease : JAD 2012;28(2):303-8
TDP-43 plasma levels are higher in amyotrophic lateral sclerosis.
Verstraete E, Kuiperij HB, van Blitterswijk MM, Veldink JH, Schelhaas HJ, van den Berg LH, Verbeek MM
Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases 2012 Sep;13(5):446-51
Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases 2012 Sep;13(5):446-51
An MND/ALS phenotype associated with C9orf72 repeat expansion: abundant p62-positive, TDP-43-negative inclusions in cerebral cortex, hippocampus and cerebellum but without associated cognitive decline.
Troakes C, Maekawa S, Wijesekera L, Rogelj B, Siklós L, Bell C, Smith B, Newhouse S, Vance C, Johnson L, Hortobágyi T, Shatunov A, Al-Chalabi A, Leigh N, Shaw CE, King A, Al-Sarraj S
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Oct;32(5):505-14
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Oct;32(5):505-14
Matrix metalloproteinase-9 expression in the nuclear compartment of neurons and glial cells in aging and stroke.
Pirici D, Pirici I, Mogoanta L, Margaritescu O, Tudorica V, Margaritescu C, Ion DA, Simionescu C, Coconu M
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Oct;32(5):492-504
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Oct;32(5):492-504
XBP1 depletion precedes ubiquitin aggregation and Golgi fragmentation in TDP-43 transgenic rats.
Tong J, Huang C, Bi F, Wu Q, Huang B, Zhou H
Journal of neurochemistry 2012 Nov;123(3):406-16
Journal of neurochemistry 2012 Nov;123(3):406-16
Wild type TDP-43 induces neuro-inflammation and alters APP metabolism in lentiviral gene transfer models.
Herman AM, Khandelwal PJ, Rebeck GW, Moussa CE
Experimental neurology 2012 May;235(1):297-305
Experimental neurology 2012 May;235(1):297-305
TDP-43 regulates global translational yield by splicing of exon junction complex component SKAR.
Fiesel FC, Weber SS, Supper J, Zell A, Kahle PJ
Nucleic acids research 2012 Mar;40(6):2668-82
Nucleic acids research 2012 Mar;40(6):2668-82
Identification of c-myc-dependent proteins in the medulloblastoma cell line D425Med.
Azizi AA, Li L, Ströbel T, Chen WQ, Slavc I, Lubec G
Amino acids 2012 Jun;42(6):2149-63
Amino acids 2012 Jun;42(6):2149-63
Mutant TDP-43 in motor neurons promotes the onset and progression of ALS in rats.
Huang C, Tong J, Bi F, Zhou H, Xia XG
The Journal of clinical investigation 2012 Jan;122(1):107-18
The Journal of clinical investigation 2012 Jan;122(1):107-18
Epitope mapping of antibodies against TDP-43 and detection of protease-resistant fragments of pathological TDP-43 in amyotrophic lateral sclerosis and frontotemporal lobar degeneration.
Tsuji H, Nonaka T, Yamashita M, Masuda-Suzukake M, Kametani F, Akiyama H, Mann DM, Tamaoka A, Hasegawa M
Biochemical and biophysical research communications 2012 Jan 6;417(1):116-21
Biochemical and biophysical research communications 2012 Jan 6;417(1):116-21
Unusual clinical presentation and neuropathology in two subjects with fused-in sarcoma (FUS) positive inclusions.
Hartikainen PH, Pikkarainen M, Hänninen T, Soininen H, Alafuzoff I
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Feb;32(1):60-8
Neuropathology : official journal of the Japanese Society of Neuropathology 2012 Feb;32(1):60-8
Frontotemporal lobar degeneration-related proteins induce only subtle memory-related deficits when bilaterally overexpressed in the dorsal hippocampus.
Dayton RD, Wang DB, Cain CD, Schrott LM, Ramirez JJ, King MA, Klein RL
Experimental neurology 2012 Feb;233(2):807-14
Experimental neurology 2012 Feb;233(2):807-14
Pick's pathology in Parkinson's disease with dementia.
Vilas D, Marti MJ, Botta-Orfila T, Colom-Cadena M, Gelpi E
Neuropathology and applied neurobiology 2012 Dec;38(7):737-43
Neuropathology and applied neurobiology 2012 Dec;38(7):737-43
Abnormal regenerative responses and impaired axonal outgrowth after nerve crush in TDP-43 transgenic mouse models of amyotrophic lateral sclerosis.
Swarup V, Audet JN, Phaneuf D, Kriz J, Julien JP
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Dec 12;32(50):18186-95
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Dec 12;32(50):18186-95
Hirano body-rich subtypes of Creutzfeldt-Jakob disease.
Martinez-Saez E, Gelpi E, Rey MJ, Ferrer I, Ribalta T, Botta-Orfila T, Nos C, Yagüe J, Sanchez-Valle R
Neuropathology and applied neurobiology 2012 Apr;38(2):153-61
Neuropathology and applied neurobiology 2012 Apr;38(2):153-61
Pathological hallmarks of amyotrophic lateral sclerosis/frontotemporal lobar degeneration in transgenic mice produced with TDP-43 genomic fragments.
Swarup V, Phaneuf D, Bareil C, Robertson J, Rouleau GA, Kriz J, Julien JP
Brain : a journal of neurology 2011 Sep;134(Pt 9):2610-26
Brain : a journal of neurology 2011 Sep;134(Pt 9):2610-26
Neurofibrillary tangle pathology and Braak staging in chronic epilepsy in relation to traumatic brain injury and hippocampal sclerosis: a post-mortem study.
Thom M, Liu JY, Thompson P, Phadke R, Narkiewicz M, Martinian L, Marsdon D, Koepp M, Caboclo L, Catarino CB, Sisodiya SM
Brain : a journal of neurology 2011 Oct;134(Pt 10):2969-81
Brain : a journal of neurology 2011 Oct;134(Pt 10):2969-81
The ALS-associated proteins FUS and TDP-43 function together to affect Drosophila locomotion and life span.
Wang JW, Brent JR, Tomlinson A, Shneider NA, McCabe BD
The Journal of clinical investigation 2011 Oct;121(10):4118-26
The Journal of clinical investigation 2011 Oct;121(10):4118-26
Cytoplasmic accumulation of TDP-43 in circulating lymphomonocytes of ALS patients with and without TARDBP mutations.
De Marco G, Lupino E, Calvo A, Moglia C, Buccinnà B, Grifoni S, Ramondetti C, Lomartire A, Rinaudo MT, Piccinini M, Giordana MT, Chiò A
Acta neuropathologica 2011 May;121(5):611-22
Acta neuropathologica 2011 May;121(5):611-22
TAR-DNA binding protein-43 and alterations in the hippocampus.
Rauramaa T, Pikkarainen M, Englund E, Ince PG, Jellinger K, Paetau A, Alafuzoff I
Journal of neural transmission (Vienna, Austria : 1996) 2011 May;118(5):683-9
Journal of neural transmission (Vienna, Austria : 1996) 2011 May;118(5):683-9
FTLD-TDP with motor neuron disease, visuospatial impairment and a progressive supranuclear palsy-like syndrome: broadening the clinical phenotype of TDP-43 proteinopathies. A report of three cases.
Rusina R, Kovacs GG, Fiala J, Hort J, Ridzoň P, Holmerová I, Ströbel T, Matěj R
BMC neurology 2011 May 10;11:50
BMC neurology 2011 May 10;11:50
Neocortical layer formation of human developing brains and lissencephalies: consideration of layer-specific marker expression.
Saito T, Hanai S, Takashima S, Nakagawa E, Okazaki S, Inoue T, Miyata R, Hoshino K, Akashi T, Sasaki M, Goto Y, Hayashi M, Itoh M
Cerebral cortex (New York, N.Y. : 1991) 2011 Mar;21(3):588-96
Cerebral cortex (New York, N.Y. : 1991) 2011 Mar;21(3):588-96
Elevated CSF TDP-43 levels in amyotrophic lateral sclerosis: specificity, sensitivity, and a possible prognostic value.
Noto Y, Shibuya K, Sato Y, Kanai K, Misawa S, Sawai S, Mori M, Uchiyama T, Isose S, Nasu S, Sekiguchi Y, Fujimaki Y, Kasai T, Tokuda T, Nakagawa M, Kuwabara S
Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases 2011 Mar;12(2):140-3
Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases 2011 Mar;12(2):140-3
TDP-43 potentiates alpha-synuclein toxicity to dopaminergic neurons in transgenic mice.
Tian T, Huang C, Tong J, Yang M, Zhou H, Xia XG
International journal of biological sciences 2011 Mar 4;7(2):234-43
International journal of biological sciences 2011 Mar 4;7(2):234-43
TDP-43 potentiates alpha-synuclein toxicity to dopaminergic neurons in transgenic mice.
Tian T, Huang C, Tong J, Yang M, Zhou H, Xia XG
International journal of biological sciences 2011 Mar 4;7(2):234-43
International journal of biological sciences 2011 Mar 4;7(2):234-43
Aggregation of the 35-kDa fragment of TDP-43 causes formation of cytoplasmic inclusions and alteration of RNA processing.
Che MX, Jiang YJ, Xie YY, Jiang LL, Hu HY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Jul;25(7):2344-53
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Jul;25(7):2344-53
Increased dopaminergic cells and protein aggregates in the olfactory bulb of patients with neurodegenerative disorders.
Mundiñano IC, Caballero MC, Ordóñez C, Hernandez M, DiCaudo C, Marcilla I, Erro ME, Tuñon MT, Luquin MR
Acta neuropathologica 2011 Jul;122(1):61-74
Acta neuropathologica 2011 Jul;122(1):61-74
Distribution and pattern of pathology in subjects with familial or sporadic late-onset cerebellar ataxia as assessed by p62/sequestosome immunohistochemistry.
Pikkarainen M, Hartikainen P, Soininen H, Alafuzoff I
Cerebellum (London, England) 2011 Dec;10(4):720-31
Cerebellum (London, England) 2011 Dec;10(4):720-31
Molecular mechanisms of MLC1 and GLIALCAM mutations in megalencephalic leukoencephalopathy with subcortical cysts.
López-Hernández T, Sirisi S, Capdevila-Nortes X, Montolio M, Fernández-Dueñas V, Scheper GC, van der Knaap MS, Casquero P, Ciruela F, Ferrer I, Nunes V, Estévez R
Human molecular genetics 2011 Aug 15;20(16):3266-77
Human molecular genetics 2011 Aug 15;20(16):3266-77
TDP-43 pathology may occur in the BRI2 gene-related dementias.
Lashley T, Holton JL, Revesz T
Acta neuropathologica 2011 Apr;121(4):559-60
Acta neuropathologica 2011 Apr;121(4):559-60
β-amyloid triggers ALS-associated TDP-43 pathology in AD models.
Herman AM, Khandelwal PJ, Stanczyk BB, Rebeck GW, Moussa CE
Brain research 2011 Apr 22;1386:191-9
Brain research 2011 Apr 22;1386:191-9
The first case of protease-sensitive prionopathy (PSPr) in The Netherlands: a patient with an unusual GSS-like clinical phenotype.
Jansen C, Head MW, van Gool WA, Baas F, Yull H, Ironside JW, Rozemuller AJ
Journal of neurology, neurosurgery, and psychiatry 2010 Sep;81(9):1052-5
Journal of neurology, neurosurgery, and psychiatry 2010 Sep;81(9):1052-5
Altered distributions of Gemini of coiled bodies and mitochondria in motor neurons of TDP-43 transgenic mice.
Shan X, Chiang PM, Price DL, Wong PC
Proceedings of the National Academy of Sciences of the United States of America 2010 Sep 14;107(37):16325-30
Proceedings of the National Academy of Sciences of the United States of America 2010 Sep 14;107(37):16325-30
Tubulin polymerization promoting protein (TPPP/p25) as a marker for oligodendroglial changes in multiple sclerosis.
Höftberger R, Fink S, Aboul-Enein F, Botond G, Olah J, Berki T, Ovadi J, Lassmann H, Budka H, Kovacs GG
Glia 2010 Nov 15;58(15):1847-57
Glia 2010 Nov 15;58(15):1847-57
Depletion of oxidative and endoplasmic reticulum stress regulators in Pick disease.
Ilieva EV, Naudí A, Kichev A, Ferrer I, Pamplona R, Portero-Otín M
Free radical biology & medicine 2010 May 15;48(10):1302-10
Free radical biology & medicine 2010 May 15;48(10):1302-10
Mutations of optineurin in amyotrophic lateral sclerosis.
Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y, Kinoshita Y, Kamada M, Nodera H, Suzuki H, Komure O, Matsuura S, Kobatake K, Morimoto N, Abe K, Suzuki N, Aoki M, Kawata A, Hirai T, Kato T, Ogasawara K, Hirano A, Takumi T, Kusaka H, Hagiwara K, Kaji R, Kawakami H
Nature 2010 May 13;465(7295):223-6
Nature 2010 May 13;465(7295):223-6
Amyotrophic lateral sclerosis: dash-like accumulation of phosphorylated TDP-43 in somatodendritic and axonal compartments of somatomotor neurons of the lower brainstem and spinal cord.
Braak H, Ludolph A, Thal DR, Del Tredici K
Acta neuropathologica 2010 Jul;120(1):67-74
Acta neuropathologica 2010 Jul;120(1):67-74
ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS.
Ling SC, Albuquerque CP, Han JS, Lagier-Tourenne C, Tokunaga S, Zhou H, Cleveland DW
Proceedings of the National Academy of Sciences of the United States of America 2010 Jul 27;107(30):13318-23
Proceedings of the National Academy of Sciences of the United States of America 2010 Jul 27;107(30):13318-23
Knockdown of transactive response DNA-binding protein (TDP-43) downregulates histone deacetylase 6.
Fiesel FC, Voigt A, Weber SS, Van den Haute C, Waldenmaier A, Görner K, Walter M, Anderson ML, Kern JV, Rasse TM, Schmidt T, Springer W, Kirchner R, Bonin M, Neumann M, Baekelandt V, Alunni-Fabbroni M, Schulz JB, Kahle PJ
The EMBO journal 2010 Jan 6;29(1):209-21
The EMBO journal 2010 Jan 6;29(1):209-21
Characterization of alternative isoforms and inclusion body of the TAR DNA-binding protein-43.
Nishimoto Y, Ito D, Yagi T, Nihei Y, Tsunoda Y, Suzuki N
The Journal of biological chemistry 2010 Jan 1;285(1):608-19
The Journal of biological chemistry 2010 Jan 1;285(1):608-19
TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration.
Wils H, Kleinberger G, Janssens J, Pereson S, Joris G, Cuijt I, Smits V, Ceuterick-de Groote C, Van Broeckhoven C, Kumar-Singh S
Proceedings of the National Academy of Sciences of the United States of America 2010 Feb 23;107(8):3858-63
Proceedings of the National Academy of Sciences of the United States of America 2010 Feb 23;107(8):3858-63
TDP-43 physically interacts with amyotrophic lateral sclerosis-linked mutant CuZn superoxide dismutase.
Higashi S, Tsuchiya Y, Araki T, Wada K, Kabuta T
Neurochemistry international 2010 Dec;57(8):906-13
Neurochemistry international 2010 Dec;57(8):906-13
Expansive gene transfer in the rat CNS rapidly produces amyotrophic lateral sclerosis relevant sequelae when TDP-43 is overexpressed.
Wang DB, Dayton RD, Henning PP, Cain CD, Zhao LR, Schrott LM, Orchard EA, Knight DS, Klein RL
Molecular therapy : the journal of the American Society of Gene Therapy 2010 Dec;18(12):2064-74
Molecular therapy : the journal of the American Society of Gene Therapy 2010 Dec;18(12):2064-74
Neurotoxic effects of TDP-43 overexpression in C. elegans.
Ash PE, Zhang YJ, Roberts CM, Saldi T, Hutter H, Buratti E, Petrucelli L, Link CD
Human molecular genetics 2010 Aug 15;19(16):3206-18
Human molecular genetics 2010 Aug 15;19(16):3206-18
Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice.
Xu YF, Gendron TF, Zhang YJ, Lin WL, D'Alton S, Sheng H, Casey MC, Tong J, Knight J, Yu X, Rademakers R, Boylan K, Hutton M, McGowan E, Dickson DW, Lewis J, Petrucelli L
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Aug 11;30(32):10851-9
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Aug 11;30(32):10851-9
Alteration of biochemical and pathological properties of TDP-43 protein by a lipid mediator, 15-deoxy-Delta(12,14)-prostaglandin J(2).
Zhang HX, Tanji K, Yoshida H, Hayakari M, Shibata T, Mori F, Uchida K, Wakabayashi K
Experimental neurology 2010 Apr;222(2):296-303
Experimental neurology 2010 Apr;222(2):296-303
Plasma phosphorylated-TDP-43 protein levels correlate with brain pathology in frontotemporal lobar degeneration.
Foulds PG, Davidson Y, Mishra M, Hobson DJ, Humphreys KM, Taylor M, Johnson N, Weintraub S, Akiyama H, Arai T, Hasegawa M, Bigio EH, Benson FE, Allsop D, Mann DM
Acta neuropathologica 2009 Nov;118(5):647-58
Acta neuropathologica 2009 Nov;118(5):647-58
Cytosolic TDP-43 expression following axotomy is associated with caspase 3 activation in NFL-/- mice: support for a role for TDP-43 in the physiological response to neuronal injury.
Moisse K, Mepham J, Volkening K, Welch I, Hill T, Strong MJ
Brain research 2009 Nov 3;1296:176-86
Brain research 2009 Nov 3;1296:176-86
The complement factor C5a receptor is upregulated in NFL-/- mouse motor neurons.
Humayun S, Gohar M, Volkening K, Moisse K, Leystra-Lantz C, Mepham J, McLean J, Strong MJ
Journal of neuroimmunology 2009 May 29;210(1-2):52-62
Journal of neuroimmunology 2009 May 29;210(1-2):52-62
VCP mutations causing frontotemporal lobar degeneration disrupt localization of TDP-43 and induce cell death.
Gitcho MA, Strider J, Carter D, Taylor-Reinwald L, Forman MS, Goate AM, Cairns NJ
The Journal of biological chemistry 2009 May 1;284(18):12384-98
The Journal of biological chemistry 2009 May 1;284(18):12384-98
TAR DNA-Binding protein 43 accumulation in protein aggregate myopathies.
Olivé M, Janué A, Moreno D, Gámez J, Torrejón-Escribano B, Ferrer I
Journal of neuropathology and experimental neurology 2009 Mar;68(3):262-73
Journal of neuropathology and experimental neurology 2009 Mar;68(3):262-73
Expression of TDP-43 C-terminal Fragments in Vitro Recapitulates Pathological Features of TDP-43 Proteinopathies.
Igaz LM, Kwong LK, Chen-Plotkin A, Winton MJ, Unger TL, Xu Y, Neumann M, Trojanowski JQ, Lee VM
The Journal of biological chemistry 2009 Mar 27;284(13):8516-24
The Journal of biological chemistry 2009 Mar 27;284(13):8516-24
Early-onset familial lewy body dementia with extensive tauopathy: a clinical, genetic, and neuropathological study.
Clarimón J, Molina-Porcel L, Gómez-Isla T, Blesa R, Guardia-Laguarta C, González-Neira A, Estorch M, Ma Grau J, Barraquer L, Roig C, Ferrer I, Lleó A
Journal of neuropathology and experimental neurology 2009 Jan;68(1):73-82
Journal of neuropathology and experimental neurology 2009 Jan;68(1):73-82
Increased TDP-43 protein in cerebrospinal fluid of patients with amyotrophic lateral sclerosis.
Kasai T, Tokuda T, Ishigami N, Sasayama H, Foulds P, Mitchell DJ, Mann DM, Allsop D, Nakagawa M
Acta neuropathologica 2009 Jan;117(1):55-62
Acta neuropathologica 2009 Jan;117(1):55-62
Sporadic amyotrophic lateral sclerosis of long duration is associated with relatively mild TDP-43 pathology.
Nishihira Y, Tan CF, Hoshi Y, Iwanaga K, Yamada M, Kawachi I, Tsujihata M, Hozumi I, Morita T, Onodera O, Nishizawa M, Kakita A, Takahashi H
Acta neuropathologica 2009 Jan;117(1):45-53
Acta neuropathologica 2009 Jan;117(1):45-53
Expression of collagen XVII and ubiquitin-binding protein p62 in motor neuron disease.
Seppänen A, Pikkarainen M, Hartikainen P, Hofmann SC, Majamaa K, Alafuzoff I
Brain research 2009 Jan 9;1247:171-7
Brain research 2009 Jan 9;1247:171-7
Divergent patterns of cytosolic TDP-43 and neuronal progranulin expression following axotomy: implications for TDP-43 in the physiological response to neuronal injury.
Moisse K, Volkening K, Leystra-Lantz C, Welch I, Hill T, Strong MJ
Brain research 2009 Jan 16;1249:202-11
Brain research 2009 Jan 16;1249:202-11
Frontotemporal dementia in a large Swedish family is caused by a progranulin null mutation.
Skoglund L, Brundin R, Olofsson T, Kalimo H, Ingvast S, Blom ES, Giedraitis V, Ingelsson M, Lannfelt L, Basun H, Glaser A
Neurogenetics 2009 Feb;10(1):27-34
Neurogenetics 2009 Feb;10(1):27-34
Abnormal hippocampal distribution of TDP-43 in patients with-late onset psychosis.
Velakoulis D, Walterfang M, Mocellin R, Pantelis C, Dean B, McLean C
The Australian and New Zealand journal of psychiatry 2009 Aug;43(8):739-45
The Australian and New Zealand journal of psychiatry 2009 Aug;43(8):739-45
Ubiquitin associated protein 1 is a risk factor for frontotemporal lobar degeneration.
Rollinson S, Rizzu P, Sikkink S, Baker M, Halliwell N, Snowden J, Traynor BJ, Ruano D, Cairns N, Rohrer JD, Mead S, Collinge J, Rossor M, Akay E, Guerreiro R, Rademakers R, Morrison KE, Pastor P, Alonso E, Martinez-Lage P, Graff-Radford N, Neary D, Heutink P, Mann DM, Van Swieten J, Pickering-Brown SM
Neurobiology of aging 2009 Apr;30(4):656-65
Neurobiology of aging 2009 Apr;30(4):656-65
Frontotemporal dementia presenting as schizophrenia-like psychosis in young people: clinicopathological series and review of cases.
Velakoulis D, Walterfang M, Mocellin R, Pantelis C, McLean C
The British journal of psychiatry : the journal of mental science 2009 Apr;194(4):298-305
The British journal of psychiatry : the journal of mental science 2009 Apr;194(4):298-305
TDP-43 accumulation in inclusion body myopathy muscle suggests a common pathogenic mechanism with frontotemporal dementia.
Weihl CC, Temiz P, Miller SE, Watts G, Smith C, Forman M, Hanson PI, Kimonis V, Pestronk A
Journal of neurology, neurosurgery, and psychiatry 2008 Oct;79(10):1186-9
Journal of neurology, neurosurgery, and psychiatry 2008 Oct;79(10):1186-9
White matter tauopathy with globular glial inclusions: a distinct sporadic frontotemporal lobar degeneration.
Kovacs GG, Majtenyi K, Spina S, Murrell JR, Gelpi E, Hoftberger R, Fraser G, Crowther RA, Goedert M, Budka H, Ghetti B
Journal of neuropathology and experimental neurology 2008 Oct;67(10):963-75
Journal of neuropathology and experimental neurology 2008 Oct;67(10):963-75
TDP-43 in cerebrospinal fluid of patients with frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
Steinacker P, Hendrich C, Sperfeld AD, Jesse S, von Arnim CA, Lehnert S, Pabst A, Uttner I, Tumani H, Lee VM, Trojanowski JQ, Kretzschmar HA, Ludolph A, Neumann M, Otto M
Archives of neurology 2008 Nov;65(11):1481-7
Archives of neurology 2008 Nov;65(11):1481-7
Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation.
Winton MJ, Igaz LM, Wong MM, Kwong LK, Trojanowski JQ, Lee VM
The Journal of biological chemistry 2008 May 9;283(19):13302-9
The Journal of biological chemistry 2008 May 9;283(19):13302-9
A distinct clinical, neuropsychological and radiological phenotype is associated with progranulin gene mutations in a large UK series.
Beck J, Rohrer JD, Campbell T, Isaacs A, Morrison KE, Goodall EF, Warrington EK, Stevens J, Revesz T, Holton J, Al-Sarraj S, King A, Scahill R, Warren JD, Fox NC, Rossor MN, Collinge J, Mead S
Brain : a journal of neurology 2008 Mar;131(Pt 3):706-20
Brain : a journal of neurology 2008 Mar;131(Pt 3):706-20
Epitope mapping of 2E2-D3, a monoclonal antibody directed against human TDP-43.
Zhang HX, Tanji K, Mori F, Wakabayashi K
Neuroscience letters 2008 Mar 28;434(2):170-4
Neuroscience letters 2008 Mar 28;434(2):170-4
Molecular pathogenesis of frontotemporal lobar degeneration: basic science seminar in neurology.
Sleegers K, Kumar-Singh S, Cruts M, Van Broeckhoven C
Archives of neurology 2008 Jun;65(6):700-4
Archives of neurology 2008 Jun;65(6):700-4
Enrichment of C-terminal fragments in TAR DNA-binding protein-43 cytoplasmic inclusions in brain but not in spinal cord of frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
Igaz LM, Kwong LK, Xu Y, Truax AC, Uryu K, Neumann M, Clark CM, Elman LB, Miller BL, Grossman M, McCluskey LF, Trojanowski JQ, Lee VM
The American journal of pathology 2008 Jul;173(1):182-94
The American journal of pathology 2008 Jul;173(1):182-94
MAPT S305I mutation: implications for argyrophilic grain disease.
Kovacs GG, Pittman A, Revesz T, Luk C, Lees A, Kiss E, Tariska P, Laszlo L, Molnár K, Molnar MJ, Tolnay M, de Silva R
Acta neuropathologica 2008 Jul;116(1):103-18
Acta neuropathologica 2008 Jul;116(1):103-18
TDP-43-immunoreactive neuronal and glial inclusions in the neostriatum in amyotrophic lateral sclerosis with and without dementia.
Zhang H, Tan CF, Mori F, Tanji K, Kakita A, Takahashi H, Wakabayashi K
Acta neuropathologica 2008 Jan;115(1):115-22
Acta neuropathologica 2008 Jan;115(1):115-22
Missense mutations in the progranulin gene linked to frontotemporal lobar degeneration with ubiquitin-immunoreactive inclusions reduce progranulin production and secretion.
Shankaran SS, Capell A, Hruscha AT, Fellerer K, Neumann M, Schmid B, Haass C
The Journal of biological chemistry 2008 Jan 18;283(3):1744-53
The Journal of biological chemistry 2008 Jan 18;283(3):1744-53
Colocalization of transactivation-responsive DNA-binding protein 43 and huntingtin in inclusions of Huntington disease.
Schwab C, Arai T, Hasegawa M, Yu S, McGeer PL
Journal of neuropathology and experimental neurology 2008 Dec;67(12):1159-65
Journal of neuropathology and experimental neurology 2008 Dec;67(12):1159-65
Enduring involvement of tau, beta-amyloid, alpha-synuclein, ubiquitin and TDP-43 pathology in the amyotrophic lateral sclerosis/parkinsonism-dementia complex of Guam (ALS/PDC).
Miklossy J, Steele JC, Yu S, McCall S, Sandberg G, McGeer EG, McGeer PL
Acta neuropathologica 2008 Dec;116(6):625-37
Acta neuropathologica 2008 Dec;116(6):625-37
Ultrastructural localization of TDP-43 in filamentous neuronal inclusions in various neurodegenerative diseases.
Lin WL, Dickson DW
Acta neuropathologica 2008 Aug;116(2):205-13
Acta neuropathologica 2008 Aug;116(2):205-13
Maturation process of TDP-43-positive neuronal cytoplasmic inclusions in amyotrophic lateral sclerosis with and without dementia.
Mori F, Tanji K, Zhang HX, Nishihira Y, Tan CF, Takahashi H, Wakabayashi K
Acta neuropathologica 2008 Aug;116(2):193-203
Acta neuropathologica 2008 Aug;116(2):193-203
TDP-43 protein in plasma may index TDP-43 brain pathology in Alzheimer's disease and frontotemporal lobar degeneration.
Foulds P, McAuley E, Gibbons L, Davidson Y, Pickering-Brown SM, Neary D, Snowden JS, Allsop D, Mann DM
Acta neuropathologica 2008 Aug;116(2):141-6
Acta neuropathologica 2008 Aug;116(2):141-6
A yeast TDP-43 proteinopathy model: Exploring the molecular determinants of TDP-43 aggregation and cellular toxicity.
Johnson BS, McCaffery JM, Lindquist S, Gitler AD
Proceedings of the National Academy of Sciences of the United States of America 2008 Apr 29;105(17):6439-44
Proceedings of the National Academy of Sciences of the United States of America 2008 Apr 29;105(17):6439-44
DEAD-box RNA helicase subunits of the Drosha complex are required for processing of rRNA and a subset of microRNAs.
Fukuda T, Yamagata K, Fujiyama S, Matsumoto T, Koshida I, Yoshimura K, Mihara M, Naitou M, Endoh H, Nakamura T, Akimoto C, Yamamoto Y, Katagiri T, Foulds C, Takezawa S, Kitagawa H, Takeyama K, O'Malley BW, Kato S
Nature cell biology 2007 May;9(5):604-11
Nature cell biology 2007 May;9(5):604-11
TDP-43 immunoreactivity in hippocampal sclerosis and Alzheimer's disease.
Amador-Ortiz C, Lin WL, Ahmed Z, Personett D, Davies P, Duara R, Graff-Radford NR, Hutton ML, Dickson DW
Annals of neurology 2007 May;61(5):435-45
Annals of neurology 2007 May;61(5):435-45
Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations.
Mackenzie IR, Bigio EH, Ince PG, Geser F, Neumann M, Cairns NJ, Kwong LK, Forman MS, Ravits J, Stewart H, Eisen A, McClusky L, Kretzschmar HA, Monoranu CM, Highley JR, Kirby J, Siddique T, Shaw PJ, Lee VM, Trojanowski JQ
Annals of neurology 2007 May;61(5):427-34
Annals of neurology 2007 May;61(5):427-34
TDP-43 is deposited in the Guam parkinsonism-dementia complex brains.
Hasegawa M, Arai T, Akiyama H, Nonaka T, Mori H, Hashimoto T, Yamazaki M, Oyanagi K
Brain : a journal of neurology 2007 May;130(Pt 5):1386-94
Brain : a journal of neurology 2007 May;130(Pt 5):1386-94
TDP-43 immunoreactivity in neuronal inclusions in familial amyotrophic lateral sclerosis with or without SOD1 gene mutation.
Tan CF, Eguchi H, Tagawa A, Onodera O, Iwasaki T, Tsujino A, Nishizawa M, Kakita A, Takahashi H
Acta neuropathologica 2007 May;113(5):535-42
Acta neuropathologica 2007 May;113(5):535-42
TDP-43-positive white matter pathology in frontotemporal lobar degeneration with ubiquitin-positive inclusions.
Neumann M, Kwong LK, Truax AC, Vanmassenhove B, Kretzschmar HA, Van Deerlin VM, Clark CM, Grossman M, Miller BL, Trojanowski JQ, Lee VM
Journal of neuropathology and experimental neurology 2007 Mar;66(3):177-83
Journal of neuropathology and experimental neurology 2007 Mar;66(3):177-83
TDP-43 in the ubiquitin pathology of frontotemporal dementia with VCP gene mutations.
Neumann M, Mackenzie IR, Cairns NJ, Boyer PJ, Markesbery WR, Smith CD, Taylor JP, Kretzschmar HA, Kimonis VE, Forman MS
Journal of neuropathology and experimental neurology 2007 Feb;66(2):152-7
Journal of neuropathology and experimental neurology 2007 Feb;66(2):152-7
Evidence that TDP-43 is not the major ubiquitinated target within the pathological inclusions of amyotrophic lateral sclerosis.
Sanelli T, Xiao S, Horne P, Bilbao J, Zinman L, Robertson J
Journal of neuropathology and experimental neurology 2007 Dec;66(12):1147-53
Journal of neuropathology and experimental neurology 2007 Dec;66(12):1147-53
TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis.
Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H, Mann D, Tsuchiya K, Yoshida M, Hashizume Y, Oda T
Biochemical and biophysical research communications 2006 Dec 22;351(3):602-11
Biochemical and biophysical research communications 2006 Dec 22;351(3):602-11
No comments: Submit comment
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
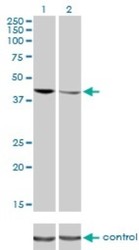
- Experimental details
- Western Blot: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Analysis of TARDBP over-expressed 293 cell line, cotransfected with TARDBP Validated Chimera RNAi ( Cat # H00023435-R01V ) (Lane 2) or non-transfected control (Lane 1). Blot probed with TARDBP monoclonal antibody (M01), clone 2E2-D3 (Cat # H00023435-M01 ). GAPDH ( 36.1 kDa ) used as specificity and loading control.
- Submitted by
- Novus Biologicals (provider)
- Main image
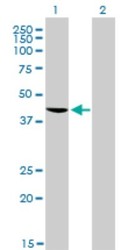
- Experimental details
- Western Blot: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Analysis of TARDBP expression in transfected 293T cell line by TARDBP monoclonal antibody (M01), clone 2E2-D3.Lane 1: TARDBP transfected lysate(44.7 KDa).Lane 2: Non-transfected lysate.
- Submitted by
- Novus Biologicals (provider)
- Main image
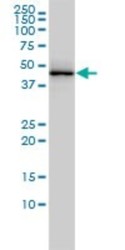
- Experimental details
- Western Blot: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - TARDBP monoclonal antibody (M01), clone 2E2-D3 Analysis of TARDBP expression in A-431.
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
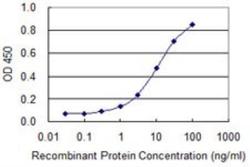
- Experimental details
- ELISA: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Detection limit for recombinant GST tagged TARDBP is 0.3 ng/ml as a capture antibody.
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
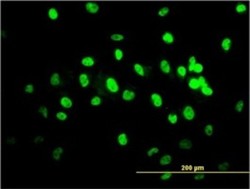
- Experimental details
- Immunocytochemistry/Immunofluorescence: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Analysis of monoclonal antibody to TARDBP on HeLa cell. Antibody concentration 10 ug/ml.
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
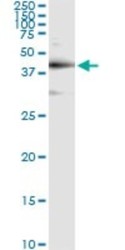
- Experimental details
- Immunoprecipitation: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Analysis of TARDBP transfected lysate using anti-TARDBP monoclonal antibody and Protein A Magnetic Bead, and immunoblotted with TARDBP MaxPab rabbit polyclonal antibody.
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
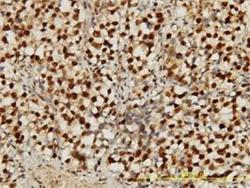
- Experimental details
- Immunohistochemistry-Paraffin: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - TARDBP on formalin-fixed paraffin-embedded human leiomyosarcoma. [antibody concentration 3 ug/ml]
- Submitted by
- Novus Biologicals (provider)
- Main image
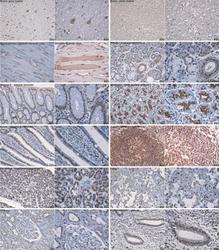
- Experimental details
- Immunohistochemistry-Paraffin: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - TARDBP in rat tissue samples. Image courtsey of product review by Daniel Pirici.
- Submitted by
- Novus Biologicals (provider)
- Main image
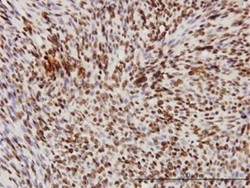
- Experimental details
- Immunohistochemistry-Paraffin: TDP-43/TARDBP Antibody (2E2-D3) [H00023435-M01] - Analysis of monoclonal antibody to TARDBP on formalin-fixed paraffin-embedded human leiomyosarcoma tissue Antibody concentration 3 ug/ml.
 Explore
Explore Validate
Validate Learn
Learn