Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-50606 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ARFRP1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- The antibody detects endogenous levels of total ARFRP1 protein.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 3.3 mg/mL
- Storage
- -20°C
Submitted references ARFRP1 functions upstream of ARL1 and ARL5 to coordinate recruitment of distinct tethering factors to the trans-Golgi network.
Ishida M, Bonifacino JS
The Journal of cell biology 2019 Nov 4;218(11):3681-3696
The Journal of cell biology 2019 Nov 4;218(11):3681-3696
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
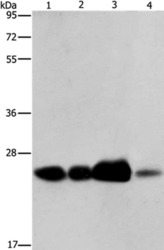
- Experimental details
- Western blot analysis of ARFRP1 was performed by loading (from left to right): 293T cell and human fetal liver tissue, human cervical cancer and fetal muscle tissue lysates (40µg) on to a 8% SDS-PAGE gel. Proteins were transferred to a membrane and the membrane was probed with a ARFRP1 antibody (Product # PA5-50606) at a 1/800 dilution for 40 seconds, followed by a secondary antibody at 1/8000.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
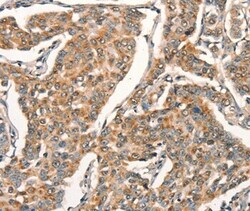
- Experimental details
- Immunohistochemical analysis of ARFRP1 in paraffin embedded Human breast cancer tissue using ARFRP1 Polyclonal Antibody (Product # PA5-50606) at a 1:25 dilution.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
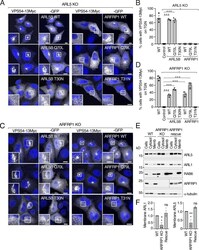
- Figure 2. Both ARL5 and ARFRP1 are required for localization of GARP to the TGN. (A) KO of TGN-localized small GTPases in HeLa cells confirmed by immunoblot analysis with antibodies to the proteins indicated on the right. In this figure and subsequent figures, ARL5 KO represents KO of both ARL5A and ARL5B, and RAB6 KO represents KO of both RAB6A and RAB6B. alpha-Tubulin was used as a loading control. The positions of molecular mass markers are indicated on the left. (B) Immunofluorescence microscopy of WT and KO HeLa cells transfected with a plasmid encoding VPS54-13Myc and stained for the Myc epitope (red), giantin (green), and nuclei (DAPI; blue). Scale bars: 10 mum. Insets are magnified views of the boxed areas. Inset scale bars: 5 mum. (C) Quantification of the percentage of cells exhibiting VPS54-13Myc staining at the TGN. Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. The statistical significance of the differences relative to WT cells was determined using Dunnett's test. **, P < 0.01; ***, P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
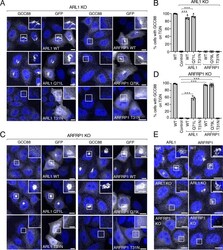
- Figure 3. ARFRP1 functions upstream of ARL5 for the recruitment of GARP to the TGN. (A) GFP-tagged ARL5B (WT, Q70L, or T30N) or ARFRP1 (WT, Q79L, or T31N) were co-expressed with VPS54-13Myc in ARL5-KO cells. Cells were stained with DAPI (blue) and examined for GFP distribution by confocal microscopy. Scale bars: 10 mum. Insets are magnified views of the boxed areas. Inset scale bars: 5 mum. (B) The percentage of cells with VPS54-13Myc staining at the TGN was quantified as described in Fig. 2 C . Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. ***, P < 0.001, in comparison to ARL5 KO cells only expressing VPS54-13Myc (control) using Dunnett's test. (C) GFP-tagged ARL5B (WT, Q70L, or T30N) or ARFRP1 (WT, Q79L, or T31N) were co-expressed with VPS54-13Myc in ARFRP1-KO cells. Cells were analyzed as in A. (D) Quantification of cells having VPS54-13Myc staining at the TGN as described in Fig. 2 C . Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. ***, P < 0.001, in comparison to ARFRP1 KO cells only expressing VPS54-13Myc (control) using Dunnett's test. (E) Subcellular fractionation of WT, ARFRP1-KO, and ARFRP1-KO-rescue HeLa cells. Whole cells and cytosolic and membrane (Memb) fractions obtained as described in Materials and methods were analyzed by SDS-PAGE and immunoblotting for the proteins indicated on the right. The positi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6. ARFRP1 functions upstream of ARL1 in the recruitment of GCC88 to the TGN. (A) ARL1-KO cells were transfected with plasmids encoding GFP-tagged ARL1 (WT, Q71L, or T31N) or GFP-tagged ARFRP1 (WT, Q79L, or T31N), immunostained for endogenous GCC88, counterstained with DAPI (blue), and imaged by confocal microscopy. (B) The percentage of cells having GCC88 staining at the TGN from experiments such as that in panel A was quantified as described in Fig. 2 C . Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. ***, P < 0.001, in comparison to untransfected ARL1 KO cells using Dunnett's test. (C) ARFRP1-KO cells were transfected and analyzed as described in A. (D) The percentage of cells having GCC88 staining at the TGN from experiments such as that in C was quantified as described in Fig. 2 C . Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. ***, P < 0.001, in comparison to untransfected ARFRP1 KO cells using Dunnett's test. (E) WT, ARL1-KO, or ARFRP1-KO cells were immunostained for endogenous ARL1 or ARFRP1, counterstained with DAPI (blue), and imaged by confocal microscopy. Scale bars: 10 mum. Insets are magnified views of the boxed areas. Inset scale bars: 5 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7. SYS1 is required for the association of ARFRP1, ARL1, GCC88, and GARP with the TGN. (A) Due to the lack of good antibodies to SYS1, KO of the SYS1 gene was demonstrated by Sanger sequencing (top) and genomic PCR (bottom). KO resulted in a 34- and 36-bp deletion. The 34-bp deletion is predicted to result in a frameshift causing premature termination at amino acid 28. The 36-bp deletion is not predicted to result in a frameshift, but to cause a deletion of more than half of the first transmembrane domain. (B) WT and SYS1-KO cells were immunostained for endogenous ARL1 or ARFRP1, counterstained with DAPI (blue), and imaged by confocal microscopy. (C) SYS1-KO cells were co-transfected with plasmids encoding VPS54-13Myc and either GFP alone or SYS1-GFP as indicated in the figure, immunostained for the Myc epitope, counterstained with DAPI (blue), and imaged by confocal microscopy. Insets are magnified views of the boxed areas. Inset scale bars: 5 mum. (D) SYS1-KO cells were transfected with plasmids encoding GFP or SYS1-GFP as indicated in the figure, immunostained for endogenous GCC88, counterstained with DAPI (blue), and imaged by confocal microscopy. Scale bars in B, C, and D: 10 mum. (E) Quantification of cells having VPS54-13Myc staining at the TGN as described in Fig. 2 C . Values are the mean +- SEM from three independent experiments. More than 100 cells per sample were counted in each experiment. (F) Quantification of cells having GCC88 staining at the TGN as des
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot