Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Immunohistochemistry [2]
- Other assay [23]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 34-4200 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Connexin 29 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- 34-4200 was used in the IHC analysis to successfully detect Connexin 29 in mouse sciatic nerve.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 50 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Differential expression of astrocytic connexins in a mouse model of prenatal alcohol exposure.
The oligodendroglial precursor cell line Oli-neu represents a cell culture system to examine functional expression of the mouse gap junction gene connexin29 (Cx29).
Connexin-47 and connexin-32 in gap junctions of oligodendrocyte somata, myelin sheaths, paranodal loops and Schmidt-Lanterman incisures: implications for ionic homeostasis and potassium siphoning.
Adherens junctions in myelinating Schwann cells stabilize Schmidt-Lanterman incisures via recruitment of p120 catenin to E-cadherin.
Connexin47, connexin29 and connexin32 co-expression in oligodendrocytes and Cx47 association with zonula occludens-1 (ZO-1) in mouse brain.
Connexin29 and connexin32 at oligodendrocyte and astrocyte gap junctions and in myelin of the mouse central nervous system.
Coupling of astrocyte connexins Cx26, Cx30, Cx43 to oligodendrocyte Cx29, Cx32, Cx47: Implications from normal and connexin32 knockout mice.
Connexin29 expression, immunocytochemistry and freeze-fracture replica immunogold labelling (FRIL) in sciatic nerve.
Ramani M, Mylvaganam S, Krawczyk M, Wang L, Zoidl C, Brien J, Reynolds JN, Kapur B, Poulter MO, Zoidl G, Carlen PL
Neurobiology of disease 2016 Jul;91:83-93
Neurobiology of disease 2016 Jul;91:83-93
The oligodendroglial precursor cell line Oli-neu represents a cell culture system to examine functional expression of the mouse gap junction gene connexin29 (Cx29).
Söhl G, Hombach S, Degen J, Odermatt B
Frontiers in pharmacology 2013;4:83
Frontiers in pharmacology 2013;4:83
Connexin-47 and connexin-32 in gap junctions of oligodendrocyte somata, myelin sheaths, paranodal loops and Schmidt-Lanterman incisures: implications for ionic homeostasis and potassium siphoning.
Kamasawa N, Sik A, Morita M, Yasumura T, Davidson KG, Nagy JI, Rash JE
Neuroscience 2005;136(1):65-86
Neuroscience 2005;136(1):65-86
Adherens junctions in myelinating Schwann cells stabilize Schmidt-Lanterman incisures via recruitment of p120 catenin to E-cadherin.
Tricaud N, Perrin-Tricaud C, Brusés JL, Rutishauser U
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Mar 30;25(13):3259-69
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Mar 30;25(13):3259-69
Connexin47, connexin29 and connexin32 co-expression in oligodendrocytes and Cx47 association with zonula occludens-1 (ZO-1) in mouse brain.
Li X, Ionescu AV, Lynn BD, Lu S, Kamasawa N, Morita M, Davidson KG, Yasumura T, Rash JE, Nagy JI
Neuroscience 2004;126(3):611-30
Neuroscience 2004;126(3):611-30
Connexin29 and connexin32 at oligodendrocyte and astrocyte gap junctions and in myelin of the mouse central nervous system.
Nagy JI, Ionescu AV, Lynn BD, Rash JE
The Journal of comparative neurology 2003 Sep 22;464(3):356-70
The Journal of comparative neurology 2003 Sep 22;464(3):356-70
Coupling of astrocyte connexins Cx26, Cx30, Cx43 to oligodendrocyte Cx29, Cx32, Cx47: Implications from normal and connexin32 knockout mice.
Nagy JI, Ionescu AV, Lynn BD, Rash JE
Glia 2003 Dec;44(3):205-18
Glia 2003 Dec;44(3):205-18
Connexin29 expression, immunocytochemistry and freeze-fracture replica immunogold labelling (FRIL) in sciatic nerve.
Li X, Lynn BD, Olson C, Meier C, Davidson KG, Yasumura T, Rash JE, Nagy JI
The European journal of neuroscience 2002 Sep;16(5):795-806
The European journal of neuroscience 2002 Sep;16(5):795-806
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
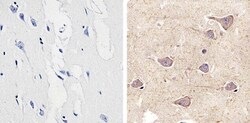
- Experimental details
- Immunohistochemistry analysis of Connexin 29 showing staining in the cytoplasm and membrane of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Connexin 29 polyclonal antibody (Product # 34-4200) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
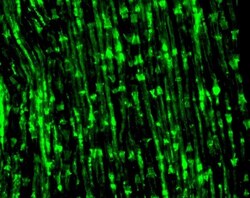
- Experimental details
- Immunofluorescence analysis of Connexin29 (Cx29) was performed on sections of adult mouse sciatic nerve. Tissue sections on slides were probed for 24 h at 4°C in a humidified chamber with rabbit polyclonal anti-Cx29 (Product # 34-4200) at an antibody concentration of 3 µg/mL diluted in 50 mM Tris-HCl, pH 7.4, containing 1.5% NaCl, 0.3% Triton X-100 (TBST) and 4% normal goat serum. After overnight incubation, sections were washed extensively for 1 h in TBST, and detection of primary antibody was performed for 1.5 h at room temperature with AlexaFluor-488-conjugated donkey anti-rabbit diluted 1:600 in TBST. Sections were then washed in TBST, then in TBS (without triton) and then coversliped with anti-fade medium. Images were taken on a Zeiss 710 confocal microscope at x40 objective magnification, and show immunofluorescence labelling of Cx29 localized along myelinated axon in adult mouse sciatic nerve, including nodal and internodal regions, and at Scmidt-Lanterman incisures. Data courtesy of Dr. James Nagy's lab.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
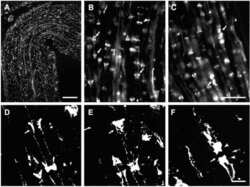
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
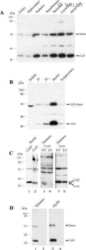
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
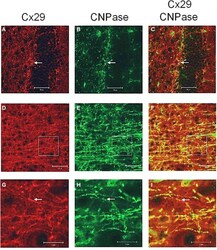
- Figure A1 Laser scan microscopy immunofluorescence analyses of Cx29 protein expression in the hippocampus (A-C) and in the neocortex (D-I) of the adult mouse brain. Pictures (G-I) represent enlargements of the white squares present in pictures (D-F) , respectively. Cx29 immunofluorescence signals (A,D,G) co-localize with myelin-associated CNPase (2', 3'-cyclic nucleotide 3'-phosphodiesterase). CNPase is present in the plasma membrane of oligodendrocytes and their processes, in periaxonal membranes and the inner mesaxons, the outer processes, the paranodal myelin loops, and the ""incisure-like"" membranes (B,E,H) . Merged pictures are (C,F,I) . Arrows in (A,B,C) indicate co-localization of Cx29 and CNPase in the soma of an oligodendrocyte. Arrows in (G,H,I) point to a co-localization of Cx29 and CNPase in myelinated fibers. However, Cx29 is hardly co-localized but present in the same neural structures as the myelin associated CNPase. This expression profile strengthened the credibility of the used Cx29 antibodies (Zymed), which was tested on tissue sections of Cx29 (-/-) deficient mice (Eiberger et al., 2006 ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure A2 Cx29 immunofluorescence analyses and microinjections of undifferentiated Oli-neu cells. (A) Distribution of the Cx29 protein mostly in plasma membranes (arrow) applying Cx29-specific and Cy3 coupled secondary antibodies in relation to the cellular nuclei after DAPI staining. (B) Omitting primary Cx29 antibodies excluded cross reactivity of Cy3 antibodies with Oli-neu cells. (C,D) Two examples of Oli-neu cells microinjected with neurobiotin and stained thereafter (arrow). (C) After filling one cell (large arrow), neurobiotin readily spreads into the various protrusions but did not pour into the attached cells at the ends (small arrows). (D) No neurobiotin transfer into the next neighboring could be demonstrated.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Northern blot analysis of total RNA from HeLa, PC12, Oli-neu cells, and mouse adult brain. (A) A 4.4 kb signal representing Cx29 expression was detected highly abundant in undifferentiated as well as differentiated Oli-neu cells and weakly in brain. A 1.6 kb signal of Cx32 expression is distinctly visible in RNA from adult brain (100%) as control but is hardly detectable (3 vs. 14%) in Oli-neu cells (diff. and undif.), respectively. Cx47 expression (2.5 kb) is also evident in adult brain but missing in Oli-neu cells. No signals of oligodendroglial connexins were seen in HeLa wild type (wt), HeLa Cx45 transfectants, and PC12 cells (diff. or undif.). Additional hybridization signals at about 10 kb are visible after hybridizing the Cx29 probe against RNA of Oli-neu cells as well as from adult brain. HeLa cells stably transfected with the tandem cloned connexin29 and eGFP reading frames (both of ~800 bp) either separated by a stop-codon or directly fused, yielded hybridization signals of the expected 1.8 kb. The blot was standardized by measuring the intensities of both the Etbr stained 18S- and 28S-rRNA. All signals have been documented after 3 weeks of exposure. (B) Immonoblot analysis of Cx29 and Cx29-eGFP stably transfected HeLa cells and Oli-neu cells (dif. and undif.) using the Cx29 polyclonal antibodies (Zymed). Two signals of about 30 and 56 kDa were prominent in Cx29 HeLa cells and Oli-neu cells. In the Cx29-eGFP HeLa cells, however, both signals seemed to be sh
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Cx29 immunofluorescence analyses and microinjections of differentiated Oli-neu cells. (A) Distribution of the Cx29 protein mostly in plasma membranes (arrows) applying Cx29-specific and Cy3 coupled secondary antibodies in relation to the cellular nuclei after DAPI staining. (B) Omitting primary Cx29 antibodies excluded cross reactivity of Cy3 antibodies with Oli-neu cells. ( C and D ) Two examples of Oli-neu cells microinjected with neurobiotin and stained thereafter (arrow) (C) No neurobiotin transfer into both cells directly attached below was detectable. (D) After filling one cell (large arrow), neurobiotin readily spreads into the three protrusions but did not migrate further into adjacent cells at their ends (small arrows). (E) Combined UV light- and trans-illumination identified the Etbr-microinjected Oli-neu cell (arrow). (F) Only the Etbr filled soma and the protrusions of the injected cell are faintly visible after 7 min (arrows).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry