Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [2]
- Immunohistochemistry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- ABIN393234 - Provider product page

- Provider
- antibodies-online
- Product name
- anti-Crystallin, gamma D (CRYGD) (AA 75-175) antibody
- Antibody type
- Monoclonal
- Description
- This antibody is purified through a protein G column, eluted with high and low pH buffers and neutralized immediately, followed by dialysis against PBS.
- Reactivity
- Human
- Host
- Mouse
- Epitope
- AA 75-175
- Isotype
- IgG
- Antibody clone number
- 4E12
- Vial size
- 100 μg
- Storage
- Maintain refrigerated at 2-8°C for up to 6 months. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles
Submitted references A novel human CRYGD mutation in a juvenile autosomal dominant cataract.
Increase in surface hydrophobicity of the cataract-associated P23T mutant of human gammaD-crystallin is responsible for its dramatically lower, retrograde solubility.
beta-Strand interactions at the domain interface critical for the stability of human lens gammaD-crystallin.
Partially folded aggregation intermediates of human gammaD-, gammaC-, and gammaS-crystallin are recognized and bound by human alphaB-crystallin chaperone.
Hydrophobic core mutations associated with cataract development in mice destabilize human gammaD-crystallin.
Roshan M, Vijaya PH, Lavanya GR, Shama PK, Santhiya ST, Graw J, Gopinath PM, Satyamoorthy K
Molecular vision 2010 May 22;16:887-96
Molecular vision 2010 May 22;16:887-96
Increase in surface hydrophobicity of the cataract-associated P23T mutant of human gammaD-crystallin is responsible for its dramatically lower, retrograde solubility.
Pande A, Ghosh KS, Banerjee PR, Pande J
Biochemistry 2010 Jul 27;49(29):6122-9
Biochemistry 2010 Jul 27;49(29):6122-9
beta-Strand interactions at the domain interface critical for the stability of human lens gammaD-crystallin.
Das P, King JA, Zhou R
Protein science : a publication of the Protein Society 2010 Jan;19(1):131-40
Protein science : a publication of the Protein Society 2010 Jan;19(1):131-40
Partially folded aggregation intermediates of human gammaD-, gammaC-, and gammaS-crystallin are recognized and bound by human alphaB-crystallin chaperone.
Acosta-Sampson L, King J
Journal of molecular biology 2010 Aug 6;401(1):134-52
Journal of molecular biology 2010 Aug 6;401(1):134-52
Hydrophobic core mutations associated with cataract development in mice destabilize human gammaD-crystallin.
Moreau KL, King J
The Journal of biological chemistry 2009 Nov 27;284(48):33285-95
The Journal of biological chemistry 2009 Nov 27;284(48):33285-95
No comments: Submit comment
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
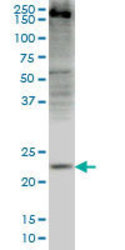
- Experimental details
- WB
- Submitted by
- antibodies-online (provider)
- Main image
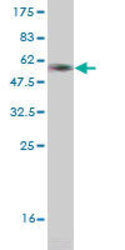
- Experimental details
- WB
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
- Experimental details
- IHC
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA