Antibody data
- Antibody Data
- Antigen structure
- References [38]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Immunohistochemistry [1]
- Other assay [28]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 32-0500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NMDAR1 Monoclonal Antibody (54.1)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This antibody specifically recognizes the ~103 kDa NMDAR1 protein. Cross-reactivity with other NMDA receptor proteins has not been observed.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 54.1
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Molecular Mechanisms Underlying the Retrieval and Extinction of Morphine Withdrawal-Associated Memories in the Basolateral Amygdala and Dentate Gyrus.
Evidence of Tinnitus Development Due to Stress: An Experimental Study in Rats.
Synaptic protein interaction networks encode experience by assuming stimulus-specific and brain-region-specific states.
Effects of Mephedrone and Amphetamine Exposure during Adolescence on Spatial Memory in Adulthood: Behavioral and Neurochemical Analysis.
Cocaine Self-Administration and Abstinence Modulate NMDA Receptor Subunits and Active Zone Proteins in the Rat Nucleus Accumbens.
Amyloid-β Oligomers Regulate ADAM10 Synaptic Localization Through Aberrant Plasticity Phenomena.
PSD-95 deficiency disrupts PFC-associated function and behavior during neurodevelopment.
Glutamate Receptor Trafficking and Protein Synthesis Mediate the Facilitation of LTP by Secreted Amyloid Precursor Protein-Alpha.
The histone demethylase KDM6B in the medial prefrontal cortex epigenetically regulates cocaine reward memory.
Juvenile treatment with mGluR2/3 agonist prevents schizophrenia-like phenotypes in adult by acting through GSK3β.
Epigenetic mechanisms underlying NMDA receptor hypofunction in the prefrontal cortex of juvenile animals in the MAM model for schizophrenia.
Hierarchical organization and genetically separable subfamilies of PSD95 postsynaptic supercomplexes.
Juvenile treatment with a novel mGluR2 agonist/mGluR3 antagonist compound, LY395756, reverses learning deficits and cognitive flexibility impairments in adults in a neurodevelopmental model of schizophrenia.
NMDA receptors participate in the progression of diabetic kidney disease by decreasing Cdc42-GTP activation in podocytes.
Pentobarbital modifies the lipid raft-protein interaction: A first clue about the anesthesia mechanism on NMDA and GABA(A) receptors.
NMDA receptors are selectively partitioned into complexes and supercomplexes during synapse maturation.
LY395756, an mGluR2 agonist and mGluR3 antagonist, enhances NMDA receptor expression and function in the normal adult rat prefrontal cortex, but fails to improve working memory and reverse MK801-induced working memory impairment.
GluN2B-Containing NMDA Receptors Regulate AMPA Receptor Traffic through Anchoring of the Synaptic Proteasome.
Redistribution of ionotropic glutamate receptors detected by laser microdissection of the rat dentate gyrus 48 h following LTP induction in vivo.
Mutant disrupted-in-schizophrenia 1 in astrocytes: focus on glutamate metabolism.
Metabotropic glutamate receptor 5 mediates phosphorylation of vascular endothelial cadherin and nuclear localization of β-catenin in response to homocysteine.
Glutamate binding to the GluN2B subunit controls surface trafficking of N-methyl-D-aspartate (NMDA) receptors.
NMDA receptors mediate synaptic competition in culture.
Long-term consequences of a prolonged febrile seizure in a dual pathology model.
Stargazin and AMPA receptor membrane expression is increased in the somatosensory cortex of Genetic Absence Epilepsy Rats from Strasbourg.
In vivo composition of NMDA receptor signaling complexes differs between membrane subdomains and is modulated by PSD-95 and PSD-93.
The importance of N-methyl-D-aspartate (NMDA) receptors in subtraction of electrosensory reafference in the dorsal nucleus of skates.
N-methyl-D-aspartate, hyperpolarization-activated cation current (Ih) and gamma-aminobutyric acid conductances govern the risk of epileptogenesis following febrile seizures in rat hippocampus.
Increased expression, but not postsynaptic localisation, of ionotropic glutamate receptors during the late-phase of long-term potentiation in the dentate gyrus in vivo.
Differential trafficking of AMPA and NMDA receptors during long-term potentiation in awake adult animals.
Expression of AMPA and NMDA receptor subunits in the cervical spinal cord of wobbler mice.
Shank expression is sufficient to induce functional dendritic spine synapses in aspiny neurons.
Long-term regulation of N-methyl-D-aspartate receptor subunits and associated synaptic proteins following hippocampal synaptic plasticity.
SNAP-25 reduction in the hippocampus of patients with schizophrenia.
Light and electron microscopic distribution of the AMPA receptor subunit, GluR2, in the spinal cord of control and G86R mutant superoxide dismutase transgenic mice.
Distribution of glutamate receptor subunit proteins GluR2(4), GluR5/6/7, and NMDAR1 in the canine and primate cerebral cortex: a comparative immunohistochemical analysis.
Androgenic regulation of tubulin gene expression in axotomized hamster facial motoneurons.
Regional, cellular, and ultrastructural distribution of N-methyl-D-aspartate receptor subunit 1 in monkey hippocampus.
Franco-García A, Fernández-Gómez FJ, Gómez-Murcia V, Hidalgo JM, Milanés MV, Núñez C
Biomedicines 2022 Mar 2;10(3)
Biomedicines 2022 Mar 2;10(3)
Evidence of Tinnitus Development Due to Stress: An Experimental Study in Rats.
Kim MJ, Park SY, Park JM, Yu HJ, Park I, Park SN
The Laryngoscope 2021 Oct;131(10):2332-2340
The Laryngoscope 2021 Oct;131(10):2332-2340
Synaptic protein interaction networks encode experience by assuming stimulus-specific and brain-region-specific states.
Lautz JD, Tsegay KB, Zhu Z, Gniffke EP, Welsh JP, Smith SEP
Cell reports 2021 Nov 30;37(9):110076
Cell reports 2021 Nov 30;37(9):110076
Effects of Mephedrone and Amphetamine Exposure during Adolescence on Spatial Memory in Adulthood: Behavioral and Neurochemical Analysis.
Grochecki P, Smaga I, Lopatynska-Mazurek M, Gibula-Tarlowska E, Kedzierska E, Listos J, Talarek S, Marszalek-Grabska M, Hubalewska-Mazgaj M, Korga-Plewko A, Dudka J, Marzec Z, Filip M, Kotlinska JH
International journal of molecular sciences 2021 Jan 8;22(2)
International journal of molecular sciences 2021 Jan 8;22(2)
Cocaine Self-Administration and Abstinence Modulate NMDA Receptor Subunits and Active Zone Proteins in the Rat Nucleus Accumbens.
Smaga I, Wydra K, Frankowska M, Fumagalli F, Sanak M, Filip M
Molecules (Basel, Switzerland) 2020 Jul 31;25(15)
Molecules (Basel, Switzerland) 2020 Jul 31;25(15)
Amyloid-β Oligomers Regulate ADAM10 Synaptic Localization Through Aberrant Plasticity Phenomena.
Marcello E, Musardo S, Vandermeulen L, Pelucchi S, Gardoni F, Santo N, Antonucci F, Di Luca M
Molecular neurobiology 2019 Oct;56(10):7136-7143
Molecular neurobiology 2019 Oct;56(10):7136-7143
PSD-95 deficiency disrupts PFC-associated function and behavior during neurodevelopment.
Coley AA, Gao WJ
Scientific reports 2019 Jul 1;9(1):9486
Scientific reports 2019 Jul 1;9(1):9486
Glutamate Receptor Trafficking and Protein Synthesis Mediate the Facilitation of LTP by Secreted Amyloid Precursor Protein-Alpha.
Mockett BG, Guévremont D, Elder MK, Parfitt KD, Peppercorn K, Morrissey J, Singh A, Hintz TJ, Kochen L, Tom Dieck S, Schuman E, Tate WP, Williams JM, Abraham WC
The Journal of neuroscience : the official journal of the Society for Neuroscience 2019 Apr 24;39(17):3188-3203
The Journal of neuroscience : the official journal of the Society for Neuroscience 2019 Apr 24;39(17):3188-3203
The histone demethylase KDM6B in the medial prefrontal cortex epigenetically regulates cocaine reward memory.
Zhang YX, Akumuo RC, España RA, Yan CX, Gao WJ, Li YC
Neuropharmacology 2018 Oct;141:113-125
Neuropharmacology 2018 Oct;141:113-125
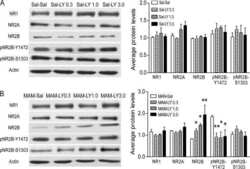
Juvenile treatment with mGluR2/3 agonist prevents schizophrenia-like phenotypes in adult by acting through GSK3β.
Xing B, Han G, Wang MJ, Snyder MA, Gao WJ
Neuropharmacology 2018 Jul 15;137:359-371
Neuropharmacology 2018 Jul 15;137:359-371
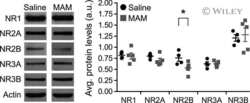
Epigenetic mechanisms underlying NMDA receptor hypofunction in the prefrontal cortex of juvenile animals in the MAM model for schizophrenia.
Gulchina Y, Xu SJ, Snyder MA, Elefant F, Gao WJ
Journal of neurochemistry 2017 Nov;143(3):320-333
Journal of neurochemistry 2017 Nov;143(3):320-333
Hierarchical organization and genetically separable subfamilies of PSD95 postsynaptic supercomplexes.
Frank RAW, Zhu F, Komiyama NH, Grant SGN
Journal of neurochemistry 2017 Aug;142(4):504-511
Journal of neurochemistry 2017 Aug;142(4):504-511
Juvenile treatment with a novel mGluR2 agonist/mGluR3 antagonist compound, LY395756, reverses learning deficits and cognitive flexibility impairments in adults in a neurodevelopmental model of schizophrenia.
Li ML, Gulchina Y, Monaco SA, Xing B, Ferguson BR, Li YC, Li F, Hu XQ, Gao WJ
Neurobiology of learning and memory 2017 Apr;140:52-61
Neurobiology of learning and memory 2017 Apr;140:52-61
NMDA receptors participate in the progression of diabetic kidney disease by decreasing Cdc42-GTP activation in podocytes.
Shen J, Wang R, He Z, Huang H, He X, Zhou J, Yan Y, Shen S, Shao X, Shen X, Weng C, Lin W, Chen J
The Journal of pathology 2016 Oct;240(2):149-60
The Journal of pathology 2016 Oct;240(2):149-60
Pentobarbital modifies the lipid raft-protein interaction: A first clue about the anesthesia mechanism on NMDA and GABA(A) receptors.
Sierra-Valdez FJ, Ruiz-Suárez JC, Delint-Ramirez I
Biochimica et biophysica acta 2016 Nov;1858(11):2603-2610
Biochimica et biophysica acta 2016 Nov;1858(11):2603-2610
NMDA receptors are selectively partitioned into complexes and supercomplexes during synapse maturation.
Frank RA, Komiyama NH, Ryan TJ, Zhu F, O'Dell TJ, Grant SG
Nature communications 2016 Apr 27;7:11264
Nature communications 2016 Apr 27;7:11264
LY395756, an mGluR2 agonist and mGluR3 antagonist, enhances NMDA receptor expression and function in the normal adult rat prefrontal cortex, but fails to improve working memory and reverse MK801-induced working memory impairment.
Li ML, Yang SS, Xing B, Ferguson BR, Gulchina Y, Li YC, Li F, Hu XQ, Gao WJ
Experimental neurology 2015 Nov;273:190-201
Experimental neurology 2015 Nov;273:190-201
GluN2B-Containing NMDA Receptors Regulate AMPA Receptor Traffic through Anchoring of the Synaptic Proteasome.
Ferreira JS, Schmidt J, Rio P, Águas R, Rooyakkers A, Li KW, Smit AB, Craig AM, Carvalho AL
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Jun 3;35(22):8462-79
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Jun 3;35(22):8462-79
Redistribution of ionotropic glutamate receptors detected by laser microdissection of the rat dentate gyrus 48 h following LTP induction in vivo.
Kennard JT, Guévremont D, Mason-Parker SE, Abraham WC, Williams JM
PloS one 2014;9(3):e92972
PloS one 2014;9(3):e92972
Mutant disrupted-in-schizophrenia 1 in astrocytes: focus on glutamate metabolism.
Abazyan S, Yang EJ, Abazyan B, Xia M, Yang C, Rojas C, Slusher B, Sattler R, Pletnikov M
Journal of neuroscience research 2014 Dec;92(12):1659-68
Journal of neuroscience research 2014 Dec;92(12):1659-68
Metabotropic glutamate receptor 5 mediates phosphorylation of vascular endothelial cadherin and nuclear localization of β-catenin in response to homocysteine.
Beard RS Jr, Reynolds JJ, Bearden SE
Vascular pharmacology 2012 Mar-Apr;56(3-4):159-67
Vascular pharmacology 2012 Mar-Apr;56(3-4):159-67
Glutamate binding to the GluN2B subunit controls surface trafficking of N-methyl-D-aspartate (NMDA) receptors.
She K, Ferreira JS, Carvalho AL, Craig AM
The Journal of biological chemistry 2012 Aug 10;287(33):27432-45
The Journal of biological chemistry 2012 Aug 10;287(33):27432-45
NMDA receptors mediate synaptic competition in culture.
She K, Craig AM
PloS one 2011;6(9):e24423
PloS one 2011;6(9):e24423
Long-term consequences of a prolonged febrile seizure in a dual pathology model.
Gibbs S, Chattopadhyaya B, Desgent S, Awad PN, Clerk-Lamalice O, Levesque M, Vianna RM, Rébillard RM, Delsemme AA, Hébert D, Tremblay L, Lepage M, Descarries L, Di Cristo G, Carmant L
Neurobiology of disease 2011 Aug;43(2):312-21
Neurobiology of disease 2011 Aug;43(2):312-21
Stargazin and AMPA receptor membrane expression is increased in the somatosensory cortex of Genetic Absence Epilepsy Rats from Strasbourg.
Kennard JT, Barmanray R, Sampurno S, Ozturk E, Reid CA, Paradiso L, D'Abaco GM, Kaye AH, Foote SJ, O'Brien TJ, Powell KL
Neurobiology of disease 2011 Apr;42(1):48-54
Neurobiology of disease 2011 Apr;42(1):48-54
In vivo composition of NMDA receptor signaling complexes differs between membrane subdomains and is modulated by PSD-95 and PSD-93.
Delint-Ramirez I, Fernández E, Bayés A, Kicsi E, Komiyama NH, Grant SG
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Jun 16;30(24):8162-70
The Journal of neuroscience : the official journal of the Society for Neuroscience 2010 Jun 16;30(24):8162-70
The importance of N-methyl-D-aspartate (NMDA) receptors in subtraction of electrosensory reafference in the dorsal nucleus of skates.
Zhang Z, Bodznick D
The Journal of experimental biology 2010 Aug 1;213(Pt 15):2700-9
The Journal of experimental biology 2010 Aug 1;213(Pt 15):2700-9
N-methyl-D-aspartate, hyperpolarization-activated cation current (Ih) and gamma-aminobutyric acid conductances govern the risk of epileptogenesis following febrile seizures in rat hippocampus.
Ouardouz M, Lema P, Awad PN, Di Cristo G, Carmant L
The European journal of neuroscience 2010 Apr;31(7):1252-60
The European journal of neuroscience 2010 Apr;31(7):1252-60
Increased expression, but not postsynaptic localisation, of ionotropic glutamate receptors during the late-phase of long-term potentiation in the dentate gyrus in vivo.
Kennard JT, Guévremont D, Mason-Parker SE, Abraham WC, Williams JM
Neuropharmacology 2009 Jan;56(1):66-72
Neuropharmacology 2009 Jan;56(1):66-72
Differential trafficking of AMPA and NMDA receptors during long-term potentiation in awake adult animals.
Williams JM, Guévremont D, Mason-Parker SE, Luxmanan C, Tate WP, Abraham WC
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 Dec 19;27(51):14171-8
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 Dec 19;27(51):14171-8
Expression of AMPA and NMDA receptor subunits in the cervical spinal cord of wobbler mice.
Bigini P, Gardoni F, Barbera S, Cagnotto A, Fumagalli E, Longhi A, Corsi MM, Di Luca M, Mennini T
BMC neuroscience 2006 Oct 26;7:71
BMC neuroscience 2006 Oct 26;7:71
Shank expression is sufficient to induce functional dendritic spine synapses in aspiny neurons.
Roussignol G, Ango F, Romorini S, Tu JC, Sala C, Worley PF, Bockaert J, Fagni L
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Apr 6;25(14):3560-70
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Apr 6;25(14):3560-70
Long-term regulation of N-methyl-D-aspartate receptor subunits and associated synaptic proteins following hippocampal synaptic plasticity.
Williams JM, Guévremont D, Kennard JT, Mason-Parker SE, Tate WP, Abraham WC
Neuroscience 2003;118(4):1003-13
Neuroscience 2003;118(4):1003-13
SNAP-25 reduction in the hippocampus of patients with schizophrenia.
Thompson PM, Egbufoama S, Vawter MP
Progress in neuro-psychopharmacology & biological psychiatry 2003 May;27(3):411-7
Progress in neuro-psychopharmacology & biological psychiatry 2003 May;27(3):411-7
Light and electron microscopic distribution of the AMPA receptor subunit, GluR2, in the spinal cord of control and G86R mutant superoxide dismutase transgenic mice.
Morrison BM, Janssen WG, Gordon JW, Morrison JH
The Journal of comparative neurology 1998 Jun 15;395(4):523-34
The Journal of comparative neurology 1998 Jun 15;395(4):523-34
Distribution of glutamate receptor subunit proteins GluR2(4), GluR5/6/7, and NMDAR1 in the canine and primate cerebral cortex: a comparative immunohistochemical analysis.
Hof PR, Vissavajjhala P, Rosenthal RE, Fiskum G, Morrison JH
Brain research 1996 Jun 3;723(1-2):77-89
Brain research 1996 Jun 3;723(1-2):77-89
Androgenic regulation of tubulin gene expression in axotomized hamster facial motoneurons.
Jones KJ, Oblinger MM
The Journal of neuroscience : the official journal of the Society for Neuroscience 1994 Jun;14(6):3620-7
The Journal of neuroscience : the official journal of the Society for Neuroscience 1994 Jun;14(6):3620-7
Regional, cellular, and ultrastructural distribution of N-methyl-D-aspartate receptor subunit 1 in monkey hippocampus.
Siegel SJ, Brose N, Janssen WG, Gasic GP, Jahn R, Heinemann SF, Morrison JH
Proceedings of the National Academy of Sciences of the United States of America 1994 Jan 18;91(2):564-8
Proceedings of the National Academy of Sciences of the United States of America 1994 Jan 18;91(2):564-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of NMDA 1 Receptor was performed by loading 30 µg of Mouse Brain (lane1) and Rat Brain (lane2) tissue lysate using Novex® NuPAGE® 12 % Bis-Tris gel (Product # NP0342BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and Pierce™ Power Blotter System (22834). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk at 4ºC overnight. NMDA 1 Receptor was detected at ~103 kDa using NMDA 1 Receptor Mouse Monoclonal Antibody (Product # 32-0500) at 1-3 µg/mL in 5 % skim milk for 3 hours at room temperature on a rocking platform. Goat Anti-Mouse - HRP Secondary Antibody (Product # 62-6520) at 1:4000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-NMDAR1 Monoclonal Antibody (54.1) (Product # 32-0500) and a 110k Da band corresponding to NMDAR1 was observed in Mouse Brain but not Mouse Liver. Tissue extracts (30 µg lysate) of Mouse Brain (Lane 1) or Mouse Liver (Lane 2) were electrophoresed using NuPAGE™ 10% Bis-Tris Protein Gel (Product # NP0301BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:4000) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of MAP2 (green) and NMDAR1 (red) on rat primary Hippocampal neurons (E18) (Product # A15587) cultured for 28 days in the B-27 Plus Neuronal Culture System (Product # A3653401). At day 28 the cells were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.1% triton x-100 for 30min, and blocked with 1% BSA for 30 min at room temperature. Cells were stained with anti-NMDAR1 antibody (Product # 32-0500) at a dilution of 1:100, and anti-MAP2 (Product # PA5-17646) at a dilution of 1:250, in 1% BSA staining buffer, overnight at 4C, and then incubated with Alexa Fluor 488 conjugated donkey anti-rabbit (Product # A-21206) and Alexa Fluor 594 donkey anti-mouse (Product # A-21203) antibodies at a dilution of 1:1000 for 30 min. at room temp. Wash 3 times with DPBS. Stain with DAPI for nucleus.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of NMDA1 receptor showing staining in the cytoplasm and membrane of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10 mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a NMDA1 receptor Mouse monoclonal antibody (Product # 32-0500) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
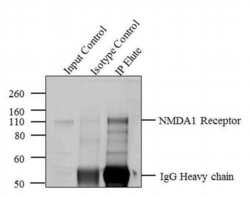
- Experimental details
- Immunoprecipitation of NMDA 1 Receptor was performed with 5 æg of the NMDA 1 Receptor Mouse Monoclonal Antibody (Product # 32-0500) on tissue extract from Rat Brain using the Dynabeads® Protein A Immunoprecipitation Kit (10006D). Normal Mouse IgG was used as a negative IP control. Subsequently, western blot analysis was performed using Novex® NuPAGE® 10 % Bis-Tris gel (Product # NP0301BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800). Proteins were transferred using iBlot® 2 Dry Blotting System (IB21001) to a nitrocellulose membrane and blocked with 5% skim milk for 1 hour at room temperature on a rocking platform. NMDA 1 Receptor was detected at ~ 103 kDa using NMDA 1 Receptor Mouse Monoclonal Antibody (Product # 32-0500) at 1-3 æg/mL in 5 % skim milk at 4ºC overnight on a rocking platform. Goat Anti-Mouse - HRP Secondary Antibody (Product # 62-6520) at 1:4000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106). Lane 1 represents 10 % of the total tissue extract (input), Lane 2 is the IP performed with Mouse IgG and Lane 3 represents IP performed with NMDA 1 Receptor Mouse Monoclonal Antibody (Product # 32-0500).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
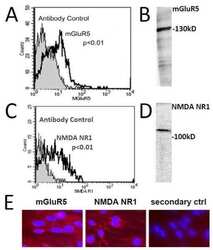
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
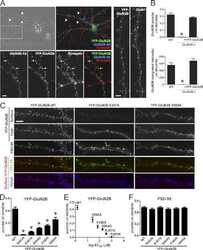
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 NMDA receptor subunits in spinal cord of wobbler mice . Representative photomicrographs showing NR1 (A,C) and NR2A (B,D) immunostaining in the cervical spinal cord of four-week-old wobbler mice (B). Scale bar, A, B 100 mum. C, D 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 western blotting analysis and quantification of the percentage of AMPA and NMDA receptor subunits expression in spinal cord of wobbler mice . Representative immunoblot from cervical spinal cord samples from 12-week-old healthy mice and age-matched wobbler mice. Left column: whole spinal cord homogenates. Right column: spinal cord TIF. Quantification of the mean values of immunodensity for CaMK, AMPA and NMDA receptor subunits, obtained from the cervical spinal wobbler mice in whole spinal cord homogenates (Homo) and in TIF. Data are representative of two independent experiments on two different homogenate preparations of four pooled animals for each group and replicated two times in each homogenate preparation. Values reported represent the percentage of the mean values of immunodensity obtained in 12-week-old wobbler mice compared to the levels measured in healthy littermates and normalized to 100. Both preparations samples were analyzed by Western blot analysis with CaMKII, GluR1, GluR2, GluR3, GluR4, NR1, and NR2A antibodies. The same amount of protein was loaded per lane.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
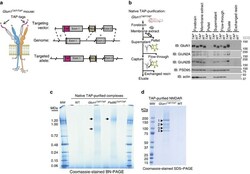
- Figure 1 Native supermolecular assembly of NMDAR subunits in the mammalian brain. ( a ) Schematic showing the higher-order assembly from individual proteins to complexes and supercomplexes (complexes of complexes). ( b ) Native protein complexes of GluN1, GluN2A and GluN2B detected in BNP immunoblot screen of mouse forebrain extracts. Approximately 0.8 and ~1.5 MDa complexes indicated by filled and open arrowheads, respectively (hereafter used to label all figures). The expected size of each protein in monomeric form indicated with pink rectangle. On left side, non-denaturing molecular mass indicated in mega-Daltons (MDa). ( c ) BNP GluN1 immunoblot of fractions from glycerol gradient (10-30%) ultracentrifugation. 'IN', forebrain extract supernatant. On right side, non-denaturing molecular mass indicated in MDa. ( d ) BNP GluN1 immunoblot of fresh human cortical biopsy samples from the inferior frontal (inf. front.), inferior temporal (inf. temp.) and superior frontal (sup. front.) lobes. Mouse forebrain extract supernatant shown for comparison. These data show the ~0.8 and ~1.5 MDa NMDA receptor complexes (1.5-NR and 0.8-NR) were conserved between mouse and humans.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2 Generation of TAP-tagged GluN1 ( Glun1 TAP/TAP ) knock-in mice for purification of native NMDARs. ( a ) Genetic engineering of TAP-tags into GluN1. Left, schematic shows tetrameric NMDAR with two GluN1 subunits (grey) engineered with tandem affinity peptide (TAP-tags, magenta) on their extracellular N-termini. GluN2 subunits (GluN2A, cyan; GluN2B, orange) shown with C-terminal cytoplasmic ESDV motifs/PDZ-ligands. TAP-tag encodes 3xFlag and His x6 . Right, schematic shows gene-targeting vector carrying TAP-tag sequence in Glun1 exon1, 5' and 3' regions of homology with genome and resultant targeted allele below. The neomycin selection cassette was subsequently deleted using Cre/loxP. Grey-filled boxes, exons; magenta, TAP cassette; brown box, neo neomycin resistance cassette; brown triangle, loxP site; dotted lines, homology arms. ( b ) Purification of native NMDARs from Glun1 TAP/TAP mouse forebrains. Left, schematic of purification steps and samples corresponding to right, immunoblots of NMDAR subunits, PSD95, actin. Dissected mouse forebrains were homogenized and fractionated. Crude membrane fraction was solubilized (membrane extract) and separated by centrifugation (supernatant and pellet). Supernatant was incubated with Flag-affinity resin capturing TAP-tagged receptors with some residual Flow-through. High yields of native receptor were released (eluate) by peptide-antigen exchange. Following elution no material remained (exchanged resin). Right, purif
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
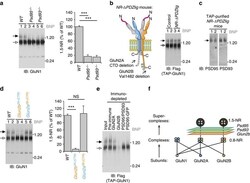
- Figure 3 Mutant mouse screen of NMDAR supermolecular assembly. ( a ) Left, BNP GluN1 immunoblot of forebrain extracts WT (lanes 1 and 2, duplicates), Psd95 -/- (lanes 3 and 4), Psd93 -/- (lanes 5 and 6). Right panel, quantification shows relative to WT ( n =6) 1.5-NR decreased to 17% ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 The GluN2A/GluN2B relative molar abundance using mouse genetics and quantitative tagging. ( a ) SDS-PAGE immunoblot of WT, Glun2b 2A(CTR)/2A(CTR) and Glun2a 2B(CTR)/2B(CTR) forebrain extract. Top panel, detected the GluN2A C-terminal domain (CTD). Second panel down, GluN2B CTD. Glun2a 2B(CTR)/2B(CTR) and Glun2b 2A(CTR)/2A(CTR) provided negative controls for the specificities of the GluN2A CTD and GluN2B CTD antibodies, respectively. Third, fourth and fifth panels down, detected GluN2A NTD, GluN2B NTD subunits and GluN1, respectively. Blue and orange labels indicate chimeric Glun2a and Glun2b knock-in mutations, respectively 20 . ( b ) Dilution series of Glun2a 2B(CTR)/2B(CTR) forebrain extract into that of Glun2b 2A(CTR)/2A(CTR) indicated sensitivity of quantification. Immunoblots detected GluN2A CTD (upper panel) and GluN2B CTD (lower panel). ( c ) Quantification of GluN2A CTD and GluN2B CTD immunoblots. Measurements from Glun2b 2A(CTR)/2A(CTR) ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 1.5-NR and 1.5-PSD95 assemble late in postnatal development. ( a ) BNP immunoblots show WT mouse forebrain extracts at developmental time points probed with antibodies against GluN1, GluN2A, GluN2B, PSD95 and PSD93. Filled arrow, ~1.5 MDa complexes that only assembly from ~P16 onwards. Sample load was normalized by forebrain mass. Molecular weight in MDa shown on right. ( b ) Quantification of changing abundance of 0.8-NR and 1.5-NR from P12 ( n =3) to P16 ( n =3) using BNP immunoblots of GluN1. 0.8-NR increased by 25%, whereas 1.5-NR increased by 445%; t -test, P >0.01; Error bars indicate s.e.m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
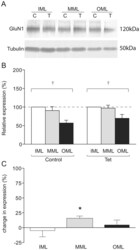
- Experimental details
- Figure 3 GluN1 expression in LTP-stimulated rats. (a) Representative Western blot showing GluN1 (upper) and tubulin (lower) expression in laser microdissected tissue from one LTP-stimulated animal. (b) Mean relative expression of GluN1 in the molecular layer zones from LTP-stimulated and naive hemispheres, determined as a proportion of the ipsilateral inner molecular layer. (c) Mean percentage difference in tubulin-normalised expression of GluN1 in molecular layer zones of the LTP-stimulated hemisphere compared to the naive hemisphere; * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Effect of repeated mephedrone or amphetamine administration during adolescence on MMP-9 ( A ) and NMDA subunits (GluN1, GluN2A, GluN2B) and PSD-95 ( B ) expression in the prefrontal cortex (PC) and hippocampus (HC) of rats (PND 87) that underwent the Barnes maze task. Representative blots for significant changes are presented, as well as full membranes are presented in the supplementary material (Figure S1) . Data are expressed as mean +- SEM. n = 8 rats/group. * p < 0.05 vs vehicle (0.9% NaCl). V--vehicle, M--mephedrone, A--amphetamine.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Changes in the expression of NMDA receptor subunits (GluN1, GluN2A and GluN2B) in the nucleus accumbens in rats following cocaine self-administration. All data are expressed as % of control (mean +- SEM). N = 6 rats/group. * p < 0.05 vs. yoked saline. ( a ), GluN1 protein levels [% of control]; ( b ), GluN2A protein levels [% of control]. ; ( c ), GluN2B protein levels [% of control].
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
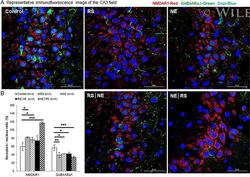
- Experimental details
- 7 Fig GABAARalpha1 and NMADR1 expression by immunofluorescence and DAPI counterstaining in the CA3 field of the hippocampus. (A) Representative immunofluorescence image of the CA3 field. (B) NE|RS group showed significantly increased expression of NMADR1 (red), whereas expression of GABAARalpha1 (green) was significantly decreased in the RS and NE|RS groups. Error bars indicate SEM. * P < .05, ** P < .005, *** P < .001. An analysis of variance with least significant difference post hoc tests for multiple comparisons was performed to examine immunofluorescence in the CA3 field of the hippocampus. GABAARalpha1 = gamma-aminobutyric acid A receptor alpha 1; NE = noise exposure; NMDAR1 = N -methyl- d -aspartate receptor 1; RS = restraint stress; SEM = standard error of mean
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Abeta oligomers characterization and effects on the synapses. a A representative Coomassie-stained polyacrylamide gel and Western blot analysis of Abeta 42-1 and oAbeta 1-42 . The image shows the presence of oligomers formation for Abeta 1-42 and monomer and aggregated forms for Abeta 42-1 . b TEM revealed globular but not fibrillar structures for oAbeta 1-42 preparation, scale bar 500 nm. c Representative confocal images of GFP-transfected primary hippocampal neurons. The analysis shows that oAbeta 1-42 (500 nM, 24 h) reduces spine density (CTRL 4.58 +- 0.18; Abeta 42-1 4.15 +- 0.12; oAbeta 1-42 3.36 +- 0.18; ** p < 0.01, *** p < 0.001; one-way ANOVA, n = 34-45). Scale bar 5 mum. d MTT test shows that exposure to oAbeta 1-42 for 24 h induces cell death starting from a concentration of 1 muM (** p < 0.01, *** p < 0.001 one-way ANOVA, oAbeta 1-42 vs CTRL, n = 4). e Representative traces of mEPSCs collected in hippocampal neurons before and after oAbeta 1-42 exposure (upper panel) and before and after chemical LTD (cLTD, lower panel). At least 21 neurons before and after each treatment (oAbeta 1-42 or cLTD) have been analyzed and the related analysis of mEPSC amplitudes, here shown as cumulative probability, includes these n of excitatory events: 1550 (before oAbeta 1-42 , black line) vs 970 (after oAbeta 1-42 , red line), CTRL vs oAbeta 1-42 p = 0.0075; 2100 (before cLTD, black line) vs 1970 (after cLTD, green line), CTRL vs cLTD p < 0.0001. f Western blot analysis of s
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunoprecipitation
Immunoprecipitation