Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Flow cytometry [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- DM1204 - Provider product page

- Provider
- Acris Antibodies GmbH
- Proper citation
- Acris Antibodies GmbH Cat#DM1204, RRID:AB_977064
- Product name
- anti CD66e / CEACAM5
- Antibody type
- Monoclonal
- Antigen
- Immunisation with extracted protein of CEACAM5
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- D14HD11
- Vial size
- 0.1 mg
- Concentration
- 2.0 mg/ml
Submitted references Extracellular IgC2 constant domains of CEACAMs mediate PI3K sensitivity during uptake of pathogens.
Neisseria meningitidis has two independent modes of recognizing its human receptor CEACAM1.
The prevalence and nature of glycan alterations on specific proteins in pancreatic cancer patients revealed using antibody-lectin sandwich arrays.
Human decay-accelerating factor and CEACAM receptor-mediated internalization and intracellular lifestyle of Afa/Dr diffusely adhering Escherichia coli in epithelial cells.
CEACAM1 dynamics during neisseria gonorrhoeae suppression of CD4+ T lymphocyte activation.
Opa proteins of pathogenic neisseriae initiate Src kinase-dependent or lipid raft-mediated uptake via distinct human carcinoembryonic antigen-related cell adhesion molecule isoforms.
CEACAM engagement by human pathogens enhances cell adhesion and counteracts bacteria-induced detachment of epithelial cells.
Engulfment of Neisseria gonorrhoeae: revealing distinct processes of bacterial entry by individual carcinoembryonic antigen-related cellular adhesion molecule family receptors.
Granulocyte CEACAM3 is a phagocytic receptor of the innate immune system that mediates recognition and elimination of human-specific pathogens.
Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes.
Distinct mechanisms of internalization of Neisseria gonorrhoeae by members of the CEACAM receptor family involving Rac1- and Cdc42-dependent and -independent pathways.
Homophilic adhesion of human CEACAM1 involves N-terminal domain interactions: structural analysis of the binding site.
Carcinoembryonic antigen family members CEACAM6 and CEACAM7 are differentially expressed in normal tissues and oppositely deregulated in hyperplastic colorectal polyps and early adenomas.
Voges M, Bachmann V, Naujoks J, Kopp K, Hauck CR
PloS one 2012;7(6):e39908
PloS one 2012;7(6):e39908
Neisseria meningitidis has two independent modes of recognizing its human receptor CEACAM1.
Kuespert K, Roth A, Hauck CR
PloS one 2011 Jan 27;6(1):e14609
PloS one 2011 Jan 27;6(1):e14609
The prevalence and nature of glycan alterations on specific proteins in pancreatic cancer patients revealed using antibody-lectin sandwich arrays.
Yue T, Goldstein IJ, Hollingsworth MA, Kaul K, Brand RE, Haab BB
Molecular & cellular proteomics : MCP 2009 Jul;8(7):1697-707
Molecular & cellular proteomics : MCP 2009 Jul;8(7):1697-707
Human decay-accelerating factor and CEACAM receptor-mediated internalization and intracellular lifestyle of Afa/Dr diffusely adhering Escherichia coli in epithelial cells.
Guignot J, Hudault S, Kansau I, Chau I, Servin AL
Infection and immunity 2009 Jan;77(1):517-31
Infection and immunity 2009 Jan;77(1):517-31
CEACAM1 dynamics during neisseria gonorrhoeae suppression of CD4+ T lymphocyte activation.
Lee HS, Ostrowski MA, Gray-Owen SD
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6827-35
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6827-35
Opa proteins of pathogenic neisseriae initiate Src kinase-dependent or lipid raft-mediated uptake via distinct human carcinoembryonic antigen-related cell adhesion molecule isoforms.
Schmitter T, Pils S, Weibel S, Agerer F, Peterson L, Buntru A, Kopp K, Hauck CR
Infection and immunity 2007 Aug;75(8):4116-26
Infection and immunity 2007 Aug;75(8):4116-26
CEACAM engagement by human pathogens enhances cell adhesion and counteracts bacteria-induced detachment of epithelial cells.
Muenzner P, Rohde M, Kneitz S, Hauck CR
The Journal of cell biology 2005 Aug 29;170(5):825-36
The Journal of cell biology 2005 Aug 29;170(5):825-36
Engulfment of Neisseria gonorrhoeae: revealing distinct processes of bacterial entry by individual carcinoembryonic antigen-related cellular adhesion molecule family receptors.
McCaw SE, Liao EH, Gray-Owen SD
Infection and immunity 2004 May;72(5):2742-52
Infection and immunity 2004 May;72(5):2742-52
Granulocyte CEACAM3 is a phagocytic receptor of the innate immune system that mediates recognition and elimination of human-specific pathogens.
Schmitter T, Agerer F, Peterson L, Munzner P, Hauck CR
The Journal of experimental medicine 2004 Jan 5;199(1):35-46
The Journal of experimental medicine 2004 Jan 5;199(1):35-46
Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes.
Boulton IC, Gray-Owen SD
Nature immunology 2002 Mar;3(3):229-36
Nature immunology 2002 Mar;3(3):229-36
Distinct mechanisms of internalization of Neisseria gonorrhoeae by members of the CEACAM receptor family involving Rac1- and Cdc42-dependent and -independent pathways.
Billker O, Popp A, Brinkmann V, Wenig G, Schneider J, Caron E, Meyer TF
The EMBO journal 2002 Feb 15;21(4):560-71
The EMBO journal 2002 Feb 15;21(4):560-71
Homophilic adhesion of human CEACAM1 involves N-terminal domain interactions: structural analysis of the binding site.
Watt SM, Teixeira AM, Zhou GQ, Doyonnas R, Zhang Y, Grunert F, Blumberg RS, Kuroki M, Skubitz KM, Bates PA
Blood 2001 Sep 1;98(5):1469-79
Blood 2001 Sep 1;98(5):1469-79
Carcinoembryonic antigen family members CEACAM6 and CEACAM7 are differentially expressed in normal tissues and oppositely deregulated in hyperplastic colorectal polyps and early adenomas.
Schölzel S, Zimmermann W, Schwarzkopf G, Grunert F, Rogaczewski B, Thompson J
The American journal of pathology 2000 Feb;156(2):595-605
The American journal of pathology 2000 Feb;156(2):595-605
No comments: Submit comment
Supportive validation
- Submitted by
- Acris Antibodies GmbH (provider)
- Main image
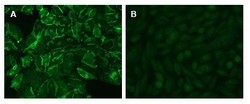
- Experimental details
- Figure 2: Immunofluorescence Microscopy of CHO cells using D14HD11 Cat#DM1204. CHO cells were transfected with an expression vector encoding CEACAM1 (A). Untransfected CHO parental cells served as Negative Control (B). Binding of D14HD11 was visualized with a FITC-conjugated secondary antibody.
Supportive validation
- Submitted by
- Acris Antibodies GmbH (provider)
- Main image

- Experimental details
- Figure 3: BOSC cells were transiently transfected with expression vectors containing either the cDNA of CEACAM1, CEACAM3-CEACAM8 or CEACAM19-21. Recognition of CEACAM4 was tested on CHO cells stably transfected with a CEACAM4 expressionvector. Expression of the constructs was confirmed with monoclonal antibodies known to recognise the corresponding proteins(CEACAM1: 4/3/17, CEACAM3,4: D14HD11, CEACAM5: 26/3/13, CEACAM6: 9A6, CEACAM7: BAC2, CEACAM8: GM-2H6,CEACAM19-21: anti-myc, green curves). An irrelevant monoclonal antibody served as a negative control (black curves). Forspecificity testing, protein G purified D14HD11 was tested on all CEACAM transfectants. A positive signal was obtained with CEA-CAM1, CEACAM3, CEACAM4, CEACAM5 and CEACAM6 expressing cells (red curves).
- Submitted by
- Acris Antibodies GmbH (provider)
- Main image
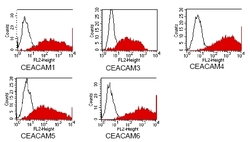
- Experimental details
- Figure 4: FACS analysis of BOSC23 cells using D14HD11 Cat#DM1204. BOSC23 cells were transiently transfected with anexpression vector encoding either CEACAM1,3,4,5,6 (red curves) or an irrelevant protein (control transfectant: blackcurves). Binding of D14HD11 was detected with a PE-conjugated secondary antibody. A positive signal wasobtained only with CEACAM1, CEACAM3, CEACAM4,CEACAM5 and CEACAM6 expressing cells.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry