Antibody data
- Antibody Data
- Antigen structure
- References [34]
- Comments [0]
- Validations
- Western blot [3]
- Immunohistochemistry [2]
- Other assay [17]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-11981 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Fibronectin Monoclonal Antibody (FBN11)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- MA5-11981 targets Fibronectin in IF, WB and IHC (P) applications and shows reactivity with Human, mouse, and Rat samples. The MA5-11981 immunogen is recombinant fibronectin protein.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- FBN11
- Vial size
- 500 µL
- Concentration
- 0.2 mg/mL
- Storage
- 4° C
Submitted references Hdac8 Inhibitor Alleviates Transverse Aortic Constriction-Induced Heart Failure in Mice by Downregulating Ace1.
More Is Not Always Better-the Double-Headed Role of Fibronectin in Staphylococcus aureus Host Cell Invasion.
Human placental villous stromal extracellular matrix regulates fetoplacental angiogenesis in severe fetal growth restriction.
Transthyretin amyloid fibrils alter primary fibroblast structure, function, and inflammatory gene expression.
TAGLN and High-mobility Group AT-Hook 2 (HMGA2) Complex Regulates TGF-β-induced Colorectal Cancer Metastasis.
Development of prediction models for lymph node metastasis in endometrioid endometrial carcinoma.
Lysophosphatidic acid promotes thrombus stability by inducing rapid formation of neutrophil extracellular traps: A new mechanism of thrombosis.
Macrophage-associated wound healing contributes to African green monkey SIV pathogenesis control.
IL-17 metabolically reprograms activated fibroblastic reticular cells for proliferation and survival.
Rhodoptilometrin, a Crinoid-Derived Anthraquinone, Induces Cell Regeneration by Promoting Wound Healing and Oxidative Phosphorylation in Human Gingival Fibroblast Cells.
Degenerate intervertebral disc-like pH induces a catabolic mechanoresponse in human nucleus pulposus cells.
Activation of hepatocyte growth factor/MET signaling initiates oncogenic transformation and enhances tumor aggressiveness in the murine prostate.
TGF-β1-induced differentiation of SHED into functional smooth muscle cells.
Decellularized extracellular matrix of human umbilical vein endothelial cells promotes endothelial differentiation of stem cells from exfoliated deciduous teeth.
Piceatannol Attenuates Renal Fibrosis Induced by Unilateral Ureteral Obstruction via Downregulation of Histone Deacetylase 4/5 or p38-MAPK Signaling.
Gallic acid prevents isoproterenol-induced cardiac hypertrophy and fibrosis through regulation of JNK2 signaling and Smad3 binding activity.
Characterization of chondrocyte sheets prepared using a co-culture method with temperature-responsive culture inserts.
Class I HDACs specifically regulate E-cadherin expression in human renal epithelial cells.
Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro.
Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in SIV-infected macaques.
RhoB deficiency in thymic medullary epithelium leads to early thymic atrophy.
The immunohistochemical characterization of human fetal olfactory bulb and olfactory ensheathing cells in culture as a source for clinical CNS restoration.
Primary human dermal fibroblast interactions with open weave three-dimensional scaffolds prepared from synthetic human elastin.
The properties of bioengineered chondrocyte sheets for cartilage regeneration.
A physical mechanism for coupling bone resorption and formation in adult human bone.
Molecular alteration of Ca(v)1.2 calcium channel in chronic myocardial infarction.
Flow shear stress affects macromolecular accumulation through modulation of internal elastic lamina fenestrae.
Melatonin protects against epirubicin-induced cardiotoxicity.
Uterine stretch regulates temporal and spatial expression of fibronectin protein and its alpha 5 integrin receptor in myometrium of unilaterally pregnant rats.
Progesterone withdrawal up-regulates fibronectin and integrins during menstruation and repair in the rhesus macaque endometrium.
Inhibition of NAD(P)H oxidase reduces fibronectin expression in stroke-prone renovascular hypertensive rat brain.
Expression of laminin and fibronectin in renal dysplasia.
Angiotensin-converting enzyme inhibitor captopril prevents volume overload cardiomyopathy in experimental chronic aortic valve regurgitation.
Mammary localization and abundance of laminin, fibronectin, and collagen IV proteins in prepubertal heifers.
Zhao T, Kee HJ, Kee SJ, Jeong MH
Oxidative medicine and cellular longevity 2022;2022:6227330
Oxidative medicine and cellular longevity 2022;2022:6227330
More Is Not Always Better-the Double-Headed Role of Fibronectin in Staphylococcus aureus Host Cell Invasion.
Niemann S, Nguyen MT, Eble JA, Chasan AI, Mrakovcic M, Böttcher RT, Preissner KT, Roßlenbroich S, Peters G, Herrmann M
mBio 2021 Oct 26;12(5):e0106221
mBio 2021 Oct 26;12(5):e0106221
Human placental villous stromal extracellular matrix regulates fetoplacental angiogenesis in severe fetal growth restriction.
Ji S, Gumina D, McPeak K, Moldovan R, Post MD, Su EJ
Clinical science (London, England : 1979) 2021 May 14;135(9):1127-1143
Clinical science (London, England : 1979) 2021 May 14;135(9):1127-1143
Transthyretin amyloid fibrils alter primary fibroblast structure, function, and inflammatory gene expression.
Dittloff KT, Iezzi A, Zhong JX, Mohindra P, Desai TA, Russell B
American journal of physiology. Heart and circulatory physiology 2021 Jul 1;321(1):H149-H160
American journal of physiology. Heart and circulatory physiology 2021 Jul 1;321(1):H149-H160
TAGLN and High-mobility Group AT-Hook 2 (HMGA2) Complex Regulates TGF-β-induced Colorectal Cancer Metastasis.
Zhou H, Li L, Xie W, Wu L, Lin Y, He X
OncoTargets and therapy 2020;13:10489-10498
OncoTargets and therapy 2020;13:10489-10498
Development of prediction models for lymph node metastasis in endometrioid endometrial carcinoma.
Berg HF, Ju Z, Myrvold M, Fasmer KE, Halle MK, Hoivik EA, Westin SN, Trovik J, Haldorsen IS, Mills GB, Krakstad C, Werner HMJ
British journal of cancer 2020 Mar;122(7):1014-1022
British journal of cancer 2020 Mar;122(7):1014-1022
Lysophosphatidic acid promotes thrombus stability by inducing rapid formation of neutrophil extracellular traps: A new mechanism of thrombosis.
Li T, Peng R, Wang F, Hua L, Liu S, Han Z, Pei J, Pei S, Zhao Z, Jiang X, Chen X
Journal of thrombosis and haemostasis : JTH 2020 Aug;18(8):1952-1964
Journal of thrombosis and haemostasis : JTH 2020 Aug;18(8):1952-1964
Macrophage-associated wound healing contributes to African green monkey SIV pathogenesis control.
Barrenas F, Raehtz K, Xu C, Law L, Green RR, Silvestri G, Bosinger SE, Nishida A, Li Q, Lu W, Zhang J, Thomas MJ, Chang J, Smith E, Weiss JM, Dawoud RA, Richter GH, Trichel A, Ma D, Peng X, Komorowski J, Apetrei C, Pandrea I, Gale M Jr
Nature communications 2019 Nov 8;10(1):5101
Nature communications 2019 Nov 8;10(1):5101
IL-17 metabolically reprograms activated fibroblastic reticular cells for proliferation and survival.
Majumder S, Amatya N, Revu S, Jawale CV, Wu D, Rittenhouse N, Menk A, Kupul S, Du F, Raphael I, Bhattacharjee A, Siebenlist U, Hand TW, Delgoffe GM, Poholek AC, Gaffen SL, Biswas PS, McGeachy MJ
Nature immunology 2019 May;20(5):534-545
Nature immunology 2019 May;20(5):534-545
Rhodoptilometrin, a Crinoid-Derived Anthraquinone, Induces Cell Regeneration by Promoting Wound Healing and Oxidative Phosphorylation in Human Gingival Fibroblast Cells.
Tseng CC, Lai YC, Kuo TJ, Su JH, Sung PJ, Feng CW, Lin YY, Chen PC, Tai MH, Cheng SY, Kuo HM, Wen ZH
Marine drugs 2019 Feb 27;17(3)
Marine drugs 2019 Feb 27;17(3)
Degenerate intervertebral disc-like pH induces a catabolic mechanoresponse in human nucleus pulposus cells.
Hodson NW, Patel S, Richardson SM, Hoyland JA, Gilbert HTJ
JOR spine 2018 Mar;1(1):e1004
JOR spine 2018 Mar;1(1):e1004
Activation of hepatocyte growth factor/MET signaling initiates oncogenic transformation and enhances tumor aggressiveness in the murine prostate.
Mi J, Hooker E, Balog S, Zeng H, Johnson DT, He Y, Yu EJ, Wu H, Le V, Lee DH, Aldahl J, Gonzalgo ML, Sun Z
The Journal of biological chemistry 2018 Dec 28;293(52):20123-20136
The Journal of biological chemistry 2018 Dec 28;293(52):20123-20136
TGF-β1-induced differentiation of SHED into functional smooth muscle cells.
Xu JG, Zhu SY, Heng BC, Dissanayaka WL, Zhang CF
Stem cell research & therapy 2017 Jan 23;8(1):10
Stem cell research & therapy 2017 Jan 23;8(1):10
Decellularized extracellular matrix of human umbilical vein endothelial cells promotes endothelial differentiation of stem cells from exfoliated deciduous teeth.
Gong T, Heng BC, Xu J, Zhu S, Yuan C, Lo EC, Zhang C
Journal of biomedical materials research. Part A 2017 Apr;105(4):1083-1093
Journal of biomedical materials research. Part A 2017 Apr;105(4):1083-1093
Piceatannol Attenuates Renal Fibrosis Induced by Unilateral Ureteral Obstruction via Downregulation of Histone Deacetylase 4/5 or p38-MAPK Signaling.
Choi SY, Piao ZH, Jin L, Kim JH, Kim GR, Ryu Y, Lin MQ, Kim HS, Kee HJ, Jeong MH
PloS one 2016;11(11):e0167340
PloS one 2016;11(11):e0167340
Gallic acid prevents isoproterenol-induced cardiac hypertrophy and fibrosis through regulation of JNK2 signaling and Smad3 binding activity.
Ryu Y, Jin L, Kee HJ, Piao ZH, Cho JY, Kim GR, Choi SY, Lin MQ, Jeong MH
Scientific reports 2016 Oct 5;6:34790
Scientific reports 2016 Oct 5;6:34790
Characterization of chondrocyte sheets prepared using a co-culture method with temperature-responsive culture inserts.
Kokubo M, Sato M, Yamato M, Mitani G, Kutsuna T, Ebihara G, Okano T, Mochida J
Journal of tissue engineering and regenerative medicine 2016 Jun;10(6):486-95
Journal of tissue engineering and regenerative medicine 2016 Jun;10(6):486-95
Class I HDACs specifically regulate E-cadherin expression in human renal epithelial cells.
Choi SY, Kee HJ, Kurz T, Hansen FK, Ryu Y, Kim GR, Lin MQ, Jin L, Piao ZH, Jeong MH
Journal of cellular and molecular medicine 2016 Dec;20(12):2289-2298
Journal of cellular and molecular medicine 2016 Dec;20(12):2289-2298
Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro.
Heng BC, Zhu S, Xu J, Yuan C, Gong T, Zhang C
Tissue & cell 2016 Apr;48(2):133-43
Tissue & cell 2016 Apr;48(2):133-43
Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in SIV-infected macaques.
Klatt NR, Canary LA, Sun X, Vinton CL, Funderburg NT, Morcock DR, Quiñones M, Deming CB, Perkins M, Hazuda DJ, Miller MD, Lederman MM, Segre JA, Lifson JD, Haddad EK, Estes JD, Brenchley JM
The Journal of clinical investigation 2013 Feb;123(2):903-7
The Journal of clinical investigation 2013 Feb;123(2):903-7
RhoB deficiency in thymic medullary epithelium leads to early thymic atrophy.
Bravo-Nuevo A, O'Donnell R, Rosendahl A, Chung JH, Benjamin LE, Odaka C
International immunology 2011 Oct;23(10):593-600
International immunology 2011 Oct;23(10):593-600
The immunohistochemical characterization of human fetal olfactory bulb and olfactory ensheathing cells in culture as a source for clinical CNS restoration.
Liu K, Li Y, Wang H, Jiang X, Zhao Y, Sun D, Chen L, Young W, Huang H, Zhou C
Anatomical record (Hoboken, N.J. : 2007) 2010 Mar;293(3):359-69
Anatomical record (Hoboken, N.J. : 2007) 2010 Mar;293(3):359-69
Primary human dermal fibroblast interactions with open weave three-dimensional scaffolds prepared from synthetic human elastin.
Rnjak J, Li Z, Maitz PK, Wise SG, Weiss AS
Biomaterials 2009 Nov;30(32):6469-77
Biomaterials 2009 Nov;30(32):6469-77
The properties of bioengineered chondrocyte sheets for cartilage regeneration.
Mitani G, Sato M, Lee JI, Kaneshiro N, Ishihara M, Ota N, Kokubo M, Sakai H, Kikuchi T, Mochida J
BMC biotechnology 2009 Mar 6;9:17
BMC biotechnology 2009 Mar 6;9:17
A physical mechanism for coupling bone resorption and formation in adult human bone.
Andersen TL, Sondergaard TE, Skorzynska KE, Dagnaes-Hansen F, Plesner TL, Hauge EM, Plesner T, Delaisse JM
The American journal of pathology 2009 Jan;174(1):239-47
The American journal of pathology 2009 Jan;174(1):239-47
Molecular alteration of Ca(v)1.2 calcium channel in chronic myocardial infarction.
Liao P, Li G, Yu DJ, Yong TF, Wang JJ, Wang J, Soong TW
Pflugers Archiv : European journal of physiology 2009 Aug;458(4):701-11
Pflugers Archiv : European journal of physiology 2009 Aug;458(4):701-11
Flow shear stress affects macromolecular accumulation through modulation of internal elastic lamina fenestrae.
Guo ZY, Yan ZQ, Bai L, Zhang ML, Jiang ZL
Clinical biomechanics (Bristol, Avon) 2008;23 Suppl 1:S104-11
Clinical biomechanics (Bristol, Avon) 2008;23 Suppl 1:S104-11
Melatonin protects against epirubicin-induced cardiotoxicity.
Guven A, Yavuz O, Cam M, Ercan F, Bukan N, Comunoglu C
Acta histochemica 2007;109(1):52-60
Acta histochemica 2007;109(1):52-60
Uterine stretch regulates temporal and spatial expression of fibronectin protein and its alpha 5 integrin receptor in myometrium of unilaterally pregnant rats.
Shynlova O, Williams SJ, Draper H, White BG, MacPhee DJ, Lye SJ
Biology of reproduction 2007 Nov;77(5):880-8
Biology of reproduction 2007 Nov;77(5):880-8
Progesterone withdrawal up-regulates fibronectin and integrins during menstruation and repair in the rhesus macaque endometrium.
Cao W, Mah K, Carroll RS, Slayden OD, Brenner RM
Human reproduction (Oxford, England) 2007 Dec;22(12):3223-31
Human reproduction (Oxford, England) 2007 Dec;22(12):3223-31
Inhibition of NAD(P)H oxidase reduces fibronectin expression in stroke-prone renovascular hypertensive rat brain.
Cui C, Chen AF, Jiang Z, Wu Q, Lin J, Wen H, Zeng J
Clinical and experimental pharmacology & physiology 2007 Apr;34(4):304-9
Clinical and experimental pharmacology & physiology 2007 Apr;34(4):304-9
Expression of laminin and fibronectin in renal dysplasia.
Menon S, Kakkar N, Radotra BD
Pediatric and developmental pathology : the official journal of the Society for Pediatric Pathology and the Paediatric Pathology Society 2004 Nov-Dec;7(6):568-76
Pediatric and developmental pathology : the official journal of the Society for Pediatric Pathology and the Paediatric Pathology Society 2004 Nov-Dec;7(6):568-76
Angiotensin-converting enzyme inhibitor captopril prevents volume overload cardiomyopathy in experimental chronic aortic valve regurgitation.
Plante E, Gaudreau M, Lachance D, Drolet MC, Roussel E, Gauthier C, Lapointe E, Arsenault M, Couet J
Canadian journal of physiology and pharmacology 2004 Mar;82(3):191-9
Canadian journal of physiology and pharmacology 2004 Mar;82(3):191-9
Mammary localization and abundance of laminin, fibronectin, and collagen IV proteins in prepubertal heifers.
Berry SD, Howard RD, Akers RM
Journal of dairy science 2003 Sep;86(9):2864-74
Journal of dairy science 2003 Sep;86(9):2864-74
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of fibronectin was performed by loading insoluble fibronectin isolated via a deoxycholate assay from 3 x 105 mouse smooth muscle cells from three wild type (WT) or integrin knock-out (KO) mice per well onto an SDS-PAGE gel. Proteins were transferred to a nitrocellulose membrane and blocked with 5% milk in TBST for 1 hour at room temperature. The membrane was probed with a fibronectin monoclonal antibody (Product # MA5-11981) at a dilution of 1:1000 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG secondary antibody at a dilution of 1:2000 for 2 hours. Detection was performed using SuperSignal West Pico (Product # 34080). Data courtesy of the Innovators Program.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockout of Fibronectin was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR616644_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of Fibronectin was performed by loading 30 µg of Hep G2 Wild Type (Lane 1), Hep G2 Wild Type treated with 1X PTI for 4hrs (Lane 2), Hep G2 Cas9 (Lane 3), Hep G2 Cas9 treated with 1X PTI for 4hrs (Lane 4), Hep G2 Fibronectin KO (Lane 5) and Hep G2 Fibronectin KO treated with 1X PTI for 4hrs (Lane 6) whole cell extracts. The samples were electrophoresed using NuPAGE™ 3-8% Tris-Acetate Protein Gel (Product # EA0378BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-Fibronectin Monoclonal Antibody (FBN11) (Product # MA5-11981, 1:2,000 dilution) and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:5,000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Loss of signal upon CRISPR mediated knockout (KO) using the LentiArray™ CRISPR product line confirms that antibody is specific to Fibronectin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Fibronectin Monoclonal Antibody (FBN11) (Product # MA5-11981) and a ~260kDa band corresponding to Fibronectin was observed across cell lines tested . Whole cell extracts (30 µg lysate) of Hep G2 (Lane 1), Hep G2 treated with 1X PTI for 4h (Lane 2), Hep G2 treated with 10uM FLI-06 for 4h (Lane 3), HEL 92.1.7 (Lane 4), HEL 92.1.7 treated with 1X PTI for 4h (Lane 5), HEL 92.1.7 treated with 10uM FLI-06 for 4h (Lane 6) were electrophoresed using NuPAGE™ 3-8% Tris-Acetate Protein Gel (Product # EA0378BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # LC2002) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:2000) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:4000) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Anti-Fibronectin Monoclonal Antibody (FBN11) (Product # MA5-11981) showed enhanced pick up in positive cell line Hep G2 upon treatment with secretion blockers as compared to in negative cell line HEL 92.1.7 treated with the same blockers.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
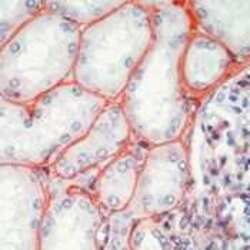
- Experimental details
- Formalin-fixed, paraffin-embedded human kidney stained with Fibronectin antibody1 using peroxidase-conjugate and AEC chromogen. Note staining of tubular basement membranes, mesangial cells and matrix.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
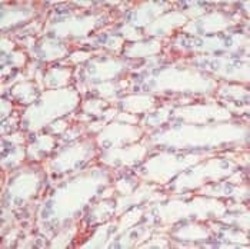
- Experimental details
- Formalin-fixed, paraffin-embedded human kidney stained with Fibronectin antibody1 using peroxidase-conjugate and AEC chromogen. Note staining of tubular basement membranes, mesangial cells and matrix.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Gallic acid pretreatment suppressed cardiac fibrosis in isoproterenol (ISP)-treated mice. Gallic acid administration was started 2 weeks before infusion of ISP (3 days) in mice; sham + vehicle, ISP + vehicle, and ISP + gallic acid group. ( A ) Representative images of hearts stained with Masson's trichrome, which were used to examine fibrosis (collagen deposition, scale bar, 100 mum). ( B ) Immunofluorescent staining with anti-collagen type I of heart sections from ISP-induced mice (scale bar, 50 mum). ( C ) Protein lysates from heart tissues were subjected to western blot with anti-collagen type I, anti-fibronectin, and anti-alpha-smooth muscle actin (SMA) antibodies. GAPDH was used as a loading control. ( D ) Collagen type I and fibronectin proteins were quantified by densitometry. ( E - G ) The mRNA levels of fibrosis-related genes (collagen type I, fibronectin, SMA) were analyzed by qRT-PCR. Data are presented as the means +- SD of 6~15 mice per group. * P < 0.05, * P < 0.01, and *** P < 0.001 versus sham + vehicle; ## P < 0.01versus ISP + vehicle.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 SHED-derived SMCs promote EC vessel formation. a Photomicrographs of EC-coated microbeads cultured in fibrin gels with SMC, SHED and SHED-induced SMC on the gel surface. b The schematic representation of calculating tube formation from each bead: (i) the number of vessels which sprout from the bead directly (in blue ); (ii) the total number of individual vessel segments (including blue and yellow ); and (iii) the total length of all the vessel segments. c Quantification of the vessel structures in the fibrin gel bead assay in the presence of SMC, SHED and SHED-induced SMC.*: p < 0.05 versus SMCs group, ^: p < 0.05 between SHED and SHED-derived SMCs. d Western blot showing fibronectin expression within each experimental group. All experiments were performed three times ( N = 3). ECs endothelial cells, SHED stem cells from human exfoliated deciduous teeth, SMCs smooth muscle cells
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 5 FIGURE Lysophosphatidic acid (LPA)-induced neutrophil extracellular traps (NETs) bound plasma proteins important for thrombus stability. Neutrophils without stimulation and NETs induced by LPA (10 muM, 240 min) were seeded on glass coverslips and incubated with 50% plasma in phosphate buffered saline. Samples were stained for (A) von Willebrand factor, (B) fibrinogen, and (C) fibronectin (400x) (n = 4)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 TSA regulates TGF -beta1-mediated ECM proteins and E-cadherin in HK 2 cells. ( A ) HK2 cells were treated with increasing concentrations of TSA (0, 10, 25, 50 100 and 250 nM) for 24 hrs and cell viability was measured by the MTT assay. ( B ) HK2 cells were co-treated with TGF-beta1 (5 ng/ml) and TSA (100 nM). ECM proteins and EMT markers were determined using Western blot analysis. Representative immunoblots are shown. beta-Actin was used as the loading control. ( C-F ) Quantification was performed by densitometry for at least four independent experiments. ( G ) Representative immunofluorescence images for E-cadherin in HK2 cells. Immunofluorescence staining was performed using anti-E-cadherin antibody and DAPI was used for staining cell nuclei. Merged images are shown. Scale bar represents 50 mum. *** P < 0.001 compared with untreated cells. # P < 0.05 and ### P < 0.001 compared with TGF-beta1-treated cells. NS, not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 PCI 34051 did not affect TGF -beta1-mediated EMT markers in HK 2 cells. ( A ) Cytotoxicity of PCI 34051 in HK 2 cells. ( B ) HK 2 cells were co-treated with TGF -beta1 (5 ng/ml) and PCI 34051 (5 muM) for 24 hrs. Western blot analysis was performed using collagen type I (collagen 1), fibronectin, N-cadherin, and E-cadherin antibodies. beta-Actin was used as the loading control. ( C-F ) Quantification was performed by densitometry for at least four independent experiments. ** P < 0.01 and *** P < 0.001 compared with untreated cells. # P < 0.05 and ### P < 0.001 compared with TGF -beta1 treated cells. NS , not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 LMK 235 did not affect TGF -beta1-mediated ECM proteins and EMT markers in HK 2 cells. ( A ) Cytotoxicity of LMK 235 in HK 2 cells. ( B ) HK 2 cells were co-treated with TGF -beta1 and LMK 235 for 24 hrs. Western blot analysis was performed using collagen I, fibronectin, N-cadherin, and E-cadherin antibodies. beta-Actin was used as the loading control. ( C-F ) Quantification was performed by densitometry for at least four independent experiments. * P < 0.05 and *** P < 0.001 compared with untreated cells. ## P < 0.01 and ### P < 0.001 compared with TGF -b1 treated cells. NS , not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 TGF-beta-induced cell migration and invasion was reversed by knockdown of TAGLN . ( A ) Cell migration and invasion in HCT116 cells (left panel) and representative images of crystal violet staining (right panel). ( B ) Cell migration and invasion in HT29 cells (left panel) and representative images of crystal violet staining (right panel). ( C ) Representative images of Western blotting assay of E-cadherin, vimentin, fibronectin, MMP2, MMP9, and GADPH . 200 x magnification. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Overexpression of HMGA2 restored altered CRC cell migration and invasion induced by inhibition of TAGLN . ( A ) Cell migration and invasion in HCT116 cells (left panel) and representative images of crystal violet staining (right panel). ( B ) Cell migration and invasion in HT29 cells (left panel) and representative images of crystal violet staining (right panel). ( C ) Representative images of Western blotting assay of nucleus and total TAGLN levels. ( D ) Representative images of Western blotting assay of E-cadherin, vimentin, fibronectin, MMP2, MMP9, and GADPH . 200 x magnification. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 TGF-beta-induced TAGLN expression increased migration and invasion of tumor promoting. ( A ) Image of tumors in nude mice inoculated with HT29 cells (Blank) and HT29 cells pretreated with TGF-beta and si- TAGLN (TGF-beta+si- TAGLN group). ( B ) Tumor volume was measured every four days. Tumor volume=1/2*L*W*W. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIG 6 Inhibition of Fn fibril formation increases the bacterial uptake in osteoblasts. Host cells were cultured for 2 days with a 70-kDa fragment of fibronectin to block Fn assembly. (A) Representative immunofluorescence microscopy images of Fn expression on pHOBs detected by anti-Fn antibody. (B) A549 cells and pHOBs cultured with the 70-kDa fragment of Fn as indicated and infected with S. aureus 6850 or S. carnosus TM300(pFnBA4) (MOI 50). One hour post infection, extracellular staphylococci were removed by lysostaphin treatment. Subsequently, the host cells were detached and the number of host cells was determined. Host cells were lysed and the number of viable intracellular bacteria was assessed by plate counting. Numbers of intracellular bacteria in non-pretreated cells were set to 100%. Data are means +- SD from 3 independent experiments. *, P < 0.05, one-way ANOVA followed by Dunnett's multiple-comparison test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 PCI34051 alleviates cardiac fibrosis in transverse aortic constriction (TAC) mice through the TGF- beta 1-Smad2/3 pathway. (a, b) Picrosirius red staining of mouse cardiac tissues; representative images and quantification are shown. Scale bar = 50 mu m. The expression levels of the fibrosis marker genes Col1a1 (c), Fn1 (d), Acta2 (e), and Tgfb1 (f) were determined using quantitative real-time polymerase chain reaction ( n = 5-6 per group). (g) The cardiac expression levels of Fn1, Acta2, Tgfb1, p-Smad2/3, and Smad2/3 were analyzed using western blotting. Actb was used as a loading control. Representative blots are shown. (h-k) Quantification of Fn1, Acta2, Tgfb1, and p-Smad2/3 to Smad2/3 ( n = 5-6 per group).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 PCI34051 alleviates pulmonary congestion and fibrosis in transverse aortic constriction (TAC) mice. (a) The ratio of lung weight-to-bodyweight (LW/BW) in the sham+vehicle, sham+PCI34051 (10 mg/kg bodyweight/day), TAC+vehicle, and TAC+PCI34051 (3, 10, or 30 mg/kg bodyweight/day) groups ( n = 5-6 per group) was determined. Representative images of mouse lungs (B). (b) Representative images of pulmonary tissues stained with hematoxylin and eosin. Scale bar = 50 mu m. (c) Representative images of pulmonary tissues stained with Picrosirius red. Scale bar = 50 mu m. (d) Pulmonary fibrosis was quantified using ImageJ software. (e-h) The mRNA levels of Col1a1 , Fn1 , Acta2 , and Tgfb1 were evaluated using quantitative real-time polymerase chain reaction ( n = 5-6 per group). (i-m) Representative western blots and quantification of Fn1, Acta2, Tgfb1, p-Smad2/3, and Smad2/3 levels in the pulmonary tissues ( n = 5-6 per group).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 PCI34051 or Hdac8 knockdown mitigates TGF- beta 1-induced fibrosis in rat cardiac fibroblasts. (a-e) Rat cardiac fibroblasts were pre-treated with TGF- beta 1 (10 ng/mL) for 1 h and cultured in the presence of vehicle or PCI34051 (10 mu M) for 8 h. mRNA levels of Hdac8 , Col1a1 , Fn1 , Acta2 , and Tgfb1 were evaluated using quantitative real-time polymerase chain reaction (qRT-PCR) ( n = 4-6 per group). (f-k) Representative immunoblots and quantification of Hdac8, Fn1, Acta2, Tgfb1, p-Smad2/3, and Smad2/3 levels in rat cardiac fibroblasts ( n = 6 per group). Actb was used as a loading control. (l-p) Rat cardiac fibroblasts were transfected with control or short-interfering RNA against Hdac8 and treated with TGF- beta 1 (10 ng/mL) for 9 h. The mRNA levels of Hdac8 , Fn1 , Acta2 , Tgfb1 , and Ace1 were determined using qRT-PCR ( n = 5 per group). (q-w) Representative immunoblots and quantification of Hdac8, Ace1, Fn1, Acta2, Tgfb1, p-Smad2/3, and Smad2/3 levels in rat cardiac fibroblasts ( n = 4 per group). Actb was used as a loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Immunohistochemistry of agarose gels following: (A) 1 and 7 days of culture in standard medium at pH 7.4 and (B) a subsequent 24 hours at pH 7.1 or 6.5. Nucleus pulposus (NP) cells/agarose gels were snap frozen in liquid nitrogen and cryosectioned. Antibodies against the pericellular marker type VI collagen, the integrin receptor alpha5beta1, and separate subunits alpha5 and beta1 and the extracellular matrix protein fibronectin (the predominant ligand of alpha5beta1 integrins) were used to probe sections. (A) NP cells did not appear to express type VI collagen or alpha5beta1 integrin following 1 day of preculture. However, NP cells did express both type VI collagen and alpha5beta1 following 7 days of preculture (black arrows indicate cells expressing proteins which were not expressed at day 0), (B) with 24 hours of culture at pH 7.1 or 6.5 not affecting this expression. (A) NP cells did express the alpha5 and beta1 integrin subunits, as well as fibronectin, following 1 and 7 days of preculture in standard medium, (B) with culture at pH 7.1 or 6.5 having no noticeable affect on the expression
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 CGSA showing axolotl wound healing signatures in AGMs. a Heatmap of Wound Healing Network genes in axolotl wound repair and acute SIV infection, ordered by day 1 of aquatic axolotl wound repair. The biological functions that correlate with D1 axolotl wound repair, and their associated genes are shown on the right. b A protein interaction network between proteins encoded by genes in the Wound Healing Network. Red and blue nodes indicate up and downregulation in aquatic (left) and terrestrial (right) axolotl at day 1. c Immunohistochemistry staining of fibronectin (FN) in rectal mucosa at baseline, D1 and D3 post SIV inoculation. Right panels show quantification by pixel density, middle and left panels show fibronectin protein expression in lamina propria. Representative images are shown below. The scale bar included in each image represents a length of 50 mum
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot