Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Western blot [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- NB500-126 - Provider product page

- Provider
- Novus Biologicals
- Proper citation
- Novus Cat#NB500-126, RRID:AB_10078274
- Product name
- Rabbit Polyclonal HDAC3 Antibody
- Antibody type
- Polyclonal
- Description
- IgG purified. Anti-Histone Deacetylase 3 specifically recognizes histone deacetylase 3 by immunoblotting and immunoprecipitation (50 kD).
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Antigen sequence
The epitope recognized by the antib
ody is resistant to routine formali
n-fixation and paraffin-embedding.- Isotype
- IgG
- Vial size
- 0.05 ml
- Concentration
- 12.5 mg/ml
- Storage
- Store at -20C. Avoid freeze-thaw cycles.
Submitted references Epigenetic Regulation of the Blimp-1 Gene (Prdm1) in B Cells Involves Bach2 and Histone Deacetylase 3.
Nuclear receptor corepressor is a novel regulator of phosphatidylinositol 3-kinase signaling.
SUMO promotes HDAC-mediated transcriptional repression.
SUMO promotes HDAC-mediated transcriptional repression.
Expression and functional characterization of recombinant human HDAC1 and HDAC3.
Role of class I and class II histone deacetylases in carcinoma cells using siRNA.
Role of class I and class II histone deacetylases in carcinoma cells using siRNA.
Three proteins define a class of human histone deacetylases related to yeast Hda1p.
Three proteins define a class of human histone deacetylases related to yeast Hda1p.
A role for histone deacetylase activity in HDAC1-mediated transcriptional repression.
A role for histone deacetylase activity in HDAC1-mediated transcriptional repression.
Tanaka H, Muto A, Shima H, Katoh Y, Sax N, Tajima S, Brydun A, Ikura T, Yoshizawa N, Masai H, Hoshikawa Y, Noda T, Nio M, Ochiai K, Igarashi K
The Journal of biological chemistry 2016 Mar 18;291(12):6316-30
The Journal of biological chemistry 2016 Mar 18;291(12):6316-30
Nuclear receptor corepressor is a novel regulator of phosphatidylinositol 3-kinase signaling.
Furuya F, Guigon CJ, Zhao L, Lu C, Hanover JA, Cheng SY
Molecular and cellular biology 2007 Sep;27(17):6116-26
Molecular and cellular biology 2007 Sep;27(17):6116-26
SUMO promotes HDAC-mediated transcriptional repression.
Yang SH, Sharrocks AD
Molecular cell 2004 Feb 27;13(4):611-7
Molecular cell 2004 Feb 27;13(4):611-7
SUMO promotes HDAC-mediated transcriptional repression.
Yang SH, Sharrocks AD
Molecular cell 2004 Feb 27;13(4):611-7
Molecular cell 2004 Feb 27;13(4):611-7
Expression and functional characterization of recombinant human HDAC1 and HDAC3.
Li J, Staver MJ, Curtin ML, Holms JH, Frey RR, Edalji R, Smith R, Michaelides MR, Davidsen SK, Glaser KB
Life sciences 2004 Apr 16;74(22):2693-705
Life sciences 2004 Apr 16;74(22):2693-705
Role of class I and class II histone deacetylases in carcinoma cells using siRNA.
Glaser KB, Li J, Staver MJ, Wei RQ, Albert DH, Davidsen SK
Biochemical and biophysical research communications 2003 Oct 17;310(2):529-36
Biochemical and biophysical research communications 2003 Oct 17;310(2):529-36
Role of class I and class II histone deacetylases in carcinoma cells using siRNA.
Glaser KB, Li J, Staver MJ, Wei RQ, Albert DH, Davidsen SK
Biochemical and biophysical research communications 2003 Oct 17;310(2):529-36
Biochemical and biophysical research communications 2003 Oct 17;310(2):529-36
Three proteins define a class of human histone deacetylases related to yeast Hda1p.
Grozinger CM, Hassig CA, Schreiber SL
Proceedings of the National Academy of Sciences of the United States of America 1999 Apr 27;96(9):4868-73
Proceedings of the National Academy of Sciences of the United States of America 1999 Apr 27;96(9):4868-73
Three proteins define a class of human histone deacetylases related to yeast Hda1p.
Grozinger CM, Hassig CA, Schreiber SL
Proceedings of the National Academy of Sciences of the United States of America 1999 Apr 27;96(9):4868-73
Proceedings of the National Academy of Sciences of the United States of America 1999 Apr 27;96(9):4868-73
A role for histone deacetylase activity in HDAC1-mediated transcriptional repression.
Hassig CA, Tong JK, Fleischer TC, Owa T, Grable PG, Ayer DE, Schreiber SL
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 31;95(7):3519-24
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 31;95(7):3519-24
A role for histone deacetylase activity in HDAC1-mediated transcriptional repression.
Hassig CA, Tong JK, Fleischer TC, Owa T, Grable PG, Ayer DE, Schreiber SL
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 31;95(7):3519-24
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 31;95(7):3519-24
No comments: Submit comment
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
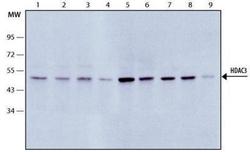
- Experimental details
- Western Blot: HDAC3 Antibody [NB500-126] - Cell line lysates (Lanes1. HEK-293T; 2. HeLa; 3. K562; 4. COS-7; 5. CHO; 6. NIH-3T3; 7. Neuro-2a; 8. PC-12; 9. NRK) were separated on SDS-PAGE and probed with 1:5,000 Rabbit Anti-HDAC3 (NB500-126). The antibody was developed using Goat Anti-Rabbit IgG-Peroxidase.
- Submitted by
- Novus Biologicals (provider)
- Main image
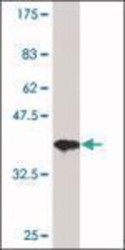
- Experimental details
- Western Blot: HDAC3 Antibody [NB500-126] - Analysis of HDAC3 Antibody [NB500-126]-Monoclonal Anti-HDAC3 antibody produced in mouse clone 3E11, purified immunoglobulin, buffered aqueous solution
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry