Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Immunohistochemistry [3]
- Other assay [26]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 34-5700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Connexin 32 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- 34-5700 was used in the immunofluorescence analysis to successfully detect Connexin 32 in gap junctions from mouse liver samples.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Re-evaluation of connexins associated with motoneurons in rodent spinal cord, sexually dimorphic motor nuclei and trigeminal motor nucleus.
The growth pattern of transplanted normal and nodular hepatocytes.
Functional heterotypic interactions between astrocyte and oligodendrocyte connexins.
Ablation of connexin30 in transgenic mice alters expression patterns of connexin26 and connexin32 in glial cells and leptomeninges.
Phenotype and growth behavior of residual β-catenin-positive hepatocytes in livers of β-catenin-deficient mice.
Connexin expression and gap junctional coupling in human cumulus cells: contribution to embryo quality.
Tissue-specific cross-reactivity of connexin32 antibodies: problems and solutions unique to the central nervous system.
Ablation of Cx47 in transgenic mice leads to the loss of MUPP1, ZONAB and multiple connexins at oligodendrocyte-astrocyte gap junctions.
Identification of connexin36 in gap junctions between neurons in rodent locus coeruleus.
Connexin-47 and connexin-32 in gap junctions of oligodendrocyte somata, myelin sheaths, paranodal loops and Schmidt-Lanterman incisures: implications for ionic homeostasis and potassium siphoning.
Connexin47, connexin29 and connexin32 co-expression in oligodendrocytes and Cx47 association with zonula occludens-1 (ZO-1) in mouse brain.
Inhibition of Mist1 homodimer formation induces pancreatic acinar-to-ductal metaplasia.
Connexin29 and connexin32 at oligodendrocyte and astrocyte gap junctions and in myelin of the mouse central nervous system.
Coupling of astrocyte connexins Cx26, Cx30, Cx43 to oligodendrocyte Cx29, Cx32, Cx47: Implications from normal and connexin32 knockout mice.
Bautista W, Rash JE, Vanderpool KG, Yasumura T, Nagy JI
The European journal of neuroscience 2014 Mar;39(5):757-70
The European journal of neuroscience 2014 Mar;39(5):757-70
The growth pattern of transplanted normal and nodular hepatocytes.
Doratiotto S, Krause P, Serra MP, Marongiu F, Sini M, Koenig S, Laconi E
Histochemistry and cell biology 2011 Jun;135(6):581-91
Histochemistry and cell biology 2011 Jun;135(6):581-91
Functional heterotypic interactions between astrocyte and oligodendrocyte connexins.
Magnotti LM, Goodenough DA, Paul DL
Glia 2011 Jan;59(1):26-34
Glia 2011 Jan;59(1):26-34
Ablation of connexin30 in transgenic mice alters expression patterns of connexin26 and connexin32 in glial cells and leptomeninges.
Lynn BD, Tress O, May D, Willecke K, Nagy JI
The European journal of neuroscience 2011 Dec;34(11):1783-93
The European journal of neuroscience 2011 Dec;34(11):1783-93
Phenotype and growth behavior of residual β-catenin-positive hepatocytes in livers of β-catenin-deficient mice.
Braeuning A, Singh Y, Rignall B, Buchmann A, Hammad S, Othman A, von Recklinghausen I, Godoy P, Hoehme S, Drasdo D, Hengstler JG, Schwarz M
Histochemistry and cell biology 2010 Nov;134(5):469-81
Histochemistry and cell biology 2010 Nov;134(5):469-81
Connexin expression and gap junctional coupling in human cumulus cells: contribution to embryo quality.
Wang HX, Tong D, El-Gehani F, Tekpetey FR, Kidder GM
Journal of cellular and molecular medicine 2009 May;13(5):972-84
Journal of cellular and molecular medicine 2009 May;13(5):972-84
Tissue-specific cross-reactivity of connexin32 antibodies: problems and solutions unique to the central nervous system.
Fowler SL, McLean AC, Bennett SA
Cell communication & adhesion 2009 Dec;16(5-6):117-30
Cell communication & adhesion 2009 Dec;16(5-6):117-30
Ablation of Cx47 in transgenic mice leads to the loss of MUPP1, ZONAB and multiple connexins at oligodendrocyte-astrocyte gap junctions.
Li X, Penes M, Odermatt B, Willecke K, Nagy JI
The European journal of neuroscience 2008 Oct;28(8):1503-17
The European journal of neuroscience 2008 Oct;28(8):1503-17
Identification of connexin36 in gap junctions between neurons in rodent locus coeruleus.
Rash JE, Olson CO, Davidson KG, Yasumura T, Kamasawa N, Nagy JI
Neuroscience 2007 Jul 29;147(4):938-56
Neuroscience 2007 Jul 29;147(4):938-56
Connexin-47 and connexin-32 in gap junctions of oligodendrocyte somata, myelin sheaths, paranodal loops and Schmidt-Lanterman incisures: implications for ionic homeostasis and potassium siphoning.
Kamasawa N, Sik A, Morita M, Yasumura T, Davidson KG, Nagy JI, Rash JE
Neuroscience 2005;136(1):65-86
Neuroscience 2005;136(1):65-86
Connexin47, connexin29 and connexin32 co-expression in oligodendrocytes and Cx47 association with zonula occludens-1 (ZO-1) in mouse brain.
Li X, Ionescu AV, Lynn BD, Lu S, Kamasawa N, Morita M, Davidson KG, Yasumura T, Rash JE, Nagy JI
Neuroscience 2004;126(3):611-30
Neuroscience 2004;126(3):611-30
Inhibition of Mist1 homodimer formation induces pancreatic acinar-to-ductal metaplasia.
Zhu L, Tran T, Rukstalis JM, Sun P, Damsz B, Konieczny SF
Molecular and cellular biology 2004 Apr;24(7):2673-81
Molecular and cellular biology 2004 Apr;24(7):2673-81
Connexin29 and connexin32 at oligodendrocyte and astrocyte gap junctions and in myelin of the mouse central nervous system.
Nagy JI, Ionescu AV, Lynn BD, Rash JE
The Journal of comparative neurology 2003 Sep 22;464(3):356-70
The Journal of comparative neurology 2003 Sep 22;464(3):356-70
Coupling of astrocyte connexins Cx26, Cx30, Cx43 to oligodendrocyte Cx29, Cx32, Cx47: Implications from normal and connexin32 knockout mice.
Nagy JI, Ionescu AV, Lynn BD, Rash JE
Glia 2003 Dec;44(3):205-18
Glia 2003 Dec;44(3):205-18
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of connexin 32 was performed on sections of adult mouse liver. Tissue sections on slides were probed for 24 h at 4°C in a humidified chamber with rabbit polyclonal anti-Cx32 (Product # 34-5700), at an antibody concentration of 1-2 µg/mL diluted in 50 mM Tris-HCl, pH 7.4, containing 1.5% NaCl, 0.3% Triton X-100 (TBST) and 4% normal goat serum. After overnight incubation, sections were washed extensively for 1 h in TBST, and detection of primary antibody was performed for 1.5 h at room temperature with AlexaFluor-488-conjugated donkey anti-rabbit diluted 1:600 in TBST. Sections were then was in TBST, then in TBS (without triton) and then coversliped with anti-fade medium. Images were taken on a Zeiss Z2 scanning microscope at x40 objective magnification, and show immunofluorescence labelling of Cx32 localized at gap junctions between liver hepatocytes. Data courtesy of Dr. James Nagy's lab.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Connexin 32 showing staining in the membrane of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Connexin 32 polyclonal antibody (Product # 34-5700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Connexin 32 showing staining in the membrane of paraffin-embedded mouse liver tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Connexin 32 polyclonal antibody (Product # 34-5700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
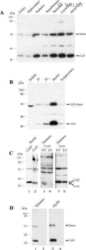
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Immunofluorescence colocalization of DPPIV ( red ) and Cx32 ( green ) in clusters of normal ( a , b , c , g ) and nodular ( d , e , f , h ) hepatocytes. a , d DPPIV only; b , e Cx32 staining only; c , f , g , h staining for DPPIV and Cx32 were merged. Note the regular pattern of expression of Cx32 in normal hepatocyte clusters ( b , c , g ), while the gap junction protein is almost undetected in the donor-derived nodular lesion ( e , f , h ). See "" Materials and methods "" for details
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Pairs of serial sections stained for immunohistochemical detection of Cx32 ( left panels , a , c , e , g , punctuated blue spots ) or Ki67 ( right panels , b , d , f , h , intense blue nuclei ). b , d , f , g , h Processed for DPPIV enzyme activity. Note that different rates of Ki67 labelling in nodular hepatocyte clusters ( b : 2%, d : 11%, f : 34%), are associated with similar low levels of Cx32 expression ( a , c , e ). However, significant Ki67 labelling in normal hepatocyte clusters ( h ) is paired with normal distribution of Cx32 gap junction protein ( g ). See "" Materials and methods "" for details
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry