Antibody data
- Antibody Data
- Antigen structure
- References [39]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Other assay [26]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-788G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-EGFR (Tyr1068) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Purified from rabbit serum by sequential epitope-specific chromatography, this product contains enough material for 10 mini-blots. The antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated epidermal growth factor receptor (EGFR). The final product is generated by affinity chromatography using an EGFR-derived peptide that is phosphorylated at tyrosine 1068. Positive controls used in western analysis were human epidermoid carcinoma (A431) cells +/- EGF.
- Reactivity
- Human, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Suppression of heparan sulfation re-sensitizes YAP1-driven melanoma to MAPK pathway inhibitors.
Lamellipodin-RICTOR Signaling Mediates Glioblastoma Cell Invasion and Radiosensitivity Downstream of EGFR.
EWI-2 controls nucleocytoplasmic shuttling of EGFR signaling molecules and miRNA sorting in exosomes to inhibit prostate cancer cell metastasis.
FAM188B Downregulation Sensitizes Lung Cancer Cells to Anoikis via EGFR Downregulation and Inhibits Tumor Metastasis In Vivo.
RNF144a induces ERK-dependent cell death under oxidative stress via downregulation of vaccinia-related kinase 3.
Entosis and apical cell extrusion constitute a tumor-suppressive mechanism downstream of Matriptase.
IGF2 Autocrine-Mediated IGF1R Activation Is a Clinically Relevant Mechanism of Osimertinib Resistance in Lung Cancer.
MET as resistance factor for afatinib therapy and motility driver in gastric cancer cells.
Overcoming acquired resistance to HSP90 inhibition by targeting JAK-STAT signalling in triple-negative breast cancer.
YXQ-EQ Induces Apoptosis and Inhibits Signaling Pathways Important for Metastasis in Non-Small Cell Lung Carcinoma Cells.
Antitumor Activity of Osimertinib, an Irreversible Mutant-Selective EGFR Tyrosine Kinase Inhibitor, in NSCLC Harboring EGFR Exon 20 Insertions.
Effects of trastuzumab and afatinib on kinase activity in gastric cancer cell lines.
E-cadherin integrates mechanotransduction and EGFR signaling to control junctional tissue polarization and tight junction positioning.
Evaluation of epidermal growth factor receptor signaling effects in gastric cancer cell lines by detailed motility-focused phenotypic characterization linked with molecular analysis.
Characterization of the efficacies of osimertinib and nazartinib against cells expressing clinically relevant epidermal growth factor receptor mutations.
Anti-tumor efficacy of theliatinib in esophageal cancer patient-derived xenografts models with epidermal growth factor receptor (EGFR) overexpression and gene amplification.
Influence of the HER receptor ligand system on sensitivity to cetuximab and trastuzumab in gastric cancer cell lines.
Amplification of EGFR Wild-Type Alleles in Non-Small Cell Lung Cancer Cells Confers Acquired Resistance to Mutation-Selective EGFR Tyrosine Kinase Inhibitors.
ZEB1 Mediates Acquired Resistance to the Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors in Non-Small Cell Lung Cancer.
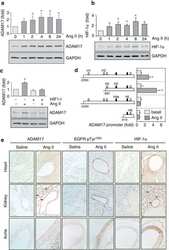
Vascular ADAM17 as a Novel Therapeutic Target in Mediating Cardiovascular Hypertrophy and Perivascular Fibrosis Induced by Angiotensin II.
Activation of EGFR Bypass Signaling by TGFα Overexpression Induces Acquired Resistance to Alectinib in ALK-Translocated Lung Cancer Cells.
In vitro modeling to determine mutation specificity of EGFR tyrosine kinase inhibitors against clinically relevant EGFR mutants in non-small-cell lung cancer.
Vascular induction of a disintegrin and metalloprotease 17 by angiotensin II through hypoxia inducible factor 1α.
Effects of AKT inhibition on HGF-mediated erlotinib resistance in non-small cell lung cancer cell lines.
Short-course treatment with gefitinib enhances curative potential of radiation therapy in a mouse model of human non-small cell lung cancer.
ADAM17 silencing by adenovirus encoding miRNA-embedded siRNA revealed essential signal transduction by angiotensin II in vascular smooth muscle cells.
Activated EGFR stimulates MUC1 expression in human uterine and pancreatic cancer cell lines.
Activation of the FGF2-FGFR1 autocrine pathway: a novel mechanism of acquired resistance to gefitinib in NSCLC.
A functional siRNA screen identifies genes modulating angiotensin II-mediated EGFR transactivation.
EGFR/Src/Akt signaling modulates Sox2 expression and self-renewal of stem-like side-population cells in non-small cell lung cancer.
Association of amphiregulin with the cetuximab sensitivity of gastric cancer cell lines.
MAPK and PI3K signalling differentially regulate angiogenic and lymphangiogenic cytokine secretion in squamous cell carcinoma of the head and neck.
Determinants of response to epidermal growth factor receptor tyrosine kinase inhibition in squamous cell carcinoma of the head and neck.
Wounding-induced synthesis of hyaluronic acid in organotypic epidermal cultures requires the release of heparin-binding egf and activation of the EGFR.
Quantitative measurement of epidermal growth factor receptor-mitogen-activated protein kinase signal transduction using a nine-plex, peptide-based immunoassay.
Hydrocortisone and indomethacin negatively modulate EGF-R signaling in human fetal intestine.
Tumor growth inhibition with cetuximab and chemotherapy in non-small cell lung cancer xenografts expressing wild-type and mutated epidermal growth factor receptor.
G protein coupling and second messenger generation are indispensable for metalloprotease-dependent, heparin-binding epidermal growth factor shedding through angiotensin II type-1 receptor.
Aplidin induces apoptosis in human cancer cells via glutathione depletion and sustained activation of the epidermal growth factor receptor, Src, JNK, and p38 MAPK.
Dieter SM, Lovecchio D, Pataskar A, Zowada MK, Körner PR, Khalizieva A, van Tellingen O, Jäger D, Glimm H, Agami R
Oncogene 2022 Aug;41(32):3953-3968
Oncogene 2022 Aug;41(32):3953-3968
Lamellipodin-RICTOR Signaling Mediates Glioblastoma Cell Invasion and Radiosensitivity Downstream of EGFR.
Moritz S, Krause M, Schlatter J, Cordes N, Vehlow A
Cancers 2021 Oct 24;13(21)
Cancers 2021 Oct 24;13(21)
EWI-2 controls nucleocytoplasmic shuttling of EGFR signaling molecules and miRNA sorting in exosomes to inhibit prostate cancer cell metastasis.
Fu C, Zhang Q, Wang A, Yang S, Jiang Y, Bai L, Wei Q
Molecular oncology 2021 May;15(5):1543-1565
Molecular oncology 2021 May;15(5):1543-1565
FAM188B Downregulation Sensitizes Lung Cancer Cells to Anoikis via EGFR Downregulation and Inhibits Tumor Metastasis In Vivo.
Jang EJ, Sung JY, Yoo HE, Jang H, Shim J, Oh ES, Goh SH, Kim YN
Cancers 2021 Jan 11;13(2)
Cancers 2021 Jan 11;13(2)
RNF144a induces ERK-dependent cell death under oxidative stress via downregulation of vaccinia-related kinase 3.
Han SH, Kim KT
Journal of cell science 2020 Nov 9;133(21)
Journal of cell science 2020 Nov 9;133(21)
Entosis and apical cell extrusion constitute a tumor-suppressive mechanism downstream of Matriptase.
Armistead J, Hatzold J, van Roye A, Fahle E, Hammerschmidt M
The Journal of cell biology 2020 Feb 3;219(2)
The Journal of cell biology 2020 Feb 3;219(2)
IGF2 Autocrine-Mediated IGF1R Activation Is a Clinically Relevant Mechanism of Osimertinib Resistance in Lung Cancer.
Manabe T, Yasuda H, Terai H, Kagiwada H, Hamamoto J, Ebisudani T, Kobayashi K, Masuzawa K, Ikemura S, Kawada I, Hayashi Y, Fukui K, Horimoto K, Fukunaga K, Soejima K
Molecular cancer research : MCR 2020 Apr;18(4):549-559
Molecular cancer research : MCR 2020 Apr;18(4):549-559
MET as resistance factor for afatinib therapy and motility driver in gastric cancer cells.
Ebert K, Mattes J, Kunzke T, Zwingenberger G, Luber B
PloS one 2019;14(9):e0223225
PloS one 2019;14(9):e0223225
Overcoming acquired resistance to HSP90 inhibition by targeting JAK-STAT signalling in triple-negative breast cancer.
Mumin NH, Drobnitzky N, Patel A, Lourenco LM, Cahill FF, Jiang Y, Kong A, Ryan AJ
BMC cancer 2019 Jan 24;19(1):102
BMC cancer 2019 Jan 24;19(1):102
YXQ-EQ Induces Apoptosis and Inhibits Signaling Pathways Important for Metastasis in Non-Small Cell Lung Carcinoma Cells.
Yan X, Shen H, Jiang H, Hu D, Wang J, Wu X
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2018;49(3):911-919
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2018;49(3):911-919
Antitumor Activity of Osimertinib, an Irreversible Mutant-Selective EGFR Tyrosine Kinase Inhibitor, in NSCLC Harboring EGFR Exon 20 Insertions.
Floc'h N, Martin MJ, Riess JW, Orme JP, Staniszewska AD, Ménard L, Cuomo ME, O'Neill DJ, Ward RA, Finlay MRV, McKerrecher D, Cheng M, Vang DP, Burich RA, Keck JG, Gandara DR, Mack PC, Cross DAE
Molecular cancer therapeutics 2018 May;17(5):885-896
Molecular cancer therapeutics 2018 May;17(5):885-896
Effects of trastuzumab and afatinib on kinase activity in gastric cancer cell lines.
Keller S, Zwingenberger G, Ebert K, Hasenauer J, Wasmuth J, Maier D, Haffner I, Schierle K, Weirich G, Luber B
Molecular oncology 2018 Apr;12(4):441-462
Molecular oncology 2018 Apr;12(4):441-462
E-cadherin integrates mechanotransduction and EGFR signaling to control junctional tissue polarization and tight junction positioning.
Rübsam M, Mertz AF, Kubo A, Marg S, Jüngst C, Goranci-Buzhala G, Schauss AC, Horsley V, Dufresne ER, Moser M, Ziegler W, Amagai M, Wickström SA, Niessen CM
Nature communications 2017 Nov 1;8(1):1250
Nature communications 2017 Nov 1;8(1):1250
Evaluation of epidermal growth factor receptor signaling effects in gastric cancer cell lines by detailed motility-focused phenotypic characterization linked with molecular analysis.
Keller S, Kneissl J, Grabher-Meier V, Heindl S, Hasenauer J, Maier D, Mattes J, Winter P, Luber B
BMC cancer 2017 Dec 13;17(1):845
BMC cancer 2017 Dec 13;17(1):845
Characterization of the efficacies of osimertinib and nazartinib against cells expressing clinically relevant epidermal growth factor receptor mutations.
Masuzawa K, Yasuda H, Hamamoto J, Nukaga S, Hirano T, Kawada I, Naoki K, Soejima K, Betsuyaku T
Oncotarget 2017 Dec 1;8(62):105479-105491
Oncotarget 2017 Dec 1;8(62):105479-105491
Anti-tumor efficacy of theliatinib in esophageal cancer patient-derived xenografts models with epidermal growth factor receptor (EGFR) overexpression and gene amplification.
Ren Y, Zheng J, Fan S, Wang L, Cheng M, Shi D, Zhang W, Tang R, Yu Y, Jiao L, Ni J, Yang H, Cai H, Yin F, Chen Y, Zhou F, Zhang W, Qing W, Su W
Oncotarget 2017 Aug 1;8(31):50832-50844
Oncotarget 2017 Aug 1;8(31):50832-50844
Influence of the HER receptor ligand system on sensitivity to cetuximab and trastuzumab in gastric cancer cell lines.
Kneissl J, Hartmann A, Pfarr N, Erlmeier F, Lorber T, Keller S, Zwingenberger G, Weichert W, Luber B
Journal of cancer research and clinical oncology 2017 Apr;143(4):573-600
Journal of cancer research and clinical oncology 2017 Apr;143(4):573-600
Amplification of EGFR Wild-Type Alleles in Non-Small Cell Lung Cancer Cells Confers Acquired Resistance to Mutation-Selective EGFR Tyrosine Kinase Inhibitors.
Nukaga S, Yasuda H, Tsuchihara K, Hamamoto J, Masuzawa K, Kawada I, Naoki K, Matsumoto S, Mimaki S, Ikemura S, Goto K, Betsuyaku T, Soejima K
Cancer research 2017 Apr 15;77(8):2078-2089
Cancer research 2017 Apr 15;77(8):2078-2089
ZEB1 Mediates Acquired Resistance to the Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors in Non-Small Cell Lung Cancer.
Yoshida T, Song L, Bai Y, Kinose F, Li J, Ohaegbulam KC, Muñoz-Antonia T, Qu X, Eschrich S, Uramoto H, Tanaka F, Nasarre P, Gemmill RM, Roche J, Drabkin HA, Haura EB
PloS one 2016;11(1):e0147344
PloS one 2016;11(1):e0147344
Vascular ADAM17 as a Novel Therapeutic Target in Mediating Cardiovascular Hypertrophy and Perivascular Fibrosis Induced by Angiotensin II.
Takayanagi T, Forrester SJ, Kawai T, Obama T, Tsuji T, Elliott KJ, Nuti E, Rossello A, Kwok HF, Scalia R, Rizzo V, Eguchi S
Hypertension (Dallas, Tex. : 1979) 2016 Oct;68(4):949-955
Hypertension (Dallas, Tex. : 1979) 2016 Oct;68(4):949-955
Activation of EGFR Bypass Signaling by TGFα Overexpression Induces Acquired Resistance to Alectinib in ALK-Translocated Lung Cancer Cells.
Tani T, Yasuda H, Hamamoto J, Kuroda A, Arai D, Ishioka K, Ohgino K, Miyawaki M, Kawada I, Naoki K, Hayashi Y, Betsuyaku T, Soejima K
Molecular cancer therapeutics 2016 Jan;15(1):162-71
Molecular cancer therapeutics 2016 Jan;15(1):162-71
In vitro modeling to determine mutation specificity of EGFR tyrosine kinase inhibitors against clinically relevant EGFR mutants in non-small-cell lung cancer.
Hirano T, Yasuda H, Tani T, Hamamoto J, Oashi A, Ishioka K, Arai D, Nukaga S, Miyawaki M, Kawada I, Naoki K, Costa DB, Kobayashi SS, Betsuyaku T, Soejima K
Oncotarget 2015 Nov 17;6(36):38789-803
Oncotarget 2015 Nov 17;6(36):38789-803
Vascular induction of a disintegrin and metalloprotease 17 by angiotensin II through hypoxia inducible factor 1α.
Obama T, Takayanagi T, Kobayashi T, Bourne AM, Elliott KJ, Charbonneau M, Dubois CM, Eguchi S
American journal of hypertension 2015 Jan;28(1):10-4
American journal of hypertension 2015 Jan;28(1):10-4
Effects of AKT inhibition on HGF-mediated erlotinib resistance in non-small cell lung cancer cell lines.
Holland WS, Chinn DC, Lara PN Jr, Gandara DR, Mack PC
Journal of cancer research and clinical oncology 2015 Apr;141(4):615-26
Journal of cancer research and clinical oncology 2015 Apr;141(4):615-26
Short-course treatment with gefitinib enhances curative potential of radiation therapy in a mouse model of human non-small cell lung cancer.
Bokobza SM, Jiang Y, Weber AM, Devery AM, Ryan AJ
International journal of radiation oncology, biology, physics 2014 Mar 15;88(4):947-54
International journal of radiation oncology, biology, physics 2014 Mar 15;88(4):947-54
ADAM17 silencing by adenovirus encoding miRNA-embedded siRNA revealed essential signal transduction by angiotensin II in vascular smooth muscle cells.
Elliott KJ, Bourne AM, Takayanagi T, Takaguri A, Kobayashi T, Eguchi K, Eguchi S
Journal of molecular and cellular cardiology 2013 Sep;62:1-7
Journal of molecular and cellular cardiology 2013 Sep;62:1-7
Activated EGFR stimulates MUC1 expression in human uterine and pancreatic cancer cell lines.
Neeraja Dharmaraj, Engel BJ, Carson DD
Journal of cellular biochemistry 2013 Oct;114(10):2314-22
Journal of cellular biochemistry 2013 Oct;114(10):2314-22
Activation of the FGF2-FGFR1 autocrine pathway: a novel mechanism of acquired resistance to gefitinib in NSCLC.
Terai H, Soejima K, Yasuda H, Nakayama S, Hamamoto J, Arai D, Ishioka K, Ohgino K, Ikemura S, Sato T, Yoda S, Satomi R, Naoki K, Betsuyaku T
Molecular cancer research : MCR 2013 Jul;11(7):759-67
Molecular cancer research : MCR 2013 Jul;11(7):759-67
A functional siRNA screen identifies genes modulating angiotensin II-mediated EGFR transactivation.
George AJ, Purdue BW, Gould CM, Thomas DW, Handoko Y, Qian H, Quaife-Ryan GA, Morgan KA, Simpson KJ, Thomas WG, Hannan RD
Journal of cell science 2013 Dec 1;126(Pt 23):5377-90
Journal of cell science 2013 Dec 1;126(Pt 23):5377-90
EGFR/Src/Akt signaling modulates Sox2 expression and self-renewal of stem-like side-population cells in non-small cell lung cancer.
Singh S, Trevino J, Bora-Singhal N, Coppola D, Haura E, Altiok S, Chellappan SP
Molecular cancer 2012 Sep 25;11:73
Molecular cancer 2012 Sep 25;11:73
Association of amphiregulin with the cetuximab sensitivity of gastric cancer cell lines.
Kneissl J, Keller S, Lorber T, Heindl S, Keller G, Drexler I, Hapfelmeier A, Höfler H, Luber B
International journal of oncology 2012 Aug;41(2):733-44
International journal of oncology 2012 Aug;41(2):733-44
MAPK and PI3K signalling differentially regulate angiogenic and lymphangiogenic cytokine secretion in squamous cell carcinoma of the head and neck.
Luangdilok S, Box C, Harrington K, Rhŷs-Evans P, Eccles S
European journal of cancer (Oxford, England : 1990) 2011 Mar;47(4):520-9
European journal of cancer (Oxford, England : 1990) 2011 Mar;47(4):520-9
Determinants of response to epidermal growth factor receptor tyrosine kinase inhibition in squamous cell carcinoma of the head and neck.
Rogers SJ, Box C, Chambers P, Barbachano Y, Nutting CM, Rhŷs-Evans P, Workman P, Harrington KJ, Eccles SA
The Journal of pathology 2009 May;218(1):122-30
The Journal of pathology 2009 May;218(1):122-30
Wounding-induced synthesis of hyaluronic acid in organotypic epidermal cultures requires the release of heparin-binding egf and activation of the EGFR.
Monslow J, Sato N, Mack JA, Maytin EV
The Journal of investigative dermatology 2009 Aug;129(8):2046-58
The Journal of investigative dermatology 2009 Aug;129(8):2046-58
Quantitative measurement of epidermal growth factor receptor-mitogen-activated protein kinase signal transduction using a nine-plex, peptide-based immunoassay.
Rauh-Adelmann C, Moskow JM, Graham JR, Yen LG, Boucher JI, Murphy CE, Nadler TK, Gordon NF, Radding JA
Analytical biochemistry 2008 Apr 15;375(2):255-64
Analytical biochemistry 2008 Apr 15;375(2):255-64
Hydrocortisone and indomethacin negatively modulate EGF-R signaling in human fetal intestine.
Kajanne R, Leppä S, Luukkainen P, Ustinov J, Thiel A, Ristimäki A, Miettinen PJ
Pediatric research 2007 Nov;62(5):570-5
Pediatric research 2007 Nov;62(5):570-5
Tumor growth inhibition with cetuximab and chemotherapy in non-small cell lung cancer xenografts expressing wild-type and mutated epidermal growth factor receptor.
Steiner P, Joynes C, Bassi R, Wang S, Tonra JR, Hadari YR, Hicklin DJ
Clinical cancer research : an official journal of the American Association for Cancer Research 2007 Mar 1;13(5):1540-51
Clinical cancer research : an official journal of the American Association for Cancer Research 2007 Mar 1;13(5):1540-51
G protein coupling and second messenger generation are indispensable for metalloprotease-dependent, heparin-binding epidermal growth factor shedding through angiotensin II type-1 receptor.
Mifune M, Ohtsu H, Suzuki H, Nakashima H, Brailoiu E, Dun NJ, Frank GD, Inagami T, Higashiyama S, Thomas WG, Eckhart AD, Dempsey PJ, Eguchi S
The Journal of biological chemistry 2005 Jul 15;280(28):26592-9
The Journal of biological chemistry 2005 Jul 15;280(28):26592-9
Aplidin induces apoptosis in human cancer cells via glutathione depletion and sustained activation of the epidermal growth factor receptor, Src, JNK, and p38 MAPK.
Cuadrado A, Garcia-Fernandez LF, Gonzalez L, Suarez Y, Losada A, Alcaide V, Martinez T, Fernandez-Sousa JM, Sanchez-Puelles JM, Munoz A
The Journal of biological chemistry 2003 Jan 3;278(1):241-50
The Journal of biological chemistry 2003 Jan 3;278(1):241-50
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- WB was performed on membrane enriched extracts (30 µg lysate) of A-431 (1), A-431 treated with EGF (200 ng/mL for 10 minutes) (2), A-431 treated with Gefitinib followed by EGF (1uM for 16 hours, 200 ng/mL for 10 minutes) (3), A-431 treated with Afatinib followed by EGF (0.5 uM for 6 hours, 200 ng/mL for 10 minutes) (4), A549 (5), A549 treated with EGF (200 ng/mL for 10 minutes) (6) and A549 treated with Afatinib followed by EGF (0.5 uM for 6 hours, 200 ng/mL for 10 minutes) (7). The blot was probed with Phospho-EGFR (Tyr1068) Rabbit Polyclonal Antibody (Product # 44-788G, 1:1000 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG Superclonal Secondary Antibody, HRP (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 170 kDa band corresponding to Phospho-EGFR (Tyr1068) was detected and observed to increase upon EGF treatment across cell lines tested. Pre-treatment with EGFR-antagonists, Gefitinib and Afatinib, resulted in inhibition of Phospho-EGFR in A-431 and A549 cell lines. Known quantity of proteinWAS electrophoresed using NovexNuPAGE4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock System (Product # EI0002) and Novex Sharp Pre-Stained Protein Standard (Product # LC5800). Proteins were then wet transferred onto a nitrocellulose membrane overnight. Primary and secondary Antibody was used following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Phospho-EGFR (Tyr1068) was performed using 70% confluent log phase A-431 cells treated with 200 ng/mL of EGF for 10 minutes. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Phospho-EGFR (Tyr1068) Rabbit Polyclonal Antibody (Product # 44-788G) at 1:100 dilution in 0.1% BSA and incubated overnight at 4 degree and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing membrane localization. Panel e represents cells treated with antagonist, Afatinib (1µM for 6hrs) followed by EGF (200 ng/mL for 10 minutes), showing no Phospho-EGFR staining. Panel f shows untreated cells with no signal. Panel g represents control cells with no primary antibody to assess background. The images were captured at 60X magnification
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
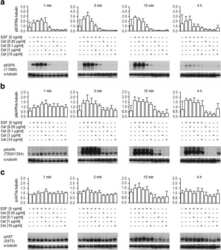
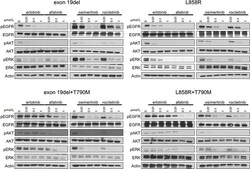
- Figure 2 Inhibition of the phosphorylation of EGFR and downstream proteins by EGFR-TKIs in BaF3 cells harboring EGFR mutations The results of immunoblotting for Ba/F3 cells with EGFR exon 19 deletion, L858R, exon 19 deletion+T790M, and L858R+T790M are shown. The cells were treated with the indicated concentrations of EGFR-TKIs for 4 h. Erlotinib, afatinib, osimertinib, and rociletinib were used as EGFR-TKIs. pEGFR, pAKT, and pERK indicate the phosphorylated form of EGFR, AKT, and ERK, respectively. Actin was used as a loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Inhibition of EGFR suppresses SP frequency and self-renewal. ( A ) H1650 and A549 cells revealed a significant decrease in SP frequency after treatment with 10 mug/ml EGFR-neutralizing antibodies for 5 days. ( B ) Frequency of SP cells in control antibody (1) and EGFR-neutralizing antibodies (2) treated cells. ( C ) Western blot analysis for ABCG2 and phospho-EGFR after EGFR-neutralizing antibody treatment. ( D ) SP frequency in NSCLC cells transfected with siRNA against EGFR or a control siRNA. ( E ) Expression of ABCG2 and EGFR in antibody treated cells. ( F and G ) SP cells were sorted and plated for self-renewal assay in the presence or absence of indicated drugs. Average number of spheres generated per well from 1000 cells is plotted (mean +- SD). Phase contrast microscopy images of the spheres in presence or absence of drugs are presented. * represent the p value of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Inhibition of EGFR signaling downregulates Sox2 expression (A) H1650-SPAdh cells were treated with EGFR or Src inhibitors for 4 days and western blot analysis was performed for the indicated proteins. ( B ) H1650-SPAdh cells were treated with MEK or PI3K inhibitor for 4 days and levels of Sox2 and activation of Akt or ERK was evaluated by western blot. ( C ) A549 and H1975 cells were treated with MEK or PI3K inhibitor for 5 days and the frequency of SP cells were examined by SP analysis. Data represents the fold change in mean (+- SD) of SP frequency. ( D ) ABCG2 expression upon inhibitor treatment as detected by western blotting. ( E ) SP cell frequency in NSCLC cell lines transfected with specific siRNAs against Src or AKT. ( F ) ABCG2 expression in siRNA transfected cells as detected by western blotting. ( G and H ) SP cells were sorted and plated for self-renewal assay in the presence or absence of indicated drugs. Average number of spheres generated from 1000 cells is plotted (mean +- SD). Phase contrast microscopy images of the spheres in presence or absence of drugs are presented. * represent the p value of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 1 Characterization of HCC4006ER cells, which are highly resistant to erlotinib or the irreversible EGFR-TKI CL387,785. A, HCC4006 and HCC4006ER cells were treated for 72 hours with increasing concentrations of erlotinib or CL387,785. Data generated by cell viability assay (CellTiter-Glo) are expressed as a percentage of the value for untreated cells. The error bars represent SEM of 3 independent experiments. B, HCC4006 and HCC4006ER cells were incubated for 24 hours at ~80-90% of cell confluence. Whole cell lysate of each cell line was collected and subjected to Proteome Profiler Human Phospho-RTK Array Kit to examine the phosphorylation levels of multiple RTKs. Detected phospho-RTKs on the array are circled. The spots in the four corners of the RTK array are positive controls. C, HCC4006 and HCC4006ER cells were incubated for 6 hours +- erlotinib (1 muM). Cell lysates were subjected to protein expression analysis with antibodies to pEGFR, EGFR, pMET, MET, pHer3, Her3, PTEN, pAkt, Akt, pErk, Erk, and beta-actin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 2 Neither Her3 loss nor persistent activation of pAkt and pErk is essential for the resistance in HCC4006ER cells. A, HCC4006ER cells with stable GFP or Her3 overexpression (HCC4006ER-GFP and HCC4006ER-Her3 cells) as well as HCC4006 and the original HCC4006ER cells were treated for 72 hours with increasing concentrations of erlotinib. Data generated by cell viability assay (CellTiter-Glo) are expressed as a percentage of the value for untreated cells. The error bars represent SEM of 3 independent experiments. B, HCC4006, HCC4006ER, HCC4006ER-GFP, and HCC4006ER-Her3 cells were incubated for 6 hours +- erlotinib (1 muM). Cell lysates were subjected to protein expression analysis with antibodies to Her3, pEGFR, EGFR, PTEN, pAkt, Akt, pErk, Erk, and beta-actin. C, HCC4006ER cells were treated for 72 h with increasing concentrations of erlotinib alone, BEZ235 alone, AZD6244 alone, or BEZ235 and AZD6244 in combination. HCC4006 cells were treated for 72 hours with increasing concentrations of erlotinib for 72 hours to plot a reference curve. Data generated by cell viability assay (CellTiter-Glo) are expressed as a percentage of the value for untreated cells. The error bars represent SEM of 3 independent experiments. Combination index (CI) at IC50 dose of BEZ235 combined with AZD6244 was calculated by CompuSyn software. CI>1, CI = 1, and CI
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 6 Effects of knockdown of ZEB1 or overexpression of miR-200c on EMT markers and erlotinib sensitivity in HCC4006ER cells. A, HCC4006ER cells were plated at 3x10 5 per well in 6-well plate and transfected with ZEB1 or control siRNA at 5 nM as a final concentration. Cells were harvested for analysis at 96 hours post-transfection. Cell lysates were subjected to protein expression analysis with antibodies to ZEB1, E-cadherin, vimentin, and beta-actin. B, HCC4006ER cells were plated at 3x10 3 cells/ well in black wall 96-well plate, transfected with ZEB1 or control siRNA at 5 nM as a final concentration, and treated for 72 hours at 48 hours after siRNA transfection with increasing concentrations of erlotinib. HCC4006 cells were treated for 72 hours with increasing concentrations of erlotinib for 72 hours to plot a reference curve. Data generated by cell viability assay (CellTiter-Glo) are expressed as a percentage of the value for erlotinib-untreated cells with control siRNA. Control siRNA or transfection reagent did not affect cell viability in HCC4006ER cells at this concentration. The error bars represent SEM of 3 independent experiments. C, HCC4006 and HCC4006ER cells were plated at 3x10 5 per well in 6-well plate. HCC4006ER cells were transfected with ZEB1 or control siRNA at 0.5 or 5 nM as indicated. HCC4006 cells and siRNA-transfected HCC4006ER cells were incubated for 6 hours +- erlotinib (1 muM) at 48 hours after siRNA transfection. Cell lysates were subjected to prot
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Effect of treatment with trastuzumab for 8 days on the expression profile of HER and pHER receptors. All gastric cancer cell lines were treated for 8 days with 10 ug/ml trastuzumab; afterwards, the expression of EGFR, HER2, HER3, HER4, pEGFR, pHER2, pHER3 and pHER4 was determined via Western blot analysis. Basal expression of HER receptors varied highly between the cell lines. No obvious correlation between basal HER/pHER receptor expression and/or changes in expression and responsiveness to trastuzumab could be observed
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Effect of treatment with cetuximab for 8 days on the expression profile of HER and pHER receptors. All gastric cancer cell lines were treated for 8 days with 10 ug/ml cetuximab; afterwards, the expression of EGFR, HER2, HER3, HER4, pEGFR, pHER2, pHER3 and pHER4 was determined via Western blot analysis. Only minor effects of the treatment on HER/pHER receptor profile were detected. No obvious correlation between basal HER/pHER receptor expression and/or changes in expression and responsiveness to cetuximab could be observed
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
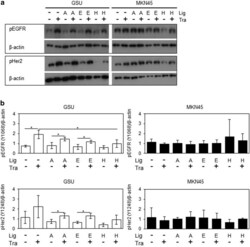
- Fig. 8 Effect of trastuzumab and concomitant ligand application on pEGFR and pHER2 levels in GSU and MKN45 cells. GSU and MKN45 cells were treated for 6 h with 10 ug/ml trastuzumab (Tra) and 15 ng/ml AREG (A) or 0.1 ng/ml EGF (E) or 0.4 ng/ml HB-EGF (H) or 0.75 ng/ml TGFalpha (T). Subsequently, pEGFR and pHER levels were determined by Western blot analysis. a Shows representative experiments; b shows results of densitometric measurements of three independent experiments. Treatment with trastuzumab induced the levels of both proteins in GSU cells but not in MKN45 cells. Concomitant application of the ligand suppressed this effect in GSU cells (significant suppression for pEGFR and HB-EGF). p values at significance levels of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 The sensitivities of lung cancer cell lines to EGFR-TKIs (A) MTS assays of PC-9, PC9-ER, and H1975 cells exposed to the indicated EGFR-TKIs. Data points represent the mean +- standard deviation. (B) PC-9, PC9-ER, and H1975 cells were treated with the indicated concentrations of EGFR-TKIs for 4 h prior to immunoblotting for the phosphorylated (p) and non-phosphorylated forms of EGFR, AKT, and ERK. Actin was used as a loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 E-cadherin regulates EGFR tissue polarization and activity. a Newborn epidermal whole-mount fluorescence analysis for EGFR and ZO-1 showing enrichment of EGFR at TJs in control SG2 layer. b Quantification of junctional EGFR fluorescence intensity. n = 6 biological replicates with 10 junctions each. Graph shows mean values +- SEM and single means of biological replicates (dots/squares). Granular layer (SG) intensity was normalized to spinous layer (SS) intensity. * P = 0.0214 with D'Agostino & Pearson omnibus normality test followed by one sample t -test. c Newborn epidermal whole-mount fluorescence analysis for EGFR and ZO-1 showing SG2, SS, and cross section (v. section), showing increased junctional ZO-1 and EGFR (arrowheads) in the SS of E-cadherin (Ecad) -/- epidermis compared to control (Ctr). a , c Partial projections and v. sections of confocal stacks from newborn epidermal whole mounts. For better visualization fluorescence intensities of SS projections have been increased relative to SG. Nuclei were stained with DAPI (blue). d Quantification of cortical EGFR enrichment in SG normalized to Ctr SS. Shown are respective means (lines) of three biological replicates with 20 measurements (dots) per replicate, * P < 0.05; n = 3 biological replicates with Kruskal-Wallis, Dunn's post hoc test. e Western blot for total and phosphorylated EGFR (pEGFR) from Ctr and Ecad epi-/- newborn epidermis. f Western blot quantification of pEGFR levels in newborn Ctr and Ecad epi-/-
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 3 Fig. EWI-2 removal promotes tumor cell motility by regulating the enrichment of exosomal miR-3934-5p. (A) Exosomes from PC3 NEG and KO cells were purified and miRNA of exosomes was extracted. Real-time PCR for miRNA was carried out and the miR-29a, miR-149-3p and miR-3934-5p expression levels in exosomes were detected. The relative miRNA expression level was quantified with Student's t -test and presented as mean +- SD ( n = 3 individual experiments). * P < 0.05; ** P < 0.01. (B) PC3 NEG and KO cells were treated with miR-29a, miR-149-3p and miR-3934-5p as well as miRctrl mimics and analyzed by western blot assay. The miR-3934-5p mimic significantly inhibited the EGFR signaling in PC3 cells with downregulated p1068-EGFR and the phosphorylated p44/42-MAPK (ERK1/2). (C) The normalized band densities of (B) were quantified with Student's t -test and are presented as mean +- SD ( n = 3 individual experiments). * P < 0.05; ** P < 0.01; *** P < 0.001. (D) The predicated interaction between miR-3934-5p and the EGFR 3'-untranslated region (3'-UTR) in TargetScan database. (E) PC3 NEG and KO cells were treated with EGFR inhibitor gefitinib and miR-3934-5p mimic, as well as DMSO as negative control and then examined for the migration through Transwell inserts, which were coated with either fibronectin (10 mug*mL -1 ) or laminin 111 (10 mug*mL -1 ). The cells that migrated through the insert pores and adhered to the bottom of the inserts were photographed. Scale bars: 200 um. (F) Numbe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 The EGFR-Lpd interplay is cell-line-dependent. ( A ) Representative immunoblot of EGFR, Lpd, phosphorylated Lpd (Y426) and beta-actin in A172 and U343MG upon EGFR knockdown and 1 h after irradiation. ( B ) Densitometric analyses showing changes in Lpd expression and phosphorylation from ( A ). Fold changes of Lpd levels are calculated by normalization to beta-actin. Fold changes of phosphorylated Lpd Y426 are determined by normalization to total Lpd expression. All protein levels were related to the corresponding beta-actin. Fold changes are relative to control-treated and unirradiated samples. ( C ) Representative Western blot from Lpd-silenced A172 and U343MG showing total and phosphorylated EGFR 1 h post-X-ray irradiation. beta-actin served as loading control. Representative Western blots are shown. ( D ) Densitometry of EGFR from ( C ). Fold changes of EGFR levels are calculated by normalization to beta-actin. Fold changes of phosphorylated EGFR are calculated by normalization to total EGFR expression. All protein levels were related to the corresponding beta-actin. Fold changes are relative to control-treated and unirradiated samples. All results are mean +- SD ( n = 3, two-sided Student's t -test, * p < 0.05, ** p < 0.01, *** p < 0.001). si, small interfering RNA; C, control; Lpd, Lamellipodin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Sensitivity of lung cancer cell lines to EGFR-TKIs A. MTS assay for PC-9, H3255, PC9-ER, and H1975 cells. The IC50 values (nM) for EGFR-TKIs are shown. Error bars indicate standard deviation. B. The results of immunoblotting for PC-9, H3255, PC9-ER, and H1975 cells are shown. The cells were treated with the indicated concentrations of EGFR-TKIs for 4 h. Erlotinib, afatinib, osimertinib, and rociletinib were used as EGFR-TKIs. pEGFR, pAKT, and pERK indicate the phosphorylated form of EGFR, AKT, and ERK, respectively. Actin was used as a loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Efficacy of EGFR-TKIs for EGFR exon 20 insertion mutations A. MTS assay (left) and immunoblotting (right) for BID007 (EGFR A763_Y764insFQEA) cells. Error bars indicate standard deviation. B. Results of immunoblotting for Ba/F3 cells with EGFR exon 20 insertion mutations. The cells were treated with the indicated concentrations of EGFR-TKIs for 4 h. Erlotinib, afatinib, osimertinib, and rociletinib were used as EGFR-TKIs. pEGFR, pAKT, and pERK indicate the phosphorylated form of EGFR, AKT, and ERK, respectively. Actin was used as a loading control. C. Apoptosis assay using cytometry. Ba/F3 cells harboring wild type EGFR and EGFR D770_N771insNPG (NPG) were treated with EGFR-TKIs for 48 h, subsequently the cells were stained with propidium iodide and annexin V-APC. The numbers indicate the proportion of annexin V-positive and/or propidium iodide-positive cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Kinetic and concentration-dependent effects of trastuzumab and afatinib on the activation of EGFR and HER 2 in NCI -N87 cells. The levels of activated receptors were determined after treatment of NCI -N87 cells with trastuzumab and/or afatinib by western blot analysis. Activation of the receptors was analyzed using pEGFR -specific (Y1068) and pHER 2-specific (Y1221/2) antibodies. (A) The levels of activated receptors were determined after 5 min of treatment of NCI -N87 cells with afatinib (0.01, 0.1, 0.5, 1 mu m ) and/or trastuzumab (5 mug*mL -1 ). (B) Cells were treated for 5, 20, 60, and 120 min with trastuzumab (5 mug*mL -1 ) and/or afatinib (0.5 mu m ). Equal loading of the lanes was confirmed by the detection of alpha-tubulin or beta-actin. The depicted results are representative of three independent experiments. The results of the densitometric analysis and P -values are shown in the Supplement (Fig. S2 , Fig. S3 , Table S1 , and Table S2 ). A, afatinib; T, trastuzumab; Ctrl, control.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot