Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Immunohistochemistry [1]
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 39-6500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FAK Monoclonal Antibody (ZF002)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- Mouse and rat FAK-1 proteins share 93% homology with human. On western blots, this antibody identifies the target band at ~110 kDa. Reactivity has been confirmed with HeLa cell lysates and mouse and rat brain homogenates by western blotting.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- ZF002
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Mechanosensitive expression of lamellipodin promotes intracellular stiffness, cyclin expression and cell proliferation.
HIFα independent mechanisms in renal carcinoma cells modulate divergent outcomes in fibronectin assembly mediated by hypoxia and CoCl(2).
Phosphatidylinositol 4-kinase III beta regulates cell shape, migration, and focal adhesion number.
The histone deacetylase complex MiDAC regulates a neurodevelopmental gene expression program to control neurite outgrowth.
mDia1 and Cdc42 Regulate Activin B-Induced Migration of Bone Marrow-Derived Mesenchymal Stromal Cells.
Rhodoptilometrin, a Crinoid-Derived Anthraquinone, Induces Cell Regeneration by Promoting Wound Healing and Oxidative Phosphorylation in Human Gingival Fibroblast Cells.
Cell circuits between B cell progenitors and IL-7(+) mesenchymal progenitor cells control B cell development.
Knockdown of glucose-regulated protein 78 decreases the invasion, metalloproteinase expression and ECM degradation in hepatocellular carcinoma cells.
Brazzo JA, Biber JC, Nimmer E, Heo Y, Ying L, Zhao R, Lee K, Krause M, Bae Y
Journal of cell science 2021 Jun 15;134(12)
Journal of cell science 2021 Jun 15;134(12)
HIFα independent mechanisms in renal carcinoma cells modulate divergent outcomes in fibronectin assembly mediated by hypoxia and CoCl(2).
Magdaleno C, Dixon L, Rajasekaran N, Varadaraj A
Scientific reports 2020 Oct 29;10(1):18560
Scientific reports 2020 Oct 29;10(1):18560
Phosphatidylinositol 4-kinase III beta regulates cell shape, migration, and focal adhesion number.
Bilodeau P, Jacobsen D, Law-Vinh D, Lee JM
Molecular biology of the cell 2020 Aug 1;31(17):1904-1916
Molecular biology of the cell 2020 Aug 1;31(17):1904-1916
The histone deacetylase complex MiDAC regulates a neurodevelopmental gene expression program to control neurite outgrowth.
Mondal B, Jin H, Kallappagoudar S, Sedkov Y, Martinez T, Sentmanat MF, Poet GJ, Li C, Fan Y, Pruett-Miller SM, Herz HM
eLife 2020 Apr 16;9
eLife 2020 Apr 16;9
mDia1 and Cdc42 Regulate Activin B-Induced Migration of Bone Marrow-Derived Mesenchymal Stromal Cells.
Wang X, Tang P, Guo F, Zhang M, Yan Y, Huang M, Chen Y, Zhang L, Zhang L
Stem cells (Dayton, Ohio) 2019 Jan;37(1):150-162
Stem cells (Dayton, Ohio) 2019 Jan;37(1):150-162
Rhodoptilometrin, a Crinoid-Derived Anthraquinone, Induces Cell Regeneration by Promoting Wound Healing and Oxidative Phosphorylation in Human Gingival Fibroblast Cells.
Tseng CC, Lai YC, Kuo TJ, Su JH, Sung PJ, Feng CW, Lin YY, Chen PC, Tai MH, Cheng SY, Kuo HM, Wen ZH
Marine drugs 2019 Feb 27;17(3)
Marine drugs 2019 Feb 27;17(3)
Cell circuits between B cell progenitors and IL-7(+) mesenchymal progenitor cells control B cell development.
Fistonich C, Zehentmeier S, Bednarski JJ, Miao R, Schjerven H, Sleckman BP, Pereira JP
The Journal of experimental medicine 2018 Oct 1;215(10):2586-2599
The Journal of experimental medicine 2018 Oct 1;215(10):2586-2599
Knockdown of glucose-regulated protein 78 decreases the invasion, metalloproteinase expression and ECM degradation in hepatocellular carcinoma cells.
Li H, Song H, Luo J, Liang J, Zhao S, Su R
Journal of experimental & clinical cancer research : CR 2012 Apr 30;31(1):39
Journal of experimental & clinical cancer research : CR 2012 Apr 30;31(1):39
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
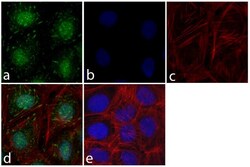
- Experimental details
- Immunofluorescent analysis of FAK was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes; permeabilized with 0.25% Triton™ X-100 for 10 minutes followed by blocking with 5% BSA for 1 hour at room temperature. The cells were incubated with FAK Mouse Monoclonal Antibody (Product # 39-6500) at 1 µg\mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytosolic and punctate staining on the membrane. Panel e shows no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of FAK showing staining in the cytoplasm of paraffin-embedded mouse spleen tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10 mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with FAK monoclonal antibody (Product # 39-6500) diluted in 3% BSA-PBS at a dilution of 1:200 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using a HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of FAK1 was done on A549 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 1 hour at room temperature. Cells were labeled with FAK1 Mouse Monoclonal Antibody (396500, red histogram) or with mouse isotype control (pink histogram) at 1-3 µg/million cells in 2.5% BSA. After incubation at room temperature for 2-3 hours, the cells were labeled with Alexa Fluor¨ 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 GRP78 knockdown decreased JNK and ERK signaling pathway. (A) Western blot analysis of JNK and p-JNK levels in cells that stably expressing shGRP78-3. (B) Western blot analysis of the ERK and p-ERK levels in cells that stably expressing shGRP78-3. (C) Western blot analysis of FAK, pY397-FAK, Src and pY416-Src in the cells that stably expressing shGRP78-3. All the experiments were repeated for three times, the results of quantative analysis were represented as x - +- SE and analyzed by one-way ANOVA. (Columns,mean of three separate experiments; bars, SE; *, values significantly different at the 5% levels).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. MiDAC regulates neurite outgrowth via the SLIT3/ROBO3 and NTN1/UNC5B signaling pathways. ( A ) WB for signaling components of the SLIT3/ROBO3 and NTN1/DCC/UNC5B signaling axes from CM and total cell lysates of WT and Dnttip1 KO1 NE after 12 days of differentiation. To enrich SLIT3 and NTN1 from CM, IPs were performed with SLIT3 and NTN1 antibodies from CM of WT and Dnttip1 KO1 NE. Actin is the loading control for the total cell lysates. ( B ) Assay to rescue the neurite outgrowth defects in Dnttip1 KO1 NE. CM of Dnttip1 KO1 NE was supplemented with the recombinant signaling ligands SLIT3 and/or NTN1 from day 7-12 without or with preblocking of Dnttip1 KO1 NE with IgG or signaling receptor antibodies against ROBO3 and/or UNC5B. MAP2 IF staining was performed after 12 days of differentiation. Nuclei were stained with DAPI. To facilitate analysis the neuronal cell body (blue) and its neurites were manually traced with ImageJ software and for each sample one traced neuron is displayed in the inlet. The white scale bar represents 50 um. ( C, D ) Quantification of ( C ) neurite length and ( D ) the total number of neurites per neuron from the MAP2 IF staining in ( B ) using ImageJ. ( C ) Neurite length was divided into two categories of short neurites =50 um (white box plots). ( C, D ) The neurites of 200 neurons were assessed per sample. One-way ANOVA was performed throughout where ***, p0.05 is not signifi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Integrins alpha4 and alpha5 are required for FN fibril assembly in response to CoCl 2 . ( a ) Caki-1 cells cultured at 21% O 2 , exposed to 1% O 2 or treated with 50 uM CoCl 2 for the indicated times. Cells were lysed and immunoblotted for pFAK and total FAK. beta-Actin was used as the loading control. ( b ) Cell lysates as in ( a ) were immunoblotted for pMLC. beta-Actin was used as the loading control. ( c ) Caki-1 cells were pretreated with 10 uM ATN-161 (Integrin alpha5 inhibitor), 1 ug/ml BIO5192 (Integrin alpha4 inhibitor) or a combination of both for 30 min followed by treatment with 50 uM CoCl 2 for 2 h and immunostained for FN (red) and counterstained with the nuclear stain DAPI (blue). Scale bar = 10 um. ( d ) Caki-1 cells transduced with shScr (200 MOI) or shalpha4 (2000 MOI), shalpha5 (2000 MOI) or a combination of shalpha4 (1000 MOI) and shalpha5 (1000 MOI) expressing the GFP reporter were left untreated or treated with 50 uM CoCl 2 for 2 h and immunostained for FN (red) and DAPI (blue). Transduced cells are shown in green (GFP reporter). The bar graph below the figure quantifies the % 'fibril-containing' cells in GFP reporter-expressing virus transduced cells. Data is representative of two independent experiments. More than 80 cells were counted in each condition.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry