Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-652G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-FAK (Tyr576) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Chicken/Avian, Xenopus
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Tumor Angiogenesis Is Differentially Regulated by Phosphorylation of Endothelial Cell Focal Adhesion Kinase Tyrosines-397 and -861.
The PDGFRα-laminin B1-keratin 19 cascade drives tumor progression at the invasive front of human hepatocellular carcinoma.
A mutation in Nischarin causes otitis media via LIMK1 and NF-κB pathways.
A Short Region of Connexin43 Reduces Human Glioma Stem Cell Migration, Invasion, and Survival through Src, PTEN, and FAK.
Tumor endothelial cells promote metastasis and cancer stem cell-like phenotype through elevated Epiregulin in esophageal cancer.
Vinculin b deficiency causes epicardial hyperplasia and coronary vessel disorganization in zebrafish.
Modulation of FAK and Src adhesion signaling occurs independently of adhesion complex composition.
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
Pelargonidin attenuates PDGF-BB-induced aortic smooth muscle cell proliferation and migration by direct inhibition of focal adhesion kinase.
Activation of endogenous FAK via expression of its amino terminal domain in Xenopus embryos.
LCB 03-0110, a novel pan-discoidin domain receptor/c-Src family tyrosine kinase inhibitor, suppresses scar formation by inhibiting fibroblast and macrophage activation.
Enhanced integrin mediated signaling and cell cycle progression on fibronectin mimetic peptide amphiphile monolayers.
Downregulation of Rap1GAP in human tumor cells alters cell/matrix and cell/cell adhesion.
Arrestin-3 is essential for the activation of Fyn by the luteinizing hormone receptor (LHR) in MA-10 cells.
Alphavbeta3 integrin and cofilin modulate K1735 melanoma cell invasion.
Analyzing FAK and Pyk2 in early integrin signaling events.
SRC catalytic but not scaffolding function is needed for integrin-regulated tyrosine phosphorylation, cell migration, and cell spreading.
SRC catalytic but not scaffolding function is needed for integrin-regulated tyrosine phosphorylation, cell migration, and cell spreading.
FAK integrates growth-factor and integrin signals to promote cell migration.
Pedrosa AR, Bodrug N, Gomez-Escudero J, Carter EP, Reynolds LE, Georgiou PN, Fernandez I, Lees DM, Kostourou V, Alexopoulou AN, Batista S, Tavora B, Serrels B, Parsons M, Iskratsch T, Hodivala-Dilke KM
Cancer research 2019 Sep 1;79(17):4371-4386
Cancer research 2019 Sep 1;79(17):4371-4386
The PDGFRα-laminin B1-keratin 19 cascade drives tumor progression at the invasive front of human hepatocellular carcinoma.
Govaere O, Petz M, Wouters J, Vandewynckel YP, Scott EJ, Topal B, Nevens F, Verslype C, Anstee QM, Van Vlierberghe H, Mikulits W, Roskams T
Oncogene 2017 Nov 23;36(47):6605-6616
Oncogene 2017 Nov 23;36(47):6605-6616
A mutation in Nischarin causes otitis media via LIMK1 and NF-κB pathways.
Crompton M, Purnell T, Tyrer HE, Parker A, Ball G, Hardisty-Hughes RE, Gale R, Williams D, Dean CH, Simon MM, Mallon AM, Wells S, Bhutta MF, Burton MJ, Tateossian H, Brown SDM
PLoS genetics 2017 Aug;13(8):e1006969
PLoS genetics 2017 Aug;13(8):e1006969
A Short Region of Connexin43 Reduces Human Glioma Stem Cell Migration, Invasion, and Survival through Src, PTEN, and FAK.
Jaraíz-Rodríguez M, Tabernero MD, González-Tablas M, Otero A, Orfao A, Medina JM, Tabernero A
Stem cell reports 2017 Aug 8;9(2):451-463
Stem cell reports 2017 Aug 8;9(2):451-463
Tumor endothelial cells promote metastasis and cancer stem cell-like phenotype through elevated Epiregulin in esophageal cancer.
Sun L, Pan J, Yu L, Liu H, Shu X, Sun L, Lou J, Yang Z, Ran Y
American journal of cancer research 2016;6(10):2277-2288
American journal of cancer research 2016;6(10):2277-2288
Vinculin b deficiency causes epicardial hyperplasia and coronary vessel disorganization in zebrafish.
Cheng F, Miao L, Wu Q, Gong X, Xiong J, Zhang J
Development (Cambridge, England) 2016 Oct 1;143(19):3522-3531
Development (Cambridge, England) 2016 Oct 1;143(19):3522-3531
Modulation of FAK and Src adhesion signaling occurs independently of adhesion complex composition.
Horton ER, Humphries JD, Stutchbury B, Jacquemet G, Ballestrem C, Barry ST, Humphries MJ
The Journal of cell biology 2016 Feb 1;212(3):349-64
The Journal of cell biology 2016 Feb 1;212(3):349-64
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
Robertson J, Jacquemet G, Byron A, Jones MC, Warwood S, Selley JN, Knight D, Humphries JD, Humphries MJ
Nature communications 2015 Feb 13;6:6265
Nature communications 2015 Feb 13;6:6265
Pelargonidin attenuates PDGF-BB-induced aortic smooth muscle cell proliferation and migration by direct inhibition of focal adhesion kinase.
Son JE, Jeong H, Kim H, Kim YA, Lee E, Lee HJ, Lee KW
Biochemical pharmacology 2014 May 15;89(2):236-45
Biochemical pharmacology 2014 May 15;89(2):236-45
Activation of endogenous FAK via expression of its amino terminal domain in Xenopus embryos.
Petridou NI, Stylianou P, Christodoulou N, Rhoads D, Guan JL, Skourides PA
PloS one 2012;7(8):e42577
PloS one 2012;7(8):e42577
LCB 03-0110, a novel pan-discoidin domain receptor/c-Src family tyrosine kinase inhibitor, suppresses scar formation by inhibiting fibroblast and macrophage activation.
Sun X, Phan TN, Jung SH, Kim SY, Cho JU, Lee H, Woo SH, Park TK, Yang BS
The Journal of pharmacology and experimental therapeutics 2012 Mar;340(3):510-9
The Journal of pharmacology and experimental therapeutics 2012 Mar;340(3):510-9
Enhanced integrin mediated signaling and cell cycle progression on fibronectin mimetic peptide amphiphile monolayers.
Shroff K, Pearce TR, Kokkoli E
Langmuir : the ACS journal of surfaces and colloids 2012 Jan 24;28(3):1858-65
Langmuir : the ACS journal of surfaces and colloids 2012 Jan 24;28(3):1858-65
Downregulation of Rap1GAP in human tumor cells alters cell/matrix and cell/cell adhesion.
Tsygankova OM, Ma C, Tang W, Korch C, Feldman MD, Lv Y, Brose MS, Meinkoth JL
Molecular and cellular biology 2010 Jul;30(13):3262-74
Molecular and cellular biology 2010 Jul;30(13):3262-74
Arrestin-3 is essential for the activation of Fyn by the luteinizing hormone receptor (LHR) in MA-10 cells.
Galet C, Ascoli M
Cellular signalling 2008 Oct;20(10):1822-9
Cellular signalling 2008 Oct;20(10):1822-9
Alphavbeta3 integrin and cofilin modulate K1735 melanoma cell invasion.
Dang D, Bamburg JR, Ramos DM
Experimental cell research 2006 Feb 15;312(4):468-77
Experimental cell research 2006 Feb 15;312(4):468-77
Analyzing FAK and Pyk2 in early integrin signaling events.
Bernard-Trifilo JA, Lim ST, Hou S, Schlaepfer DD, Ilic D
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
Current protocols in cell biology 2006 Apr;Chapter 14:Unit 14.7
SRC catalytic but not scaffolding function is needed for integrin-regulated tyrosine phosphorylation, cell migration, and cell spreading.
Cary LA, Klinghoffer RA, Sachsenmaier C, Cooper JA
Molecular and cellular biology 2002 Apr;22(8):2427-40
Molecular and cellular biology 2002 Apr;22(8):2427-40
SRC catalytic but not scaffolding function is needed for integrin-regulated tyrosine phosphorylation, cell migration, and cell spreading.
Cary LA, Klinghoffer RA, Sachsenmaier C, Cooper JA
Molecular and cellular biology 2002 Apr;22(8):2427-40
Molecular and cellular biology 2002 Apr;22(8):2427-40
FAK integrates growth-factor and integrin signals to promote cell migration.
Sieg DJ, Hauck CR, Ilic D, Klingbeil CK, Schaefer E, Damsky CH, Schlaepfer DD
Nature cell biology 2000 May;2(5):249-56
Nature cell biology 2000 May;2(5):249-56
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
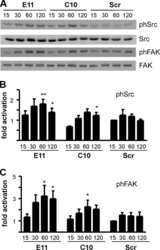
- Figure 2 FAK is heavily phosphorylated in mesodermal tissues and integrin-free regions of cells. (A) Intensity color coded confocal section of the dorsal lip region from a whole mount immunostained gastrula stage embryo using a P-Y576 FAK antibody. Mesodermal cells (white arrow) display much higher levels of phospho-FAK than endodermal cells lining the forming archenteron (white arrowheads) and the endodermal cells of the blastopore (red arrow). (B) Same as A but showing the anterior mesendoderm and the animal cap from a whole mount immunostained gastrula stage embryo. The superficial cells of the animal cap (white arrowhead) show lower levels of phospho-FAK signal compared to deep cells (white arrow) and mesendodermal cells (red arrow). (C) High magnification color coded narrow optical section of superficial cells of the animal cap reveals that the apical surface of these cells display similar levels of phospho-FAK compared to the basolateral region while (D) the apical region of the deep cells of the animal cap facing the fibronectin ECM display significantly elevated levels of phospho-FAK compared to the basolateral region. In addition, in the deep cells of the ectoderm labeling of phospho-FAK in the basolateral region is relatively uniform but the apical region displays distinct foci of higher signal intensity (E-G) Confocal optical sections from whole mount immunostained embryos using integrin-beta1 (green) and P-Y397 FAK antibodies (red). Integrin-beta1 and P-Y397 FAK c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
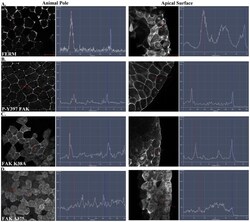
- Experimental details
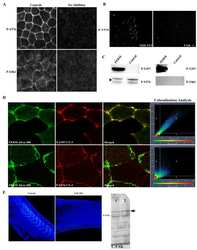
- Figure 4 The FERM domain is necessary and sufficient for membrane localization of FAK at integrin-free regions. Confocal images and intensity profiles of the indicated constructs after whole mount immunostaining. The first column are top views of superficial cells of the animal cap in intact embryos and the second column are views from sagittally sectioned embryos that reveal the localization of each construct on the apical surface of superficial cells. Apical region of superficial blastomeres is to the right. (A) The FERM domain shows strong plasma membrane localization in the top view and is strongly localized to the apical surface. (B) Endogenous phosphorylated FAK shows very strong plasma membrane localization in the top view and is localized on the basolateral and apical surface of the cell. (C) Full length FAK with the point mutation K38A exhibits strong membrane localization. (D) Deletion of the FERM domain (HA-Delta375 FAK construct) abolishes the plasma membrane localization of FAK. Scale bars: 25 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Inhibition of FAK activity by FAK [i] in human fibroblasts. (A) Immunoblotting of FAK phosphorylation sites FAK Y397 and FAK Y576 and total FAK in total cell lysates. HFF cells spread on FN for 1 h were treated with DMSO or the FAK inhibitor AZ13256675 (FAK [i]) for 1 h using half-log dilutions. Cells kept in suspension for 30 min (Susp) were used to detect basal FAK activity. Untreated cells spread on FN for 2 h were used to detect maximal FAK activity. FAK Y397 and FAK Y576 were used to assess FAK catalytic activity. Molecular mass values (kD) are displayed. (B and C) Quantification of immunoblotted membranes in A. FAK phosphorylation values FAK Y397 (B) and FAK Y576 (C) were normalized to total FAK (mean +- SEM, n = 3). (D) Dose-response curve using FAK Y397 as a readout for FAK activity to determine percentage inhibition relative to cells treated with DMSO. Gray lines and shading show values for suspension and untreated FN conditions (not used to calculate the trendline, mean +- SEM, n = 3). The 50% FAK Y397 inhibitory concentration (IC 50 ) was calculated as 0.11 uM FAK [i] using the formula y = 12.23ln( x ) + 76.99 ( y, percentage inhibition; x, FAK [i] concentration). (E) HFFs spread on FN for 1 h were treated with DMSO, 0.1 uM FAK [i], or 3 uM FAK [i] for 1 h. IACs were visualized by staining for FAK Y397 (green) and vinculin (red) by immunofluorescence. The actin cytoskeleton was visualized by staining with fluorophore-conjugated phalloidin. Bars: (main) 20
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
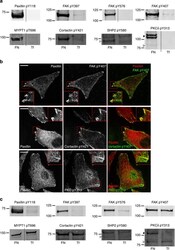
- Experimental details
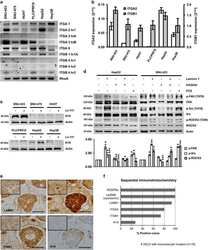
- Figure 4 Validation of adhesion complex-localized phosphorylation sites. ( a ) Immunoblot analyses of the protein complexes isolated from cells spread on FN or Tf for 120 min. Samples were probed using phosphospecific antibodies targeting: paxillin pTyr118; FAK pTyr397, pTyr576 and pTyr407; MYPT1 pThr696; cortactin pTyr421; SHP2 pTyr580; and PKCdelta pTyr313. ( b ) Immunofluorescence images of cells spread on FN for 120 min. Cells were stained using the phosphospecific antibodies FAK pTyr407, cortactin pTyr421 and PKCdelta pTyr313 (all green). In each case, cells were also stained using an antibody targeting the focal adhesion marker paxillin (red). Scale bar, 20 mum. ( c ) Immunoblot analyses of the WCLs of cells spread on FN or Tf for 120 min. Samples were probed using the same antibodies as used to probe isolated complexes in a . Dashed lines in a , c indicate where the images have been cropped for display purposes. White arrows to left of blots highlight the expected MWs of the corresponding proteins. Black arrows to the left of blots highlight a putative high MW isoform of PKCdelta.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
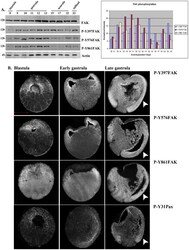
- Experimental details
- Figure 1 FAK expression and phosphorylation during development. (A) Western Blots from extracts of equal numbers of embryos probed with a monoclonal antibody against the C-terminus of FAK or polyclonal antibodies against the phosphorylated tyrosine residues indicated. FAK is phosphorylated on all three residues both before and after gastrulation. The intensity values from the densitometry analysis of the western blots were normalized against total FAK amount. (B) Blastula (1 st column), early gastrula (2 nd column) and late gastrula embryos (3 rd column) stained with P-Y397, P-Y576, P-Y861 and P-Y31paxillin antibodies as indicated. Phosphorylated FAK and paxillin can be detected on the plasma membrane from early blastula stages including the apical region of superficial blastomeres. During gastrulation elevated levels of phosphorylation are detected in the highly morphogenetic mesodermal tissues (white arrowheads). Scale bar: 400 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
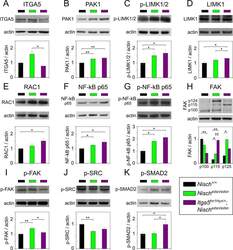
- Experimental details
- Fig 7 Protein expression analysis of NISCH interacting partners and downstream pathways in middle ear by western blot. Middle ear epithelial cells extracts of Nisch +/+ , Nisch edsn/edsn and Itga5 tm1Hyn/+ ; Nisch edsn/edsn mice at 8 wk, probed with (A) ITGA5, (B) PAK1, (C) p-LIMK1/2, (D) LIMK1, (E) RAC1, (F) NF-kappaB p65, (G) p-NFkappaB p65, (H) FAK, (I) p-FAK, (J) p-SRC and (K) p-SMAD2 antibodies. The results presented are from four independent experiments for all the antibodies except for ITGA5, PAK1, LIMK1, p-NFkappaB p65 and FAK, where three independent experiments were used to present the data. ns P > 0.05; * P < 0.05; ** P < 0.01; *** P < 0.001. Error bars indicate standard error of mean. A Student's t-test was performed to analyse the data.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
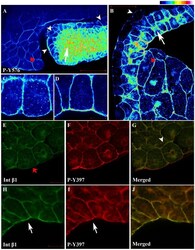
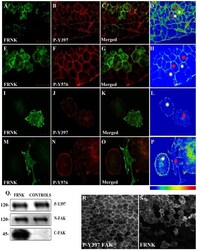
- Figure 3 FRNK does not act as a dominant negative in early Xenopus embryos. (A-D) Optical sections of whole mount immunostained embryos injected with 1 ng GFP-FRNK at the two dorsal blastomeres at the four-cell stage. Embryos were stained with anti-GFP (A) and anti-P-Y397 (B). C is the merged image and D an intensity color coded image of the anti-P-Y397 signal. FRNK injected cells are indicated with red stars and control cells with white stars. FRNK expression fails to reduce the phosphorylation levels of endogenous FAK on tyrosine 397. (E-H) Same as A-D, but the embryos were stained with anti-GFP (E) and anti-P-Y576 (F). FRNK expressing cells display similar levels of phosphorylation on tyrosine 576 as neighboring control cells. (I-L) Confocal images of A6 Xenopus cells transfected with GFP-FRNK. Cells were stained with anti-GFP (I) and anti-P-Y397 (J). K is the merged image and L an intensity color coded image of the anti-P-Y397 signal. FRNK expression leads to reduction of the phosphorylation levels of FAK on tyrosine 397 at the focal adhesions. (M-P) Same as I-L but the cells were stained with anti-GFP (M) and anti-P-Y576 (N) antibodies. FRNK expression leads to downregulation of the endogenous phosphorylation levels of FAK on tyrosine 576 at the focal adhesions. (Q) Western blot analysis of control and injected gastrula stage embryos with 1 ng FRNK at the animal pole of both blastomeres of two cell stage embryos. FRNK expression fails to reduce endogenous FAK phosphoryla
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
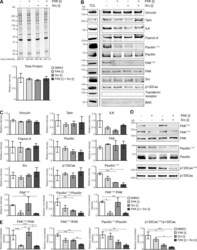
- Figure 5. Effects of FAK, Src, and combined FAK and Src inhibition on IAC composition. (A and B) HFF cells spread on FN for 1 h were treated with DMSO, FAK [i], Src [i], or combined FAK [i] + Src [i] for 1 h. IACs were isolated using the workflow in Fig. 2 A and were analyzed by SDS-PAGE (A) and immunoblotting (B). Representative total protein intensity values for each lane are indicated. Graph shows intensity values normalized to the DMSO condition (mean +- SEM, n = 3). Cell lysates from cells spread on FN (TCL) were used as a positive control. Molecular mass values (kD) are displayed. (C) For quantification of immunoblotted membranes in B, the band intensity values for each protein are shown relative to the DMSO condition (mean +- SEM, n = 3). Quantification was not performed for Tf receptor or BAK. (D) Total cell lysates collected from HFF cells spread on FN for 1 h and treated with DMSO or inhibitors for 1 h were analyzed by immunoblotting. Molecular mass values (kD) are displayed. (E) For quantification of immunoblotted membranes in D, phosphorylation values normalized to the corresponding total protein values are shown relative to the DMSO condition (mean +- SEM; n = 4, FAK Y397 , FAK Y576 , and paxillin Y118 ; n = 3, p130Cas Y249 ). t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Representative images are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
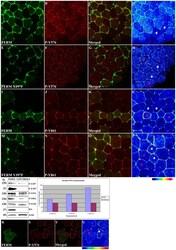
- Figure 5 The FERM domain leads to activation of endogenous FAK in a tyrosine 397 dependent manner. HA-FERM and HA-FERM Y397F injected embryos in one blastomere at the animal pole of two cell stage embryos were processed for whole mount immunostaining using an HA antibody (green) to reveal expressing cells and the respective phospho-specific antibodies (red) as indicated. In each case individual signals for each secondary are shown in addition to a merged image and finally an intensity color coded image of the respective phospho-specific antibody signal. HA-FERM and HA-FERM Y397F injected cells are indicated with red stars and un-injected cells with white stars. (A-D) Levels of phosphorylated tyrosine 576 are elevated in HA-FERM overexpressing cells compared to controls. (E-H) Overexpression of HA-FERM Y397F has no effect on the endogenous levels of phosphorylated tyrosine 576. HA-FERM Y397F expressing cells have the same levels of phosphorylated endogenous FAK on tyrosine 576 with neighboring control cells. (I-L) Levels of phosphorylated tyrosine 861 are elevated in HA-FERM expressing cells compared to controls. (M-P) Overexpression of HA-FERM Y397F has no effect on the endogenous levels of phosphorylated tyrosine 861. HA-FERM Y397F expressing cells have the same levels of phosphorylated endogenous FAK on tyrosine 861 with neighboring control cells. (Q) Total lysates from HA-FERM injected gastrula stage embryos contain comparable levels of endogenous FAK as un-injected contro
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Other assay
Other assay