MA5-13890
antibody from Invitrogen Antibodies
Targeting: CD44
CD44R, CSPG8, HCELL, IN, MC56, MDU2, MDU3, MIC4, Pgp1
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [77]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [5]
- Immunohistochemistry [2]
- Flow cytometry [2]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-13890 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD44 Monoclonal Antibody (156-3C11)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA5-13890 targets CD44 in FACS, IHC (P), ICC/IF, IP and WB applications and shows reactivity with Human, Baboon and Non-human primate samples. The MA5-13890 immunogen is stimulated human leukocytes. MA5-13890 was used in the immunofluorescence analysis to successfully detect CD44 in human mesenchymal stem cells derived from iPSCs.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 156-3C11
- Vial size
- 500 µL
- Concentration
- 0.2 mg/mL
- Storage
- 4° C
Submitted references An instantly fixable and self-adaptive scaffold for skull regeneration by autologous stem cell recruitment and angiogenesis.
Lack of CD44 overexpression and application of concurrent chemoradiotherapy with cisplatin independently indicate excellent prognosis in patients with HPV-positive oropharyngeal cancer.
Quantification of Antibody Persistence for Cell Surface Protein Labeling.
The Human TOR Signaling Regulator Is the Key Indicator of Liver Cancer Patients' Overall Survival: TIPRL/LC3/CD133/CD44 as Potential Biomarkers for Early Liver Cancers.
Filoviruses Infect Rhesus Macaque Synoviocytes in Vivo and Primary Human Synoviocytes in Vitro.
Distinguishing Specific CXCL12 Isoforms on Their Angiogenesis and Vascular Permeability Promoting Properties.
Extracellular Domains I and II of cell-surface glycoprotein CD44 mediate its trans-homophilic dimerization and tumor cluster aggregation.
A Comprehensive Molecular and Clinical Analysis of the piRNA Pathway Genes in Ovarian Cancer.
CDK5-dependent phosphorylation and nuclear translocation of TRIM59 promotes macroH2A1 ubiquitination and tumorigenicity.
The Histone Demethylase LSD1/ΚDM1A Mediates Chemoresistance in Breast Cancer via Regulation of a Stem Cell Program.
Hyaluronan concentration and molecular mass in psoriatic arthritis: biomarkers of disease severity, resistance to treatment, and outcome.
Inhibition of GSK3 and MEK induced cancer stem cell generation via the Wnt and MEK signaling pathways.
Hyaluronan in vocal folds and false vocal folds in patients with recurrent respiratory papillomatosis.
Role of Corneal Stromal Cells on Epithelial Cell Function during Wound Healing.
REST is a crucial regulator for acquiring EMT-like and stemness phenotypes in hormone-refractory prostate cancer.
Soluble CD83 Inhibits T Cell Activation by Binding to the TLR4/MD-2 Complex on CD14(+) Monocytes.
Overexpression of Specific CD44 Isoforms Is Associated with Aggressive Cell Features in Acquired Endocrine Resistance.
Combination of Cancer Stem Cell Markers CD44 and CD24 Is Superior to ALDH1 as a Prognostic Indicator in Breast Cancer Patients with Distant Metastases.
Expression of cell adhesion molecule CD44 in mucoepidermoid carcinoma and its association with the tumor behavior.
Response and resistance to BET bromodomain inhibitors in triple-negative breast cancer.
MIF allele-dependent regulation of the MIF coreceptor CD44 and role in rheumatoid arthritis.
Increased chemoresistance via Snail-Raf kinase inhibitor protein signaling in colorectal cancer in response to a nicotine derivative.
Effects of hypoxia and its relationship with apoptosis, stem cells, and angiogenesis on the thymus of children with congenital heart defects: a morphological and immunohistochemical study.
The clinicopathological and prognostic significance of CD24, CD44, CD133, ALDH1 expressions in invasive ductal carcinoma of the breast: CD44/CD24 expression in breast cancer.
An epigenetically distinct breast cancer cell subpopulation promotes collective invasion.
Interleukin-6 and pro inflammatory status in the breast tumor microenvironment.
The pattern of CD44 and matrix metalloproteinase 9 expression is a useful predictor of ulcerative colitis-associated dysplasia and neoplasia.
Tau and PTEN status as predictive markers for response to trastuzumab and paclitaxel in patients with HER2-positive breast cancer.
Immunohistochemical expression of CD44 standard and E-cadherin in atypical leiomyoma and leiomyosarcoma of the uterus.
Tetraspanin CD9 determines invasiveness and tumorigenicity of human breast cancer cells.
Isolation and characterization of cancer stem cells from high-grade serous ovarian carcinomas.
Y-box-binding protein 1 (YB1) in breast carcinomas: relation to aggressive tumor phenotype and identification of patients at high risk for relapse.
Genetic and phenotypic diversity in breast tumor metastases.
CD44/CD24 as potential prognostic markers in node-positive invasive ductal breast cancer patients treated with adjuvant chemotherapy.
MicroRNA-9 is associated with epithelial-mesenchymal transition, breast cancer stem cell phenotype, and tumor progression in breast cancer.
Triple negative breast tumors in African-American and Hispanic/Latina women are high in CD44+, low in CD24+, and have loss of PTEN.
Estrogen switches pure mucinous breast cancer to invasive lobular carcinoma with mucinous features.
Histological and prognostic importance of CD44(+) /CD24(+) /EpCAM(+) expression in clinical pancreatic cancer.
Immunohistochemical detection of breast cancer stem cells in hormone receptor-positive breast cancer and their role in response to endocrine therapy and clinical outcome.
Overexpression of CD44 accompanies acquired tamoxifen resistance in MCF7 cells and augments their sensitivity to the stromal factors, heregulin and hyaluronan.
CD44 is associated with tumor recurrence and is an independent poor prognostic factor for patients with localized clear cell renal cell carcinoma after nephrectomy.
Neoadjuvant chemotherapy with gemcitabine for pancreatic cancer increases in situ expression of the apoptosis marker M30 and stem cell marker CD44.
Epigenetic regulation of cancer stem cell genes in triple-negative breast cancer.
The critical role of CD133(+)CD44(+/high) tumor cells in hematogenous metastasis of liver cancers.
Identification of prognostic biomarkers for glioblastomas using protein expression profiling.
Expression of CD44 and MMP-2: possible association with histopathological features of pleuro-pulmonary solitary fibrous tumors.
A comprehensive characterization study of human bone marrow mscs with an emphasis on molecular and ultrastructural properties.
An increase in cancer stem cell population after primary systemic therapy is a poor prognostic factor in breast cancer.
The JAK2/STAT3 signaling pathway is required for growth of CD44⁺CD24⁻ stem cell-like breast cancer cells in human tumors.
Novel immunofluorescence protocol for multimarker assessment of putative disseminating breast cancer stem cells.
Role of versican, hyaluronan and CD44 in ovarian cancer metastasis.
Aggressive progression of breast cancer with microscopic pulmonary emboli possessing a stem cell-like phenotype independent of its origin.
Simple karyotype and bcl-6 expression predict a diagnosis of Burkitt lymphoma and better survival in IG-MYC rearranged high-grade B-cell lymphomas.
Cellular and genetic diversity in the progression of in situ human breast carcinomas to an invasive phenotype.
TGF-β Receptor Inhibitors Target the CD44(high)/Id1(high) Glioma-Initiating Cell Population in Human Glioblastoma.
Expression of CD44s in incidental prostate cancer is more strongly associated with Gleason scores on subsequent radical prostatectomies than conventional prognostic parameters.
Human mesenchymal stem cells induce E-cadherin degradation in breast carcinoma spheroids by activating ADAM10.
Plasmablastic lymphomas with MYC/IgH rearrangement: report of three cases and review of the literature.
CD44-positive cells are responsible for gemcitabine resistance in pancreatic cancer cells.
Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential Paclitaxel and epirubicin-based chemotherapy for breast cancers.
Identification of CD44v6(+)/CD24- breast carcinoma cells in primary human tumors by quantum dot-conjugated antibodies.
The CD44+/CD24- phenotype is enriched in basal-like breast tumors.
The clinicopathologic and prognostic significance of CD44+/CD24(-/low) and CD44-/CD24+ tumor cells in invasive breast carcinomas.
Periodontal tissue engineering by transplantation of multilayered sheets of phenotypically modified gingival fibroblasts.
Cell-surface MMP-9 regulates the invasive capacity of leukemia blast cells with monocytic features.
Cell-surface MMP-9 regulates the invasive capacity of leukemia blast cells with monocytic features.
Rare steroid receptor-negative basal-like tumorigenic cells in luminal subtype human breast cancer xenografts.
Moesin expression is a marker of basal breast carcinomas.
Estrogen insensitivity in a model of estrogen receptor positive breast cancer lymph node metastasis.
IL-6 triggers malignant features in mammospheres from human ductal breast carcinoma and normal mammary gland.
Formation of hyaluronan- and versican-rich pericellular matrix by prostate cancer cells promotes cell motility.
Estrogen receptor positive breast cancer metastasis: altered hormonal sensitivity and tumor aggressiveness in lymphatic vessels and lymph nodes.
Most early disseminated cancer cells detected in bone marrow of breast cancer patients have a putative breast cancer stem cell phenotype.
SHAP potentiates the CD44-mediated leukocyte adhesion to the hyaluronan substratum.
Downregulation of BRG-1 repressed expression of CD44s in cervical neuroendocrine carcinoma and adenocarcinoma.
Novel prognostic value of nuclear epidermal growth factor receptor in breast cancer.
Loss of CD10 (neutral endopeptidase) is a frequent and early event in human prostate cancer.
Lu G, Xu Y, Liu Q, Chen M, Sun H, Wang P, Li X, Wang Y, Li X, Hui X, Luo E, Liu J, Jiang Q, Liang J, Fan Y, Sun Y, Zhang X
Nature communications 2022 May 6;13(1):2499
Nature communications 2022 May 6;13(1):2499
Lack of CD44 overexpression and application of concurrent chemoradiotherapy with cisplatin independently indicate excellent prognosis in patients with HPV-positive oropharyngeal cancer.
Biesaga B, Kołodziej-Rzepa M, Janecka-Widła A, Słonina D, Halaszka K, Przewoźnik M, Mucha-Małecka A
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2021;43(1):99-113
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2021;43(1):99-113
Quantification of Antibody Persistence for Cell Surface Protein Labeling.
Dempsey ME, Woodford-Berry O, Darling EM
Cellular and molecular bioengineering 2021 Jun;14(3):267-277
Cellular and molecular bioengineering 2021 Jun;14(3):267-277
The Human TOR Signaling Regulator Is the Key Indicator of Liver Cancer Patients' Overall Survival: TIPRL/LC3/CD133/CD44 as Potential Biomarkers for Early Liver Cancers.
Jun SY, Yoon HR, Yoon JY, Jeon SJ, Lee JJ, Halder D, Kim JM, Kim NS
Cancers 2021 Jun 11;13(12)
Cancers 2021 Jun 11;13(12)
Filoviruses Infect Rhesus Macaque Synoviocytes in Vivo and Primary Human Synoviocytes in Vitro.
Cooper TK, Logue J, Liu DX, Perry DL, Hart RJ, Hischak AMW, Bernbaum JG, Gerhardt DM, Rojas O, Bohannon JK, Hagen KR, Johnson RF, Crozier I, Jahrling PB, Hensley LE, Bennett RS
The American journal of pathology 2020 Sep;190(9):1867-1880
The American journal of pathology 2020 Sep;190(9):1867-1880
Distinguishing Specific CXCL12 Isoforms on Their Angiogenesis and Vascular Permeability Promoting Properties.
Chang CW, Seibel AJ, Avendano A, Cortes-Medina MG, Song JW
Advanced healthcare materials 2020 Feb;9(4):e1901399
Advanced healthcare materials 2020 Feb;9(4):e1901399
Extracellular Domains I and II of cell-surface glycoprotein CD44 mediate its trans-homophilic dimerization and tumor cluster aggregation.
Kawaguchi M, Dashzeveg N, Cao Y, Jia Y, Liu X, Shen Y, Liu H
The Journal of biological chemistry 2020 Feb 28;295(9):2640-2649
The Journal of biological chemistry 2020 Feb 28;295(9):2640-2649
A Comprehensive Molecular and Clinical Analysis of the piRNA Pathway Genes in Ovarian Cancer.
Lee E, Lokman NA, Oehler MK, Ricciardelli C, Grutzner F
Cancers 2020 Dec 22;13(1)
Cancers 2020 Dec 22;13(1)
CDK5-dependent phosphorylation and nuclear translocation of TRIM59 promotes macroH2A1 ubiquitination and tumorigenicity.
Sang Y, Li Y, Zhang Y, Alvarez AA, Yu B, Zhang W, Hu B, Cheng SY, Feng H
Nature communications 2019 Sep 5;10(1):4013
Nature communications 2019 Sep 5;10(1):4013
The Histone Demethylase LSD1/ΚDM1A Mediates Chemoresistance in Breast Cancer via Regulation of a Stem Cell Program.
Verigos J, Karakaidos P, Kordias D, Papoudou-Bai A, Evangelou Z, Harissis HV, Klinakis A, Magklara A
Cancers 2019 Oct 17;11(10)
Cancers 2019 Oct 17;11(10)
Hyaluronan concentration and molecular mass in psoriatic arthritis: biomarkers of disease severity, resistance to treatment, and outcome.
Hellman U, Engström-Laurent A, Larsson A, Lindqvist U
Scandinavian journal of rheumatology 2019 Jul;48(4):284-293
Scandinavian journal of rheumatology 2019 Jul;48(4):284-293
Inhibition of GSK3 and MEK induced cancer stem cell generation via the Wnt and MEK signaling pathways.
Liao S, Gan L, Qin W, Liu C, Mei Z
Oncology reports 2018 Oct;40(4):2005-2013
Oncology reports 2018 Oct;40(4):2005-2013
Hyaluronan in vocal folds and false vocal folds in patients with recurrent respiratory papillomatosis.
Holm A, Hellman U, Laurent C, Laurell G, Nylander K, Olofsson K
Acta oto-laryngologica 2018 Nov;138(11):1020-1027
Acta oto-laryngologica 2018 Nov;138(11):1020-1027
Role of Corneal Stromal Cells on Epithelial Cell Function during Wound Healing.
Kowtharapu BS, Murín R, Jünemann AGM, Stachs O
International journal of molecular sciences 2018 Feb 4;19(2)
International journal of molecular sciences 2018 Feb 4;19(2)
REST is a crucial regulator for acquiring EMT-like and stemness phenotypes in hormone-refractory prostate cancer.
Chang YT, Lin TP, Campbell M, Pan CC, Lee SH, Lee HC, Yang MH, Kung HJ, Chang PC
Scientific reports 2017 Mar 3;7:42795
Scientific reports 2017 Mar 3;7:42795
Soluble CD83 Inhibits T Cell Activation by Binding to the TLR4/MD-2 Complex on CD14(+) Monocytes.
Horvatinovich JM, Grogan EW, Norris M, Steinkasserer A, Lemos H, Mellor AL, Tcherepanova IY, Nicolette CA, DeBenedette MA
Journal of immunology (Baltimore, Md. : 1950) 2017 Mar 15;198(6):2286-2301
Journal of immunology (Baltimore, Md. : 1950) 2017 Mar 15;198(6):2286-2301
Overexpression of Specific CD44 Isoforms Is Associated with Aggressive Cell Features in Acquired Endocrine Resistance.
Bellerby R, Smith C, Kyme S, Gee J, Günthert U, Green A, Rakha E, Barrett-Lee P, Hiscox S
Frontiers in oncology 2016;6:145
Frontiers in oncology 2016;6:145
Combination of Cancer Stem Cell Markers CD44 and CD24 Is Superior to ALDH1 as a Prognostic Indicator in Breast Cancer Patients with Distant Metastases.
Horimoto Y, Arakawa A, Sasahara N, Tanabe M, Sai S, Himuro T, Saito M
PloS one 2016;11(10):e0165253
PloS one 2016;11(10):e0165253
Expression of cell adhesion molecule CD44 in mucoepidermoid carcinoma and its association with the tumor behavior.
Binmadi N, Elsissi A, Elsissi N
Head & face medicine 2016 Jan 29;12:8
Head & face medicine 2016 Jan 29;12:8
Response and resistance to BET bromodomain inhibitors in triple-negative breast cancer.
Shu S, Lin CY, He HH, Witwicki RM, Tabassum DP, Roberts JM, Janiszewska M, Huh SJ, Liang Y, Ryan J, Doherty E, Mohammed H, Guo H, Stover DG, Ekram MB, Brown J, D'Santos C, Krop IE, Dillon D, McKeown M, Ott C, Qi J, Ni M, Rao PK, Duarte M, Wu SY, Chiang CM, Anders L, Young RA, Winer E, Letai A, Barry WT, Carroll JS, Long H, Brown M, Liu XS, Meyer CA, Bradner JE, Polyak K
Nature 2016 Jan 21;529(7586):413-417
Nature 2016 Jan 21;529(7586):413-417
MIF allele-dependent regulation of the MIF coreceptor CD44 and role in rheumatoid arthritis.
Yoo SA, Leng L, Kim BJ, Du X, Tilstam PV, Kim KH, Kong JS, Yoon HJ, Liu A, Wang T, Song Y, Sauler M, Bernhagen J, Ritchlin CT, Lee P, Cho CS, Kim WU, Bucala R
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 6;113(49):E7917-E7926
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 6;113(49):E7917-E7926
Increased chemoresistance via Snail-Raf kinase inhibitor protein signaling in colorectal cancer in response to a nicotine derivative.
Lee TY, Liu CL, Chang YC, Nieh S, Lin YS, Jao SW, Chen SF, Liu TY
Oncotarget 2016 Apr 26;7(17):23512-20
Oncotarget 2016 Apr 26;7(17):23512-20
Effects of hypoxia and its relationship with apoptosis, stem cells, and angiogenesis on the thymus of children with congenital heart defects: a morphological and immunohistochemical study.
Ceyran AB, Şenol S, Güzelmeriç F, Tunçer E, Tongut A, Özbek B, Şavluk Ö, Aydın A, Ceyran H
International journal of clinical and experimental pathology 2015;8(7):8038-47
International journal of clinical and experimental pathology 2015;8(7):8038-47
The clinicopathological and prognostic significance of CD24, CD44, CD133, ALDH1 expressions in invasive ductal carcinoma of the breast: CD44/CD24 expression in breast cancer.
Kapucuoğlu N, Bozkurt KK, Başpınar Ş, Koçer M, Eroğlu HE, Akdeniz R, Akçil M
Pathology, research and practice 2015 Oct;211(10):740-7
Pathology, research and practice 2015 Oct;211(10):740-7
An epigenetically distinct breast cancer cell subpopulation promotes collective invasion.
Westcott JM, Prechtl AM, Maine EA, Dang TT, Esparza MA, Sun H, Zhou Y, Xie Y, Pearson GW
The Journal of clinical investigation 2015 May;125(5):1927-43
The Journal of clinical investigation 2015 May;125(5):1927-43
Interleukin-6 and pro inflammatory status in the breast tumor microenvironment.
Sanguinetti A, Santini D, Bonafè M, Taffurelli M, Avenia N
World journal of surgical oncology 2015 Mar 28;13:129
World journal of surgical oncology 2015 Mar 28;13:129
The pattern of CD44 and matrix metalloproteinase 9 expression is a useful predictor of ulcerative colitis-associated dysplasia and neoplasia.
AbdElazeem MA, El-Sayed M
Annals of diagnostic pathology 2015 Dec;19(6):369-74
Annals of diagnostic pathology 2015 Dec;19(6):369-74
Tau and PTEN status as predictive markers for response to trastuzumab and paclitaxel in patients with HER2-positive breast cancer.
Koo DH, Lee HJ, Ahn JH, Yoon DH, Kim SB, Gong G, Son BH, Ahn SH, Jung KH
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2015 Aug;36(8):5865-71
Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine 2015 Aug;36(8):5865-71
Immunohistochemical expression of CD44 standard and E-cadherin in atypical leiomyoma and leiomyosarcoma of the uterus.
Kaygusuz EI
Journal of obstetrics and gynaecology : the journal of the Institute of Obstetrics and Gynaecology 2015 Apr;35(3):279-82
Journal of obstetrics and gynaecology : the journal of the Institute of Obstetrics and Gynaecology 2015 Apr;35(3):279-82
Tetraspanin CD9 determines invasiveness and tumorigenicity of human breast cancer cells.
Rappa G, Green TM, Karbanová J, Corbeil D, Lorico A
Oncotarget 2015 Apr 10;6(10):7970-91
Oncotarget 2015 Apr 10;6(10):7970-91
Isolation and characterization of cancer stem cells from high-grade serous ovarian carcinomas.
He QZ, Luo XZ, Wang K, Zhou Q, Ao H, Yang Y, Li SX, Li Y, Zhu HT, Duan T
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2014;33(1):173-84
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2014;33(1):173-84
Y-box-binding protein 1 (YB1) in breast carcinomas: relation to aggressive tumor phenotype and identification of patients at high risk for relapse.
Mylona E, Melissaris S, Giannopoulou I, Theohari I, Papadimitriou C, Keramopoulos A, Nakopoulou L
European journal of surgical oncology : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology 2014 Mar;40(3):289-96
European journal of surgical oncology : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology 2014 Mar;40(3):289-96
Genetic and phenotypic diversity in breast tumor metastases.
Almendro V, Kim HJ, Cheng YK, Gönen M, Itzkovitz S, Argani P, van Oudenaarden A, Sukumar S, Michor F, Polyak K
Cancer research 2014 Mar 1;74(5):1338-48
Cancer research 2014 Mar 1;74(5):1338-48
CD44/CD24 as potential prognostic markers in node-positive invasive ductal breast cancer patients treated with adjuvant chemotherapy.
Adamczyk A, Niemiec JA, Ambicka A, Mucha-Małecka A, Mituś J, Ryś J
Journal of molecular histology 2014 Feb;45(1):35-45
Journal of molecular histology 2014 Feb;45(1):35-45
MicroRNA-9 is associated with epithelial-mesenchymal transition, breast cancer stem cell phenotype, and tumor progression in breast cancer.
Gwak JM, Kim HJ, Kim EJ, Chung YR, Yun S, Seo AN, Lee HJ, Park SY
Breast cancer research and treatment 2014 Aug;147(1):39-49
Breast cancer research and treatment 2014 Aug;147(1):39-49
Triple negative breast tumors in African-American and Hispanic/Latina women are high in CD44+, low in CD24+, and have loss of PTEN.
Wu Y, Sarkissyan M, Elshimali Y, Vadgama JV
PloS one 2013;8(10):e78259
PloS one 2013;8(10):e78259
Estrogen switches pure mucinous breast cancer to invasive lobular carcinoma with mucinous features.
Jambal P, Badtke MM, Harrell JC, Borges VF, Post MD, Sollender GE, Spillman MA, Horwitz KB, Jacobsen BM
Breast cancer research and treatment 2013 Jan;137(2):431-48
Breast cancer research and treatment 2013 Jan;137(2):431-48
Histological and prognostic importance of CD44(+) /CD24(+) /EpCAM(+) expression in clinical pancreatic cancer.
Ohara Y, Oda T, Sugano M, Hashimoto S, Enomoto T, Yamada K, Akashi Y, Miyamoto R, Kobayashi A, Fukunaga K, Morishita Y, Ohkohchi N
Cancer science 2013 Aug;104(8):1127-34
Cancer science 2013 Aug;104(8):1127-34
Immunohistochemical detection of breast cancer stem cells in hormone receptor-positive breast cancer and their role in response to endocrine therapy and clinical outcome.
Hashimoto K, Shimizu C, Tsuda H, Saji S, Osaki A, Shigekawa T, Aogi K
Oncology 2012;82(3):168-74
Oncology 2012;82(3):168-74
Overexpression of CD44 accompanies acquired tamoxifen resistance in MCF7 cells and augments their sensitivity to the stromal factors, heregulin and hyaluronan.
Hiscox S, Baruha B, Smith C, Bellerby R, Goddard L, Jordan N, Poghosyan Z, Nicholson RI, Barrett-Lee P, Gee J
BMC cancer 2012 Oct 6;12:458
BMC cancer 2012 Oct 6;12:458
CD44 is associated with tumor recurrence and is an independent poor prognostic factor for patients with localized clear cell renal cell carcinoma after nephrectomy.
Jeong BJ, Liang ZL, Huang SM, Lim JS, Kim JM, Lee HJ
Experimental and therapeutic medicine 2012 May;3(5):811-817
Experimental and therapeutic medicine 2012 May;3(5):811-817
Neoadjuvant chemotherapy with gemcitabine for pancreatic cancer increases in situ expression of the apoptosis marker M30 and stem cell marker CD44.
Tajima H, Ohta T, Kitagawa H, Okamoto K, Sakai S, Kinoshita J, Makino I, Furukawa H, Hayashi H, Nakamura K, Oyama K, Inokuchi M, Nakagawara H, Fujita H, Takamura H, Ninomiya I, Fushida S, Tani T, Fujimura T, Kitamura S, Ikeda H, Tsuneyama K
Oncology letters 2012 Jun;3(6):1186-1190
Oncology letters 2012 Jun;3(6):1186-1190
Epigenetic regulation of cancer stem cell genes in triple-negative breast cancer.
Kagara N, Huynh KT, Kuo C, Okano H, Sim MS, Elashoff D, Chong K, Giuliano AE, Hoon DS
The American journal of pathology 2012 Jul;181(1):257-67
The American journal of pathology 2012 Jul;181(1):257-67
The critical role of CD133(+)CD44(+/high) tumor cells in hematogenous metastasis of liver cancers.
Hou Y, Zou Q, Ge R, Shen F, Wang Y
Cell research 2012 Jan;22(1):259-72
Cell research 2012 Jan;22(1):259-72
Identification of prognostic biomarkers for glioblastomas using protein expression profiling.
Jung Y, Joo KM, Seong DH, Choi YL, Kong DS, Kim Y, Kim MH, Jin J, Suh YL, Seol HJ, Shin CS, Lee JI, Kim JH, Song SY, Nam DH
International journal of oncology 2012 Apr;40(4):1122-32
International journal of oncology 2012 Apr;40(4):1122-32
Expression of CD44 and MMP-2: possible association with histopathological features of pleuro-pulmonary solitary fibrous tumors.
Demırağ F, Cakir E, Alpar S, Taştepe I, Kaya S
Turk patoloji dergisi 2011 May;27(2):127-33
Turk patoloji dergisi 2011 May;27(2):127-33
A comprehensive characterization study of human bone marrow mscs with an emphasis on molecular and ultrastructural properties.
Karaöz E, Okçu A, Gacar G, Sağlam O, Yürüker S, Kenar H
Journal of cellular physiology 2011 May;226(5):1367-82
Journal of cellular physiology 2011 May;226(5):1367-82
An increase in cancer stem cell population after primary systemic therapy is a poor prognostic factor in breast cancer.
Lee HE, Kim JH, Kim YJ, Choi SY, Kim SW, Kang E, Chung IY, Kim IA, Kim EJ, Choi Y, Ryu HS, Park SY
British journal of cancer 2011 May 24;104(11):1730-8
British journal of cancer 2011 May 24;104(11):1730-8
The JAK2/STAT3 signaling pathway is required for growth of CD44⁺CD24⁻ stem cell-like breast cancer cells in human tumors.
Marotta LL, Almendro V, Marusyk A, Shipitsin M, Schemme J, Walker SR, Bloushtain-Qimron N, Kim JJ, Choudhury SA, Maruyama R, Wu Z, Gönen M, Mulvey LA, Bessarabova MO, Huh SJ, Silver SJ, Kim SY, Park SY, Lee HE, Anderson KS, Richardson AL, Nikolskaya T, Nikolsky Y, Liu XS, Root DE, Hahn WC, Frank DA, Polyak K
The Journal of clinical investigation 2011 Jul;121(7):2723-35
The Journal of clinical investigation 2011 Jul;121(7):2723-35
Novel immunofluorescence protocol for multimarker assessment of putative disseminating breast cancer stem cells.
Balic M, Rapp N, Stanzer S, Lin H, Strutz J, Szkandera J, Daidone MG, Samonigg H, Cote RJ, Dandachi N
Applied immunohistochemistry & molecular morphology : AIMM 2011 Jan;19(1):33-40
Applied immunohistochemistry & molecular morphology : AIMM 2011 Jan;19(1):33-40
Role of versican, hyaluronan and CD44 in ovarian cancer metastasis.
Ween MP, Oehler MK, Ricciardelli C
International journal of molecular sciences 2011 Jan 31;12(2):1009-29
International journal of molecular sciences 2011 Jan 31;12(2):1009-29
Aggressive progression of breast cancer with microscopic pulmonary emboli possessing a stem cell-like phenotype independent of its origin.
Kawasaki H, Ogura H, Arai Y, Baba S, Kosugi I, Tsutsui Y, Iwashita T
Pathology international 2010 Mar;60(3):228-34
Pathology international 2010 Mar;60(3):228-34
Simple karyotype and bcl-6 expression predict a diagnosis of Burkitt lymphoma and better survival in IG-MYC rearranged high-grade B-cell lymphomas.
Seegmiller AC, Garcia R, Huang R, Maleki A, Karandikar NJ, Chen W
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2010 Jul;23(7):909-20
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2010 Jul;23(7):909-20
Cellular and genetic diversity in the progression of in situ human breast carcinomas to an invasive phenotype.
Park SY, Gönen M, Kim HJ, Michor F, Polyak K
The Journal of clinical investigation 2010 Feb;120(2):636-44
The Journal of clinical investigation 2010 Feb;120(2):636-44
TGF-β Receptor Inhibitors Target the CD44(high)/Id1(high) Glioma-Initiating Cell Population in Human Glioblastoma.
Anido J, Sáez-Borderías A, Gonzàlez-Juncà A, Rodón L, Folch G, Carmona MA, Prieto-Sánchez RM, Barba I, Martínez-Sáez E, Prudkin L, Cuartas I, Raventós C, Martínez-Ricarte F, Poca MA, García-Dorado D, Lahn MM, Yingling JM, Rodón J, Sahuquillo J, Baselga J, Seoane J
Cancer cell 2010 Dec 14;18(6):655-68
Cancer cell 2010 Dec 14;18(6):655-68
Expression of CD44s in incidental prostate cancer is more strongly associated with Gleason scores on subsequent radical prostatectomies than conventional prognostic parameters.
Gunia S, May M, Koch S, Dietel M, Erbersdobler A
Pathobiology : journal of immunopathology, molecular and cellular biology 2009;76(6):286-92
Pathobiology : journal of immunopathology, molecular and cellular biology 2009;76(6):286-92
Human mesenchymal stem cells induce E-cadherin degradation in breast carcinoma spheroids by activating ADAM10.
Dittmer A, Hohlfeld K, Lützkendorf J, Müller LP, Dittmer J
Cellular and molecular life sciences : CMLS 2009 Sep;66(18):3053-65
Cellular and molecular life sciences : CMLS 2009 Sep;66(18):3053-65
Plasmablastic lymphomas with MYC/IgH rearrangement: report of three cases and review of the literature.
Bogusz AM, Seegmiller AC, Garcia R, Shang P, Ashfaq R, Chen W
American journal of clinical pathology 2009 Oct;132(4):597-605
American journal of clinical pathology 2009 Oct;132(4):597-605
CD44-positive cells are responsible for gemcitabine resistance in pancreatic cancer cells.
Hong SP, Wen J, Bang S, Park S, Song SY
International journal of cancer 2009 Nov 15;125(10):2323-31
International journal of cancer 2009 Nov 15;125(10):2323-31
Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential Paclitaxel and epirubicin-based chemotherapy for breast cancers.
Tanei T, Morimoto K, Shimazu K, Kim SJ, Tanji Y, Taguchi T, Tamaki Y, Noguchi S
Clinical cancer research : an official journal of the American Association for Cancer Research 2009 Jun 15;15(12):4234-41
Clinical cancer research : an official journal of the American Association for Cancer Research 2009 Jun 15;15(12):4234-41
Identification of CD44v6(+)/CD24- breast carcinoma cells in primary human tumors by quantum dot-conjugated antibodies.
Snyder EL, Bailey D, Shipitsin M, Polyak K, Loda M
Laboratory investigation; a journal of technical methods and pathology 2009 Aug;89(8):857-66
Laboratory investigation; a journal of technical methods and pathology 2009 Aug;89(8):857-66
The CD44+/CD24- phenotype is enriched in basal-like breast tumors.
Honeth G, Bendahl PO, Ringnér M, Saal LH, Gruvberger-Saal SK, Lövgren K, Grabau D, Fernö M, Borg A, Hegardt C
Breast cancer research : BCR 2008;10(3):R53
Breast cancer research : BCR 2008;10(3):R53
The clinicopathologic and prognostic significance of CD44+/CD24(-/low) and CD44-/CD24+ tumor cells in invasive breast carcinomas.
Mylona E, Giannopoulou I, Fasomytakis E, Nomikos A, Magkou C, Bakarakos P, Nakopoulou L
Human pathology 2008 Jul;39(7):1096-102
Human pathology 2008 Jul;39(7):1096-102
Periodontal tissue engineering by transplantation of multilayered sheets of phenotypically modified gingival fibroblasts.
Nakajima K, Abe T, Tanaka M, Hara Y
Journal of periodontal research 2008 Dec;43(6):681-8
Journal of periodontal research 2008 Dec;43(6):681-8
Cell-surface MMP-9 regulates the invasive capacity of leukemia blast cells with monocytic features.
Paupert J, Mansat-De Mas V, Demur C, Salles B, Muller C
Cell cycle (Georgetown, Tex.) 2008 Apr 15;7(8):1047-53
Cell cycle (Georgetown, Tex.) 2008 Apr 15;7(8):1047-53
Cell-surface MMP-9 regulates the invasive capacity of leukemia blast cells with monocytic features.
Paupert J, Mansat-De Mas V, Demur C, Salles B, Muller C
Cell cycle (Georgetown, Tex.) 2008 Apr 15;7(8):1047-53
Cell cycle (Georgetown, Tex.) 2008 Apr 15;7(8):1047-53
Rare steroid receptor-negative basal-like tumorigenic cells in luminal subtype human breast cancer xenografts.
Horwitz KB, Dye WW, Harrell JC, Kabos P, Sartorius CA
Proceedings of the National Academy of Sciences of the United States of America 2008 Apr 15;105(15):5774-9
Proceedings of the National Academy of Sciences of the United States of America 2008 Apr 15;105(15):5774-9
Moesin expression is a marker of basal breast carcinomas.
Charafe-Jauffret E, Monville F, Bertucci F, Esterni B, Ginestier C, Finetti P, Cervera N, Geneix J, Hassanein M, Rabayrol L, Sobol H, Taranger-Charpin C, Xerri L, Viens P, Birnbaum D, Jacquemier J
International journal of cancer 2007 Oct 15;121(8):1779-85
International journal of cancer 2007 Oct 15;121(8):1779-85
Estrogen insensitivity in a model of estrogen receptor positive breast cancer lymph node metastasis.
Harrell JC, Dye WW, Harvell DM, Pinto M, Jedlicka P, Sartorius CA, Horwitz KB
Cancer research 2007 Nov 1;67(21):10582-91
Cancer research 2007 Nov 1;67(21):10582-91
IL-6 triggers malignant features in mammospheres from human ductal breast carcinoma and normal mammary gland.
Sansone P, Storci G, Tavolari S, Guarnieri T, Giovannini C, Taffurelli M, Ceccarelli C, Santini D, Paterini P, Marcu KB, Chieco P, Bonafè M
The Journal of clinical investigation 2007 Dec;117(12):3988-4002
The Journal of clinical investigation 2007 Dec;117(12):3988-4002
Formation of hyaluronan- and versican-rich pericellular matrix by prostate cancer cells promotes cell motility.
Ricciardelli C, Russell DL, Ween MP, Mayne K, Suwiwat S, Byers S, Marshall VR, Tilley WD, Horsfall DJ
The Journal of biological chemistry 2007 Apr 6;282(14):10814-25
The Journal of biological chemistry 2007 Apr 6;282(14):10814-25
Estrogen receptor positive breast cancer metastasis: altered hormonal sensitivity and tumor aggressiveness in lymphatic vessels and lymph nodes.
Harrell JC, Dye WW, Allred DC, Jedlicka P, Spoelstra NS, Sartorius CA, Horwitz KB
Cancer research 2006 Sep 15;66(18):9308-15
Cancer research 2006 Sep 15;66(18):9308-15
Most early disseminated cancer cells detected in bone marrow of breast cancer patients have a putative breast cancer stem cell phenotype.
Balic M, Lin H, Young L, Hawes D, Giuliano A, McNamara G, Datar RH, Cote RJ
Clinical cancer research : an official journal of the American Association for Cancer Research 2006 Oct 1;12(19):5615-21
Clinical cancer research : an official journal of the American Association for Cancer Research 2006 Oct 1;12(19):5615-21
SHAP potentiates the CD44-mediated leukocyte adhesion to the hyaluronan substratum.
Zhuo L, Kanamori A, Kannagi R, Itano N, Wu J, Hamaguchi M, Ishiguro N, Kimata K
The Journal of biological chemistry 2006 Jul 21;281(29):20303-14
The Journal of biological chemistry 2006 Jul 21;281(29):20303-14
Downregulation of BRG-1 repressed expression of CD44s in cervical neuroendocrine carcinoma and adenocarcinoma.
Kuo KT, Liang CW, Hsiao CH, Lin CH, Chen CA, Sheu BC, Lin MC
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2006 Dec;19(12):1570-7
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2006 Dec;19(12):1570-7
Novel prognostic value of nuclear epidermal growth factor receptor in breast cancer.
Lo HW, Xia W, Wei Y, Ali-Seyed M, Huang SF, Hung MC
Cancer research 2005 Jan 1;65(1):338-48
Cancer research 2005 Jan 1;65(1):338-48
Loss of CD10 (neutral endopeptidase) is a frequent and early event in human prostate cancer.
Freedland SJ, Seligson DB, Liu AY, Pantuck AJ, Paik SH, Horvath S, Wieder JA, Zisman A, Nguyen D, Tso CL, Palotie AV, Belldegrun AS
The Prostate 2003 Apr 1;55(1):71-80
The Prostate 2003 Apr 1;55(1):71-80
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of CD44 Std using CD44 Std Monoclonal Antibody (Product # MA5-13890) on HUVEC Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane enriched extracts (30 µg lysate) of MDA-MB-231 (Lane 1), U-937 (Lane 2), and U-87 MG (Lane 3). The blots were probed with Mouse Anti-CD44 Monoclonal Antibody (Product # MA5-13890, 2 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal Secondary Antibody, HRP conjugate (Product # A28177, 0.25 µg/mL, 1:4000 dilution). A 82 kDa band corresponding to CD44 was observed across the cell lines tested. Known quantity of protein samples were electrophoresed using Novex®NuPAGE®4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
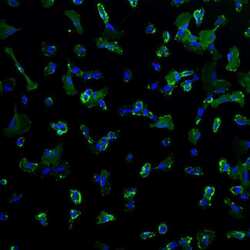
- Experimental details
- Immunofluorescence analysis of mesenchymal stem cell marker CD44 (green) in human mesenchymal stem cells CY-2 derived from iPSCs. The cells were fixed with 4% Paraformaldehyde for 30 mins and blocked with blocking buffer for 1 hour at room temperature. Cells were stained with CD44 mouse monoclonal antibody (Product # MA5-13890) at a dilution of 1:100 in staining buffer overnight at 4°C, and then incubated with secondary antibody AlexaFluor 488 donkey anti-mouse (Product # A-21202) at a dilution of 1:250 for 1 hour at room temperature. Nuclear (blue) were stained with NucBlue Fixed Cell Stain (Product # R37606). Images were taken at 20X magnification. Data courtesy of Dr. Wei Zheng's lab.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
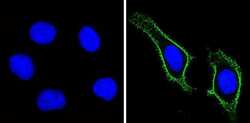
- Experimental details
- Immunofluorescent analysis of CD44 Std (green) showing staining in the membrane of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a CD44 Std monoclonal antibody (Product # MA5-13890) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
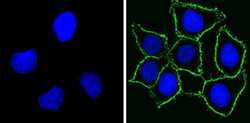
- Experimental details
- Immunofluorescent analysis of CD44 Std (green) showing staining in the membrane of MDA-MB-231 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a CD44 Std monoclonal antibody (Product # MA5-13890) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of CD44 Monoclonal Antibody was performed using 70% confluent log phase Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with CD44 (156-3C11) Mouse Monoclonal Antibody (Product # MA5-13890) at 2 µg/mL in 0.1% BSA, incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing membranous localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
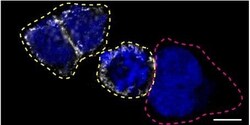
- Experimental details
- Immunofluorescence of CD44 was performed using HAP1 wild-type and CD44 KO cells that were transfected with a green or a far red fluorescent dye, respectively. Post-transfection, WT and KO cells were mixed and plated to a 1:1 ratio on coverslips as a mosaic and incubated for 24 hrs. Cells were fixed in 4% PFA (in PBS) for 15 min; cells were permeabilized with 0.1% Triton X-100 for 10 min at RT and blocked with PBS with 5% BSA, 5% goat serum, and 0.01% Triton X-100 for 30 min. Cells were stained with the CD44 monoclonal antibody (Product # MA5-13890) at a 1:200 dilution overnight at 4°C. Secondary antibody incubation was performed using 0.5 µg/mL of Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 555 antibody (Product # A21424) together with DAPI for 1 hr. Imaging was performed with a 40X oil objective and analysis was performed using Image J. Cell image represents a single focal plane; WT and KO cells are outlined with a yellow (WT) or magenta (KO) dashed line. Data courtesy of YCharOS Inc., an open science company with the mission of characterizing commercially available antibodies using knockout validation.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
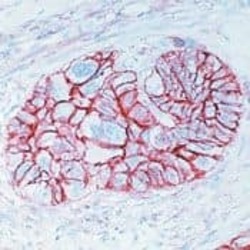
- Experimental details
- Formalin-fixed, paraffin-embedded human esophageal carcinoma stained with CD44 antibody using peroxidase-conjugate and AEC chromogen. Note cell membrane staining of tumor cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
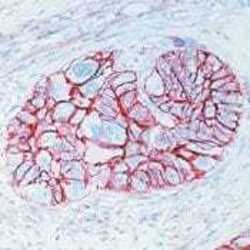
- Experimental details
- Formalin-fixed, paraffin-embedded human esophageal carcinoma stained with CD44 antibody using peroxidase-conjugate and AEC chromogen. Note cell membrane staining of tumor cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
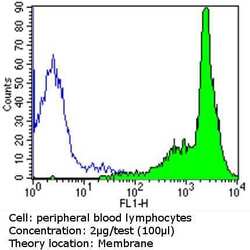
- Experimental details
- Flow cytometry analysis of CD44 Std in PBMC cells (green) compared to an isotype control (blue). Human blood was collected, combined with a hydrophilic polysaccharide, centrifuged, transferred to a conical tube and washed with PBS. 50 µL of cell solution was added to each tube at a dilution of 2x10^7 cells/mL, followed by the addition of 50 µL of isotype control and primary antibody (Product # MA5-13890) at a dilution of 2 µg/test. Cells were incubated for 30 min at 4ºC and washed with a cell buffer, followed by incubation with a DyLight 488-conjugated goat anti-mouse IgG (H+L) secondary for 30 min at 4ºC in the dark. FACS analysis was performed using 400 µL of cell buffer.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
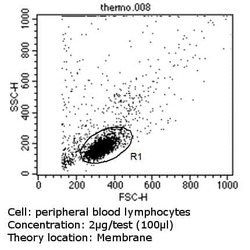
- Experimental details
- Flow cytometry analysis of CD44 Std in PBMC cells (green) compared to an isotype control (blue). Human blood was collected, combined with a hydrophilic polysaccharide, centrifuged, transferred to a conical tube and washed with PBS. 50 µL of cell solution was added to each tube at a dilution of 2x10^7 cells/mL, followed by the addition of 50 µL of isotype control and primary antibody (Product # MA5-13890) at a dilution of 2 µg/test. Cells were incubated for 30 min at 4ºC and washed with a cell buffer, followed by incubation with a DyLight 488-conjugated goat anti-mouse IgG (H+L) secondary for 30 min at 4ºC in the dark. FACS analysis was performed using 400 µL of cell buffer.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of CD44 Std using CD44 Std Monoclonal Antibody (Product # MA5-13890) on Native Human HeLa Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 PCa cell lines profiled for REST expression and characterization of EMT and stemness signature. ( A and B ) LNCaP, CWR22Rv1, PC3, and DU145 were examined for REST protein by immunoblotting ( A ) and REST mRNA by RT-qPCR ( B ). n = 3. ( C ) Immunoblotting of protein expression for REST, NE marker SYP, and EMT and stemness markers as indicated in LNCaP and CWR22Rv1 cells. n = 3. ( D ) mRNA levels of NE and EMT markers and CD44 in LNCaP and CWR22Rv1 cells. n = 2. ( E and F ) Immunofluorescence micrographs ( E ) and flow cytometric analysis ( F ) of CD44 expression in LNCaP and CWR22Rv1 cells. n = 2. ( G and H ) LNCaP and CWR22Rv1 cells were seeded in transwell (1 x 10 4 ) or cultured in defined serum-free sphere medium with growth factors (2 x 10 4 ) and measured at 3 and 14 days, respectively. The number of migrated cells was quantified by the average from 5 microscopic fields ( G ). The number of spheres was calculated ( H ). Representative images (left) and quantification (right) in both PCa cell lines. n = 3. Data represent means +- SD. ** p < 0.01, *** p < 0.001 by Student's-t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Short-term overexpression of REST inhibits EMT and stemness properties of CWR22Rv1 cells. ( A ) Immunoblotting of REST, NE marker SYP, and EMT and stemness markers in CWR22Rv1-TR-REST cells receiving 1 mug/ml Dox for 3 days (P1) compared to no Dox control. n = 3. ( B ) CWR22Rv1-TR-REST cells treated as described in ( A ) were seeded on the transwell membrane and after three days the cells were stained with Hoechst. Representative images (left) and quantification (right) of migrated CWR22Rv1 cells. n = 3. ( C and D ) Immunofluorescent micrographs ( C ) and flow cytometric analysis ( D ) of CD44 expression in CWR22Rv1 cells treated as described in ( A ). n = 2. ( E ) CWR22Rv1-TR-REST cells treated as described in ( A ) were then cultured in defined serum-free sphere medium for 14 days. Representative images (left) and quantification (right) of sphere formation in CWR22Rv1 cells. n = 3. Data represent means +- SD. *** p < 0.001 by Student's-t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Long-term overexpression of REST inhibits EMT and stemness properties of CWR22Rv1 cells. ( A ) Immunoblotting of REST, NE marker SYP, and EMT and stemness markers in CWR22Rv1-TR-REST cells receiving 1 mug/ml Dox for 45 days (P15) compared to no Dox control. n = 2. ( B ) CWR22Rv1-TR-REST cells treated as described in ( A ) were seeded on the transwell membrane and after three days the cells were stained with Hoechst. Representative images (left) and quantification (right) of migrated CWR22Rv1 cells. n = 3. ( C and D ) Immunofluorescent micrographs ( C ) and flow cytometric analysis ( D ) of CD44 expression in CWR22Rv1 cells treated as described in ( A ). n = 2. ( E ) CWR22Rv1-TR-REST cells treated as described in ( A ) were then cultured in defined serum-free sphere medium for 14 days. Representative images (left) and quantification (right) of sphere formation in CWR22Rv1 cells. n = 3. Data represent means +- SD. *** p < 0.001 by Student's-t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of CD44 was performed on HAP1 cell lysates. Antibody-bead conjugates were prepared by adding 1 µg of CD44 recombinant monoclonal antibody (Product # MA5-13890) with 30 µL of protein G-Sepharose beads and rocked overnight at 4°C. 1 mg of lysate was incubated with an antibody-bead conjugate for 2 hours at 4°C. Following centrifugation and multiple washes, 10% starting material (SM), 10% unbound fraction (UB) and immunoprecipitated fraction (IP) were processed for immunoblot using a different CD44 monoclonal antibody. Ponceau stained transfer of blot is shown. Data courtesy of YCharOS Inc., an open science company with the mission of characterizing commercially available antibodies using knockout validation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 CD9 + PMPs on BCCs A. Confocal images were taken every 25 min of live CD9-GFP-transfected MDA cells cultured with DiD-labeled MSCs. Retraction of magnupodium (asterisk) occurs on a 1 h-time scale. Arrows, branched CD9 + filopodia detached from cells and degraded over time. B . C . Fixed, permeabilized MDA cells were stained with anti-CD9 Ab followed by Cy5-conjugated secondary Ab. Images are maximum intensity projections (MIPs) of confocal z-stacks. Representative z-optical sections taken at different heights from the coverslip shown to the right of the MIP. Arrow, branched CD9 + filopodia; white asterisk, lamellipodium at the tip of magnupodium (B). CD9 + PMPs such as a lamellipodium (arrow) at the tip of magnupodium and short filopodia (arrowheads) promote the intercellular interaction between two MDA cells. High power views of images of outlined areas are shown (C). D . CD9-GFP-transfected MDA cells stained with anti-CD44 Ab followed by Cy5-conjugated secondary Ab. Image is representative z-optical section of microvilli. Insets show GFP and Cy5 channels. E - G . CD9 + PMPs on MDA-CD9-GFP, MA-11-CD9-GFP, and MCF-7-CD9-GFP cells. Branched CD9 + filopodia (arrowhead) are emerging from MDA cells over long distance (see magnification of the outlined area) (E). Short CD9 + filopodia (arrowhead) are emerging from MA-11 (F) and MCF-7 (G). Note numerous CD9 + long microvilli in MA-11 cells and large dorsal ruffles (intense green areas) in MCF-7 cells (F, G, respectively, s
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Cell cycle is accelerated and the apoptosis rate is suppressed by CHIR and PD in HMLE cells. (A) Cell cycle distribution and apoptosis rate in HMLE cells following treatment with CHIR and PD were determined using flow cytometry analysis. (B) Flow cytometry analysis of CD44 and CD24 for control cells, and cells treated with CHIR and PD. All assays were performed in duplicates and data are presented as the mean +- standard deviation. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Immunohistochemical double-staining of human breast tumors for CD44 and CD24. CD44 is stained with Permanent Red and CD24 with diaminobenzidene (DAB). Magnification x 20. (a) A tumor positive for both CD44 + /CD24 - (white arrow) and CD44 - /CD24 + (black arrow) cancer cells, although the predominant phenotype is CD44 - /CD24 - . (b) Almost all cells in this tumor are CD44 + /CD24 - . No CD24 staining is seen. (c) A tumor with predominantly CD44 + /CD24 - cells. A few CD44 + /CD24 + cells are also present (black arrow). (d) A tumor positive for the CD44 - /CD24 + phenotype. No CD44 staining is present.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 The in vivo invasion analysis using the chicken chorioallantoic membrane (CAM) assay. ( a - f ) CD44 stained OV-90 cells transfected with ( a ) empty vector (pcDNA3.1(+)) and overexpression constructs, ( b ) PIWIL1 , ( c ) P117 , ( d ) PIWIL2 , ( e ) PL2L60 and ( f ) MAEL . CAMs were counterstained with haematoxylin. Invasion was quantified by the number of OV-90 cells that successfully breached the ECT and beyond (* marked next to breached area). CM = cancer cells mixed with matrigel, ECT = ectoderm, MES = mesoderm, END = endoderm. Scale bar = 100 um. ( g ) Significantly more CAM area was invaded by P117 and PL2L60 overexpressing OV-90 cells. Both mutants were more invasive than its respective wildtype form where only overexpression of P117 was significant. n = 4-18 per construct transfected and carried out in biological duplicates. Statistical tests Mann-Whitney U (non-normal distribution) or unpaired t -test (normal distribution) were used accordingly. * p < 0.05; *** p < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 TRIM59 nuclear translocation is required for GSC tumorigenicity. a Effects of EGFR inhibitor erlotinib or CDK5 inhibitor Roscovitine treatment on p-EGFR, p-CDK5, p-STAT3, and mH2A1 protein levels in GSC R39 and R83 cells. Arrows, EGFR. Arrow heads, EGFRvIII. b qRT-PCR analyses of erlotinib or Roscovitine treatment on STAT3-targeted gene PIM1 mRNA expression. c Limiting dilution glioma sphere-forming assays in GSC R39 and R83 cells with erlotinib or Roscovitine treatment. d - f Effects of TRIM59 shRNA knockdown and re-expression of shRNA-resistant TRIM59 WT, S308A, or S308D mutant on TRIM59, mH2A1 protein levels ( d ), p-STAT3 ( d ), STAT3-targeted gene PIM1 expression ( e ), and glioma sphere formation ( f ) in GSC R39 and R83 cells. g , h Representative bioluminescence images of tumor growth ( g ) and quantification of bioluminescence ( h ). n = 5. i Representative images of IHC analysis of indicated protein expression in GSC R39 xenograft tumors. Scale bar, 50 mum. j , k Effects of TRIM59 shRNA knockdown and re-expression of shRNA-resistant TRIM59 WT, S308A, or S308D mutant on TRIM59, mH2A1 protein levels ( j ), p-STAT3 ( j ), and STAT3-targeted gene PIM1 expression ( k ) in GSC R39 xenograft tumors. Data are representative of three independent experiments with similar results. Data were expressed as means +- SD. * P < 0.05, ** P < 0.01, by two-tailed Student's t test. Source data are provided as a Source Data file
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Endogenous stem cells recruitment by HCLS. a1 Schematic depiction of in vitro BMSCs recruitment by incubating various scaffolds in a total rabbit cranial bone marrow for 48 h. a2 CD44 immunofluorescent staining imaged by CLSM. a3 Schematic depiction of transwell assay in studying BMSCs migration to various scaffolds. a4 Quantification of migrated BMSCs by dissolving crystal violet and spectrophotometrically measured at 573 nm, the optical density (OD) resulted was normalized by control. BMSCs alone was used as control (** p = 0.0028, *** p = 0.0001, *** p = 0.0002, *** p = 6.7070 x 10 -5 ). n = 3 biologically independent replicates. b SEM images of endogenous cells on the surface of various scaffolds. c CLSM images (rhodamine-phalloidin/DAPI staining) of endogenous cells on the surface of various scaffolds. d CLSM images of CD44/F-actin/DAPI staining of various scaffolds. e1 Confocal quantitative images (CD44/DAPI staining) of ESCs in scaffolds. e2 High-magnification images (CD44/DAPI staining) of ESCs in scaffolds. e3 Quantitative analysis of the total number of cells in scaffolds (* p = 0.0470, ** p = 0.0023). e4 Quantitative analysis of the number of ESCs in scaffolds (*** p = 0.0008, *** p = 0.0003). n = 3 biologically independent replicates. f1 - 3 BMP-2 and Runx2 immunofluorescence staining at one week of different treatments and semiquantitative analysis ( f1 : *** p = 0.0009, *** p = 5.5298 x 10 -5 ; f2 : ** p = 0.0078, *** p = 5.8847 x 10 -5 ). g1 CLSM images of CD31
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
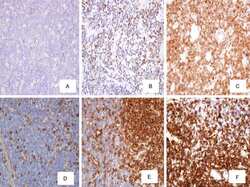
- Figure 3 Immunohistochemistry in breast tumor sections from triple-negative breast cancer (TNBC) patients for LSD1 and the stemness marker CD44. ( A - D ) Representative images from four different tumors (cases 4, 8, 9, and 10 in Table S1 ) showing high enrichment in CD44 + cells and high co-expression of CD44 and LSD1. Magnification 200x. ( E ) Very high (-100%) and ( F ) lower (-40%) concomitant expression of LSD1 and CD44 in neoplastic cells. ( G ) Strong and ( H ) moderate nuclear staining for LSD1 in neoplastic cells. Magnification 600x. ( I , J ) Non-neoplastic cells of the adjacent breast parenchyma with weak nuclear LSD1 staining from two different tumors (magnification 400x). The cell surface antigen CD44 exhibits membranous staining (chromogenic substrate: magenta) and LSD1 is nuclear (chromogenic substrate: DAB).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
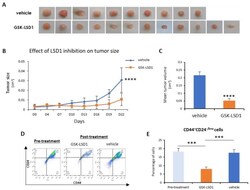
- Figure 5 LSD1 inhibition reduced tumor growth and targeted the CSC subpopulation in vivo. MDA-MB 468 breast cancer cells were injected orthotopically in mice (into two fat pads/mouse). Treatment with the GSK-LSD1 inhibitor ( n = 6) or with the vehicle control ( n = 5) started when the tumors were detectable by palpation and lasted for 22 days, when the tumors were removed and measured. ( A ) Representative images of the tumors excised from mice treated with vehicle or GSK-LSD1 inhibitor. ( B ) Graphical representation of changes in tumor size over the course of the experiment. ( C ) Graphical representation of the effects LSD1 inhibition on the mean tumor volume as measured at the end of the experiment. ( D ) The FACS analysis of dissociated tumor cells isolated pre-treatment or post-treatment with GSK-LSD1 or vehicle. Single cells were stained with antibodies against the CD44 and CD24 cell surface proteins. ( E ) Quantification of FACS analysis presented in ( D ). Error bars represent SEM. *** p < 0.001, **** p < 0.0001.
 Explore
Explore Validate
Validate Learn
Learn