Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-11921 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- L-Plastin Monoclonal Antibody (LPL4A.1)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- MA5-11921 targets Plastin-L in IF, IP, and WB applications and shows reactivity with Human samples.
- Antibody clone number
- LPL4A.1
- Concentration
- 0.2 mg/mL
Submitted references Active hematopoiesis triggers exosomal release of PRDX2 that promotes osteoclast formation.
Exosomal Release of L-Plastin by Breast Cancer Cells Facilitates Metastatic Bone Osteolysis.
The pro-oxidative drug WF-10 inhibits serial killing by primary human cytotoxic T-cells.
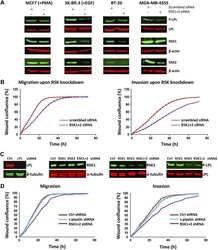
L-plastin Ser5 phosphorylation in breast cancer cells and in vitro is mediated by RSK downstream of the ERK/MAPK pathway.
Fascin Rigidity and L-plastin Flexibility Cooperate in Cancer Cell Invadopodia and Filopodia.
Metastasis of prostate cancer and melanoma cells in a preclinical in vivo mouse model is enhanced by L-plastin expression and phosphorylation.
L-plastin nanobodies perturb matrix degradation, podosome formation, stability and lifetime in THP-1 macrophages.
Nanobody-induced perturbation of LFA-1/L-plastin phosphorylation impairs MTOC docking, immune synapse formation and T cell activation.
Proteomic analysis of mononuclear cells of patients with minimal-change nephrotic syndrome of childhood.
Analysis of the expression of biomarkers in urinary bladder cancer using a tissue microarray.
Phosphorylation of ectopically expressed L-plastin enhances invasiveness of human melanoma cells.
Antileukoproteinase: modulation of neutrophil function and therapeutic effects on anti-type II collagen antibody-induced arthritis.
Sadvakassova G, Tiedemann K, Steer KJD, Mikolajewicz N, Stavnichuk M, In-Kyung Lee I, Sabirova Z, Schranzhofer M, Komarova SV
Physiological reports 2021 Feb;9(3):e14745
Physiological reports 2021 Feb;9(3):e14745
Exosomal Release of L-Plastin by Breast Cancer Cells Facilitates Metastatic Bone Osteolysis.
Tiedemann K, Sadvakassova G, Mikolajewicz N, Juhas M, Sabirova Z, Tabariès S, Gettemans J, Siegel PM, Komarova SV
Translational oncology 2019 Mar;12(3):462-474
Translational oncology 2019 Mar;12(3):462-474
The pro-oxidative drug WF-10 inhibits serial killing by primary human cytotoxic T-cells.
Wabnitz GH, Balta E, Schindler S, Kirchgessner H, Jahraus B, Meuer S, Samstag Y
Cell death discovery 2016;2:16057
Cell death discovery 2016;2:16057
L-plastin Ser5 phosphorylation in breast cancer cells and in vitro is mediated by RSK downstream of the ERK/MAPK pathway.
Lommel MJ, Trairatphisan P, Gäbler K, Laurini C, Muller A, Kaoma T, Vallar L, Sauter T, Schaffner-Reckinger E
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Mar;30(3):1218-33
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2016 Mar;30(3):1218-33
Fascin Rigidity and L-plastin Flexibility Cooperate in Cancer Cell Invadopodia and Filopodia.
Van Audenhove I, Denert M, Boucherie C, Pieters L, Cornelissen M, Gettemans J
The Journal of biological chemistry 2016 Apr 22;291(17):9148-60
The Journal of biological chemistry 2016 Apr 22;291(17):9148-60
Metastasis of prostate cancer and melanoma cells in a preclinical in vivo mouse model is enhanced by L-plastin expression and phosphorylation.
Riplinger SM, Wabnitz GH, Kirchgessner H, Jahraus B, Lasitschka F, Schulte B, van der Pluijm G, van der Horst G, Hämmerling GJ, Nakchbandi I, Samstag Y
Molecular cancer 2014 Jan 18;13:10
Molecular cancer 2014 Jan 18;13:10
L-plastin nanobodies perturb matrix degradation, podosome formation, stability and lifetime in THP-1 macrophages.
De Clercq S, Boucherie C, Vandekerckhove J, Gettemans J, Guillabert A
PloS one 2013;8(11):e78108
PloS one 2013;8(11):e78108
Nanobody-induced perturbation of LFA-1/L-plastin phosphorylation impairs MTOC docking, immune synapse formation and T cell activation.
De Clercq S, Zwaenepoel O, Martens E, Vandekerckhove J, Guillabert A, Gettemans J
Cellular and molecular life sciences : CMLS 2013 Mar;70(5):909-22
Cellular and molecular life sciences : CMLS 2013 Mar;70(5):909-22
Proteomic analysis of mononuclear cells of patients with minimal-change nephrotic syndrome of childhood.
González E, Neuhaus T, Kemper MJ, Girardin E
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2009 Jan;24(1):149-55
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2009 Jan;24(1):149-55
Analysis of the expression of biomarkers in urinary bladder cancer using a tissue microarray.
Harris LD, De La Cerda J, Tuziak T, Rosen D, Xiao L, Shen Y, Sabichi AL, Czerniak B, Grossman HB
Molecular carcinogenesis 2008 Sep;47(9):678-85
Molecular carcinogenesis 2008 Sep;47(9):678-85
Phosphorylation of ectopically expressed L-plastin enhances invasiveness of human melanoma cells.
Klemke M, Rafael MT, Wabnitz GH, Weschenfelder T, Konstandin MH, Garbi N, Autschbach F, Hartschuh W, Samstag Y
International journal of cancer 2007 Jun 15;120(12):2590-9
International journal of cancer 2007 Jun 15;120(12):2590-9
Antileukoproteinase: modulation of neutrophil function and therapeutic effects on anti-type II collagen antibody-induced arthritis.
Sehnert B, Cavcic A, Böhm B, Kalden JR, Nandakumar KS, Holmdahl R, Burkhardt H
Arthritis and rheumatism 2004 Jul;50(7):2347-59
Arthritis and rheumatism 2004 Jul;50(7):2347-59
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of Plastin-L using Plastin-L Monoclonal Antibody (Product # MA5-11921) on Jurkat Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane enriched extracts (30 µg lysate) of Jurkat (Lane 1), THP-1 (Lane 2), IM-9 (Lane 3), Raji (Lane 4), Ramos (Lane 5), K-562 (Lane 6), A-431 (Lane 7), KARPAS 299 (Lane 8) and RAW 264.7 (Lane 9). The blot was probed with Anti-L-Plastin Mouse Monoclonal Antibody (Product # MA5-11921, 2 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.4 µg/mL, 1:2500 dilution). A 70 kDa band corresponding to L-Plastin was observed across the cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® ; 10% Bis-Tris gel (Product # NP0302BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
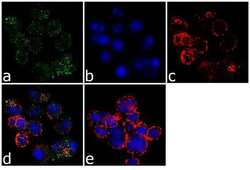
- Experimental details
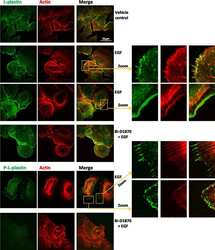
- Immunofluorescence analysis of Plastin-L was performed using 70% confluent log phase Jurkat cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with L-Plastin (LPL4A.1) Mouse Monoclonal Antibody (Product # MA5-11921) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
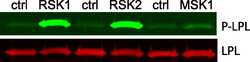
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
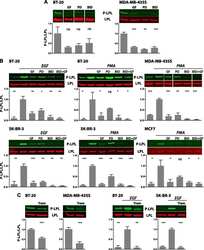
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
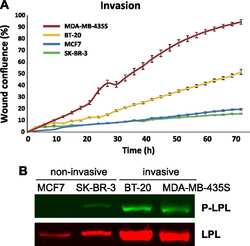
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
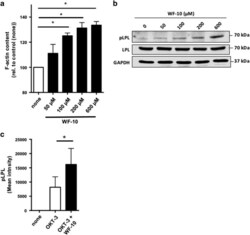
- Experimental details
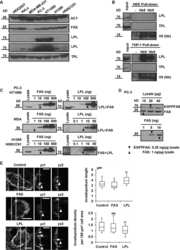
- Figure 5 WF-10 induced L-plastin phosphorylation. ( a ) F-actin content in CTLs was analyzed by flow cytometry. CTLs were incubated with WF-10 for 2 h and stained with SiR actin ( n =3; S.E.M.; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
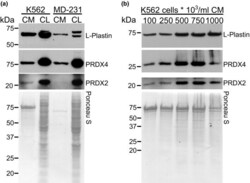
- Experimental details
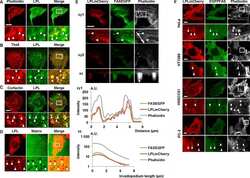
- FIGURE 4 Identification of L-plastin and PRDXs in K562 cells. Conditioned media (CM) was collected and TCA precipitated, and the protein was extracted from cell lysates (CL) of K562 and MDA-MB-231 cells (MD-231). a) Intracellular and released L-plastin, PRDX2 and PRDX4 in K562 and MDA-MB-231 cells were assessed by immunoblotting. b) K562 cells were cultured at different cell densities (10 5 -10 6 cells/ml) and the levels of L-plastin, PRDX2 and PRDX4 in CM were assessed by immunoblotting. Ponceau stain (lower gels) was used as loading control. Shown are representative immunoblots from one out five independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
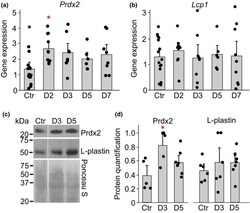
- Experimental details
- FIGURE 5 Acute anemia in mice increase the level of Prdx2 in bone marrow. Acute anemia was induced in female C57BL/6 mice by collecting 10% of total blood volume from saphenous vein, after which groups of animals were sacrificed at days 2, 3, 5 and 7. a-b) RNA was isolated from bone marrow and gene expression for Prdx2 and Lcp1 was assessed. c-d) Protein levels of Prdx2 and Lcp1 in bone marrow were assessed by immunoblotting. c) Representative immunoblots for Prdx2 ( top ), Lcp1 ( middle ) and Ponceau S-stained gel ( bottom ) used for normalization. d) Average protein expression of Prdx2 and L-plastin. For each immunoblot, protein signal was first normalized to ponceau staining, and then to the maximum signal within the immunoblot. Data are means +-SEM, N = 5-15 mice per condition, * p < 0.05 by one-way ANOVA with Bonferroni post-test, red : compared to control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 6 Exosomes from K562 cells induce osteoclast differentiation. a) RAW 264.7 cells were primed with RANKL (50 ng/ml) for 2 days and then cultured for an additional 2 days without RANKL treatment (negative control, NC), with RANKL (50 ng/ml, positive control, PC) or 10% CM from K562 cells +/- exosome inhibitor GW4869 (10 uM). N = 3. b) K562 cells were cultured for 24 h then exosomes were purified, and the distribution of particle sizes was analyzed by Nano sight. c) Representative transmission electron microscopy image of exosomes purified from K562 CM. d-h) RAW 264.7 cells were primed with RANKL (50 ng/ml) for 2 days and then cultured for an additional 2 days without RANKL treatment (negative control, NC), with RANKL (50 ng/ml, positive control, PC) or with purified exosomes (Exo, 10 ul) from K562 cells. d) Representative images of TRAP-stained osteoclasts formed in RAW cultures on day 5. Scale bar applies to all images. e) Average osteoclast number; f) average area per osteoclast; g) number of nuclei per osteoclast; h) average area/nucleus. N = 37-50 osteoclasts/condition; i) Immunoblotting for exosomal markers TFR-2 and TSG101, L-plastin, PRDX4 and PRDX2 in K562 CM and purified exosomes (Exo). Data are means +-SEM, * p < 0.05, ** p < 0.01 and *** p < 0.001 assessed by Student's t-test; red : compared to negative control, blue : compared to -GW4869 or positive control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
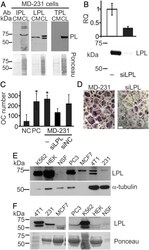
- Experimental details
- Figure 1 L-plastin is an osteoclastogenic factor secreted from MDA-MB-231 breast cancer cells. (A) Immunoblotting for I, L, and T-plastin (IPL, LPL, and TPL) in MDA-MB-231 cell lysates (CL) and conditioned medium (CM). Equal loading was visualized by Ponceau staining (lower gels). (B-D) L-plastin was transiently depleted in MDA-MB-231 cells (MD-231) using siRNA for L-plastin specific oligos (siLPL) or negative control (siNC). (B) L-plastin mRNA (top) and protein (bottom) expression in parental (-) and siLPL cells. (C, D) RAW 264.7 cells were primed with RANKL (50 ng/ml) for 2 days and then cultured for additional 2 days without treatment (negative control, NC), with RANKL (50 ng/ml, PC), or with 10% CM from siNC or siLPL MDA-MB-231. Average numbers (C) and representative images (D) of osteoclasts (OC) formed in indicated conditions. Scale bar of 100 mum applies to both images. Data are means +- SD, N = 2. (E, F) Protein expressions of L-plastin in whole cell lysates (E) and CM (F) in human erythroleukemia cells K562, human embryonic kidney cells 293 (HEK), normal skin fibroblasts (NSF), prostate carcinoma cells (PC3), human breast carcinoma cells MCF7 and MDA-MB-231 (231), and mouse breast carcinoma cells (4T1) were determined using immunoblotting. Protein loading was assessed using alpha-tubulin for cell lysates (E) and Ponceau stain for CM (F). Figure 1
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot