Antibody data
- Antibody Data
- Antigen structure
- References [41]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [2]
- Immunohistochemistry [2]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-16409 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Vimentin Monoclonal Antibody (SP20)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- Predicted to react with Rat, Hamster, Cow, and Xenopus laevis based on sequence homology. This antibody is not suitable for mouse tissues. Recommended Postive Controls - WB: U-2 OS, Hu tonsil and HeLa cell lysates. Flow: HeLa cells. ICC/IF: HAP1-VIM cells, human limbal epithelial cells. IHC-P: Human breast cancer and melanoma tissue. IHC-Fr: Colorectal cancer tissue.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- SP20
- Vial size
- 500 µL
- Concentration
- 0.006 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references A surrogate marker for very early-stage tau pathology is detectable by molecular magnetic resonance imaging.
An epithelial-mesenchymal plasticity signature identifies two novel LncRNAs with the opposite regulation.
Poria Acid, Triterpenoids Extracted from Poria cocos, Inhibits the Invasion and Metastasis of Gastric Cancer Cells.
TRPM7 Modulates Human Pancreatic Stellate Cell Activation.
Betulonic Acid, as One of the Active Components of the Celastrus orbiculatus Extract, Inhibits the Invasion and Metastasis of Gastric Cancer Cells by Mediating Cytoskeleton Rearrangement In Vitro.
Downregulation of MMP-9 Enhances the Anti-Migratory Effect of Cyclophosphamide in MDA-MB-231 and MCF-7 Breast Cancer Cell Lines.
Chondroitin polymerizing factor promotes breast carcinoma cell proliferation, invasion and migration and affects expression of epithelial-mesenchymal transition-related markers.
3D bioprinting of high cell-density heterogeneous tissue models through spheroid fusion within self-healing hydrogels.
Quiescin Sulfhydryl Oxidase 1 Regulates the Proliferation, Migration and Invasion of Human Glioblastoma Cells via PI3K/Akt Pathway.
Human Cardiac Organoids for Modeling Genetic Cardiomyopathy.
p120-catenin-dependent collective brain infiltration by glioma cell networks.
The vacuolated morphology of chordoma cells is dependent on cytokeratin intermediate filaments.
MicroRNAs as Urinary Biomarker for Oncocytoma.
Legumain Regulates Differentiation Fate of Human Bone Marrow Stromal Cells and Is Altered in Postmenopausal Osteoporosis.
Bone Formation by Sheep Stem Cells in an Ectopic Mouse Model: Comparison of Adipose and Bone Marrow Derived Cells and Identification of Donor-Derived Bone by Antibody Staining.
Mutant PIK3CA Induces EMT in a Cell Type Specific Manner.
CD146/MCAM defines functionality of human bone marrow stromal stem cell populations.
Cell type of origin as well as genetic alterations contribute to breast cancer phenotypes.
A systematic analysis of commonly used antibodies in cancer diagnostics.
Donor cornea transfer from Optisol GS to organ culture storage: a two-step procedure to increase donor tissue lifespan.
Pancreatic cancer-associated stellate cells promote differentiation of myeloid-derived suppressor cells in a STAT3-dependent manner.
Molecular Magnetic Resonance Imaging of Tumors with a PTPµ Targeted Contrast Agent.
Molecular Magnetic Resonance Imaging of Tumors with a PTPµ Targeted Contrast Agent.
Medulloblastoma with focal divergent/teratoid differentiation.
Multiple types of data are required to identify the mechanisms influencing the spatial expansion of melanoma cell colonies.
Podoplanin expressing cancer associated fibroblasts are associated with unfavourable prognosis in adenocarcinoma of the esophagus.
Derivation of myoepithelial progenitor cells from bipotent mammary stem/progenitor cells.
Residual tumor cells that drive disease relapse after chemotherapy do not have enhanced tumor initiating capacity.
Immunohistochemical characterization of nonhuman primate ovarian sex cord-stromal tumors.
Knock-down of plasminogen-activator inhibitor-1 enhances expression of E-cadherin and promotes epithelial differentiation of human pancreatic adenocarcinoma cells.
Cadherin-11, a marker of the mesenchymal phenotype, regulates glioblastoma cell migration and survival in vivo.
Mucinous epithelial lesions in endometrial curettage material: a diagnostic challenge.
Gonadoblastoma in the ovaries of a lesser galago (Galago senegalensis braccatus).
Sporadic haemangioblastoma of the kidney with rhabdoid features and focal CD10 expression: report of a case and literature review.
Cotyledonoid leiomyoma of uterus: a case report.
IL-6 promotes prostate tumorigenesis and progression through autocrine cross-activation of IGF-IR.
Primary myoepithelial carcinoma of the larynx: case report and review of the literature.
A novel assessment of the quality of immunohistostaining overcomes the limitations of current methods.
Basal-HER2 phenotype shows poorer survival than basal-like phenotype in hormone receptor-negative invasive breast cancers.
Laparoscopic resection of urinary bladder paraganglioma: a case report.
Nek8 mutation causes overexpression of galectin-1, sorcin, and vimentin and accumulation of the major urinary protein in renal cysts of jck mice.
Parekh P, Mu Q, Badachhape A, Bhavane R, Srivastava M, Devkota L, Sun X, Bhandari P, Eriksen JL, Tanifum E, Ghaghada K, Annapragada A
Theranostics 2022;12(12):5504-5521
Theranostics 2022;12(12):5504-5521
An epithelial-mesenchymal plasticity signature identifies two novel LncRNAs with the opposite regulation.
Kumar P, Khadirnaikar S, Bhandari N, Chatterjee A, Shukla S
Frontiers in cell and developmental biology 2022;10:885785
Frontiers in cell and developmental biology 2022;10:885785
Poria Acid, Triterpenoids Extracted from Poria cocos, Inhibits the Invasion and Metastasis of Gastric Cancer Cells.
Wang H, Luo Y, Chu Z, Ni T, Ou S, Dai X, Zhang X, Liu Y
Molecules (Basel, Switzerland) 2022 Jun 6;27(11)
Molecules (Basel, Switzerland) 2022 Jun 6;27(11)
TRPM7 Modulates Human Pancreatic Stellate Cell Activation.
Auwercx J, Kischel P, Lefebvre T, Jonckheere N, Vanlaeys A, Guénin S, Radoslavova S, Van Seuningen I, Ouadid-Ahidouch H, Kocher HM, Dhennin-Duthille I, Gautier M
Cells 2022 Jul 21;11(14)
Cells 2022 Jul 21;11(14)
Betulonic Acid, as One of the Active Components of the Celastrus orbiculatus Extract, Inhibits the Invasion and Metastasis of Gastric Cancer Cells by Mediating Cytoskeleton Rearrangement In Vitro.
Chu Z, Luo Y, Ni T, Zhu M, Feng X, Liu Y, Wang H
Molecules (Basel, Switzerland) 2022 Feb 2;27(3)
Molecules (Basel, Switzerland) 2022 Feb 2;27(3)
Downregulation of MMP-9 Enhances the Anti-Migratory Effect of Cyclophosphamide in MDA-MB-231 and MCF-7 Breast Cancer Cell Lines.
Izdebska M, Zielińska W, Krajewski A, Hałas-Wiśniewska M, Mikołajczyk K, Gagat M, Grzanka A
International journal of molecular sciences 2021 Nov 26;22(23)
International journal of molecular sciences 2021 Nov 26;22(23)
Chondroitin polymerizing factor promotes breast carcinoma cell proliferation, invasion and migration and affects expression of epithelial-mesenchymal transition-related markers.
Li Y, Gong H, Feng L, Mao D, Xiao Y, Wang Y, Huang L
FEBS open bio 2021 Feb;11(2):423-434
FEBS open bio 2021 Feb;11(2):423-434
3D bioprinting of high cell-density heterogeneous tissue models through spheroid fusion within self-healing hydrogels.
Daly AC, Davidson MD, Burdick JA
Nature communications 2021 Feb 2;12(1):753
Nature communications 2021 Feb 2;12(1):753
Quiescin Sulfhydryl Oxidase 1 Regulates the Proliferation, Migration and Invasion of Human Glioblastoma Cells via PI3K/Akt Pathway.
Geng Y, Xu C, Wang Y, Zhang L
OncoTargets and therapy 2020;13:5721-5729
OncoTargets and therapy 2020;13:5721-5729
Human Cardiac Organoids for Modeling Genetic Cardiomyopathy.
Filippo Buono M, von Boehmer L, Strang J, Hoerstrup SP, Emmert MY, Nugraha B
Cells 2020 Jul 20;9(7)
Cells 2020 Jul 20;9(7)
p120-catenin-dependent collective brain infiltration by glioma cell networks.
Gritsenko PG, Atlasy N, Dieteren CEJ, Navis AC, Venhuizen JH, Veelken C, Schubert D, Acker-Palmer A, Westerman BA, Wurdinger T, Leenders W, Wesseling P, Stunnenberg HG, Friedl P
Nature cell biology 2020 Jan;22(1):97-107
Nature cell biology 2020 Jan;22(1):97-107
The vacuolated morphology of chordoma cells is dependent on cytokeratin intermediate filaments.
Resutek L, Hsieh AH
Journal of cellular physiology 2019 Apr;234(4):3458-3468
Journal of cellular physiology 2019 Apr;234(4):3458-3468
MicroRNAs as Urinary Biomarker for Oncocytoma.
von Brandenstein M, Schlosser M, Herden J, Heidenreich A, Störkel S, Fries JWU
Disease markers 2018;2018:6979073
Disease markers 2018;2018:6979073
Legumain Regulates Differentiation Fate of Human Bone Marrow Stromal Cells and Is Altered in Postmenopausal Osteoporosis.
Jafari A, Qanie D, Andersen TL, Zhang Y, Chen L, Postert B, Parsons S, Ditzel N, Khosla S, Johansen HT, Kjærsgaard-Andersen P, Delaisse JM, Abdallah BM, Hesselson D, Solberg R, Kassem M
Stem cell reports 2017 Feb 14;8(2):373-386
Stem cell reports 2017 Feb 14;8(2):373-386
Bone Formation by Sheep Stem Cells in an Ectopic Mouse Model: Comparison of Adipose and Bone Marrow Derived Cells and Identification of Donor-Derived Bone by Antibody Staining.
Kjærgaard K, Dreyer CH, Ditzel N, Andreasen CM, Chen L, Sheikh SP, Overgaard S, Ding M
Stem cells international 2016;2016:3846971
Stem cells international 2016;2016:3846971
Mutant PIK3CA Induces EMT in a Cell Type Specific Manner.
Bhagirath D, Zhao X, Mirza S, West WW, Band H, Band V
PloS one 2016;11(12):e0167064
PloS one 2016;11(12):e0167064
CD146/MCAM defines functionality of human bone marrow stromal stem cell populations.
Harkness L, Zaher W, Ditzel N, Isa A, Kassem M
Stem cell research & therapy 2016 Jan 11;7:4
Stem cell research & therapy 2016 Jan 11;7:4
Cell type of origin as well as genetic alterations contribute to breast cancer phenotypes.
Bhagirath D, Zhao X, West WW, Qiu F, Band H, Band V
Oncotarget 2015 Apr 20;6(11):9018-30
Oncotarget 2015 Apr 20;6(11):9018-30
A systematic analysis of commonly used antibodies in cancer diagnostics.
Gremel G, Bergman J, Djureinovic D, Edqvist PH, Maindad V, Bharambe BM, Khan WA, Navani S, Elebro J, Jirström K, Hellberg D, Uhlén M, Micke P, Pontén F
Histopathology 2014 Jan;64(2):293-305
Histopathology 2014 Jan;64(2):293-305
Donor cornea transfer from Optisol GS to organ culture storage: a two-step procedure to increase donor tissue lifespan.
Haug K, Azqueta A, Johnsen-Soriano S, Shahdadfar A, Drolsum LK, Moe MC, Røger MT, Romero FJ, Collins AR, Nicolaissen B
Acta ophthalmologica 2013 May;91(3):219-25
Acta ophthalmologica 2013 May;91(3):219-25
Pancreatic cancer-associated stellate cells promote differentiation of myeloid-derived suppressor cells in a STAT3-dependent manner.
Mace TA, Ameen Z, Collins A, Wojcik S, Mair M, Young GS, Fuchs JR, Eubank TD, Frankel WL, Bekaii-Saab T, Bloomston M, Lesinski GB
Cancer research 2013 May 15;73(10):3007-18
Cancer research 2013 May 15;73(10):3007-18
Molecular Magnetic Resonance Imaging of Tumors with a PTPµ Targeted Contrast Agent.
Burden-Gulley SM, Zhou Z, Craig SE, Lu ZR, Brady-Kalnay SM
Translational oncology 2013 Jun;6(3):329-37
Translational oncology 2013 Jun;6(3):329-37
Molecular Magnetic Resonance Imaging of Tumors with a PTPµ Targeted Contrast Agent.
Burden-Gulley SM, Zhou Z, Craig SE, Lu ZR, Brady-Kalnay SM
Translational oncology 2013 Jun;6(3):329-37
Translational oncology 2013 Jun;6(3):329-37
Medulloblastoma with focal divergent/teratoid differentiation.
Majumdar K, Tyagi I, Saran RK, Sakhuja P, Sharma A
Brain tumor pathology 2013 Jan;30(1):50-6
Brain tumor pathology 2013 Jan;30(1):50-6
Multiple types of data are required to identify the mechanisms influencing the spatial expansion of melanoma cell colonies.
Treloar KK, Simpson MJ, Haridas P, Manton KJ, Leavesley DI, McElwain DL, Baker RE
BMC systems biology 2013 Dec 12;7:137
BMC systems biology 2013 Dec 12;7:137
Podoplanin expressing cancer associated fibroblasts are associated with unfavourable prognosis in adenocarcinoma of the esophagus.
Schoppmann SF, Jesch B, Riegler MF, Maroske F, Schwameis K, Jomrich G, Birner P
Clinical & experimental metastasis 2013 Apr;30(4):441-6
Clinical & experimental metastasis 2013 Apr;30(4):441-6
Derivation of myoepithelial progenitor cells from bipotent mammary stem/progenitor cells.
Zhao X, Malhotra GK, Band H, Band V
PloS one 2012;7(4):e35338
PloS one 2012;7(4):e35338
Residual tumor cells that drive disease relapse after chemotherapy do not have enhanced tumor initiating capacity.
Hegde GV, de la Cruz C, Eastham-Anderson J, Zheng Y, Sweet-Cordero EA, Jackson EL
PloS one 2012;7(10):e45647
PloS one 2012;7(10):e45647
Immunohistochemical characterization of nonhuman primate ovarian sex cord-stromal tumors.
Durkes A, Garner M, Juan-Sallés C, Ramos-Vara J
Veterinary pathology 2012 Sep;49(5):834-8
Veterinary pathology 2012 Sep;49(5):834-8
Knock-down of plasminogen-activator inhibitor-1 enhances expression of E-cadherin and promotes epithelial differentiation of human pancreatic adenocarcinoma cells.
Lupu-Meiri M, Geras-Raaka E, Lupu R, Shapira H, Sandbank J, Segal L, Gershengorn MC, Oron Y
Journal of cellular physiology 2012 Nov;227(11):3621-8
Journal of cellular physiology 2012 Nov;227(11):3621-8
Cadherin-11, a marker of the mesenchymal phenotype, regulates glioblastoma cell migration and survival in vivo.
Kaur H, Phillips-Mason PJ, Burden-Gulley SM, Kerstetter-Fogle AE, Basilion JP, Sloan AE, Brady-Kalnay SM
Molecular cancer research : MCR 2012 Mar;10(3):293-304
Molecular cancer research : MCR 2012 Mar;10(3):293-304
Mucinous epithelial lesions in endometrial curettage material: a diagnostic challenge.
Pavlakis K, Vrekoussis T, Messini I, Voulgaris Z, Chrysanthakis D, Yiannou P, Stofas A, Panoskaltsis T
Applied immunohistochemistry & molecular morphology : AIMM 2012 Dec;20(6):607-13
Applied immunohistochemistry & molecular morphology : AIMM 2012 Dec;20(6):607-13
Gonadoblastoma in the ovaries of a lesser galago (Galago senegalensis braccatus).
Irizarry Rovira AR, Lynch S, David M, Ramos Vara JA
Journal of comparative pathology 2012 Aug-Oct;147(2-3):204-8
Journal of comparative pathology 2012 Aug-Oct;147(2-3):204-8
Sporadic haemangioblastoma of the kidney with rhabdoid features and focal CD10 expression: report of a case and literature review.
Yin WH, Li J, Chan JK
Diagnostic pathology 2012 Apr 12;7:39
Diagnostic pathology 2012 Apr 12;7:39
Cotyledonoid leiomyoma of uterus: a case report.
Ersöz S, Turgutalp H, Mungan S, Güvendı G, Güven S
Turk patoloji dergisi 2011;27(3):257-60
Turk patoloji dergisi 2011;27(3):257-60
IL-6 promotes prostate tumorigenesis and progression through autocrine cross-activation of IGF-IR.
Rojas A, Liu G, Coleman I, Nelson PS, Zhang M, Dash R, Fisher PB, Plymate SR, Wu JD
Oncogene 2011 May 19;30(20):2345-55
Oncogene 2011 May 19;30(20):2345-55
Primary myoepithelial carcinoma of the larynx: case report and review of the literature.
Yu G, Qu G, Kong L, Pan X, Wang W, Lv J
Pathology, research and practice 2011 Feb 15;207(2):127-30
Pathology, research and practice 2011 Feb 15;207(2):127-30
A novel assessment of the quality of immunohistostaining overcomes the limitations of current methods.
Eisenthal A, Trejo L, Shtabsky A, Bedny F, Brazowski E
Pathology, research and practice 2008;204(5):323-8
Pathology, research and practice 2008;204(5):323-8
Basal-HER2 phenotype shows poorer survival than basal-like phenotype in hormone receptor-negative invasive breast cancers.
Liu H, Fan Q, Zhang Z, Li X, Yu H, Meng F
Human pathology 2008 Feb;39(2):167-74
Human pathology 2008 Feb;39(2):167-74
Laparoscopic resection of urinary bladder paraganglioma: a case report.
Dilbaz B, Bayoglu Y, Oral S, Cavusoglu D, Uluoglu O, Dilbaz S
Surgical laparoscopy, endoscopy & percutaneous techniques 2006 Feb;16(1):58-61
Surgical laparoscopy, endoscopy & percutaneous techniques 2006 Feb;16(1):58-61
Nek8 mutation causes overexpression of galectin-1, sorcin, and vimentin and accumulation of the major urinary protein in renal cysts of jck mice.
Valkova N, Yunis R, Mak SK, Kang K, Kültz D
Molecular & cellular proteomics : MCP 2005 Jul;4(7):1009-18
Molecular & cellular proteomics : MCP 2005 Jul;4(7):1009-18
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on Whole cell extracts (30 µg lysate) of HeLa (Lane 1), A-431 (Lane 2), SHSY-5Y (Lane 3), HepG2 (Lane 4) and NIH/3T3 (Lane 5). The blot was probed with Anti-Vimentin Monoclonal Antibody (Product # MA5-16409, 1:2000 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/ml, 1:4000 dilution). A 54 kDa band corresponding to Vimentin was observed across all the human cell lines positive for Vimentin (Lanes 1 and 3) but not mouse cell line (Lane 5), while this band was absent in the cell lines which do not express Vimentin protein (Lanes 2 and 4).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
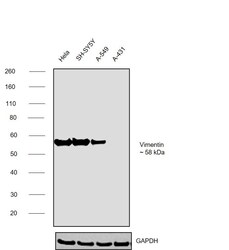
- Experimental details
- Western blot was performed using Anti-Vimentin Monoclonal Antibody (SP20) (Product # MA5-16409) and a ~58kDa band corresponding to vimentin was observed across cell lines tested except in A-431. Whole cell extracts (30 µg lysate) of HeLa (Lane 1), SH-SY5Y (Lane 2), A-549 (Lane 3), A-431 (Lane 4) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX), 12 well. Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:20000) using the iBright™ FL1500 Imaging System (Product # A44115). Chemiluminescent detection was performed using SuperSignal™ West Pico PLUS Chemiluminescent Substrate (Product # 34580).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Hela Cells using anti-Vimentin Monoclonal Antibody (Product # MA5-16409). The recommened dilution for this antibody in western blot applications is 1:100.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of vimentin was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Vimentin Monoclonal Antibody (SP20) (Product # MA5-16409) at 1:1000 in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with Hoechst 33342 (Product # H1399). F-actin (Panel c: Red) was stained with Alexa Fluor™ Plus 647 Phalloidin (Product # A30107, 1:2000 dilution). Panel d represents the merged image showing cytoplasmic and cytoskeletal localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 40X magnification in Cell Insight CX7 LZR High-Content Screening (HCS) Platform (Product # CX7A1110LZR).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Vimentin was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Vimentin Rabbit Monoclonal Antibody (Product # MA5-16409) at 1:200 dilution in 0.1% BSA and incubated overnight at 4 degree Celsius and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic, cytoskeletal and nuclear localization. Panel e represents negative control, A-431 cells. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of vimentin was performed using formalin-fixed paraffin-embedded human ovary adenocarcinoma tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a decloaking chamber at 110 degree celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Vimentin Monoclonal Antibody (SP20) (Product # MA5-16409) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree celsius in a humidified chamber. Detection was performed using Goat anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32731) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of Vimentin using anti-Vimentin Monoclonal Antibody (Product # MA5-16409) in Melanoma Cancer Tissue. The recommened dilution for this antibody in immunohistochemistry applications is 1:200.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Fabrication of cardiac spheroids for disease modeling applications. a (i) Development of healthy and scarred spheroids through mixing iPSC-derived cardiomyocytes (iPSC-CMs) with primary adult cardiac fibroblasts (CFs) at defined ratios of cell numbers (4:1 for healthy; 1:4 for scarred). Top: Images (cardiac troponin-T (cTnT) (red; iPSC-CMs); vimentin (green; CFs)) taken 3 days after cell seeding. Scalebar 50 um. Bottom: Immunofluorescence staining for alpha-actinin (green; sarcomeres) and cTnT (red; iPSC-CMs) in healthy and scarred spheroids at 3 days. Scalebar 10 um. (ii) Quantification of cellular composition through staining for cTnT (iPSC-CMs) and vimentin (CFs) ( n = 3 biologically independent samples, mean +- s.d, two-sided student t test, healthy - cTnT + vs. Vimentin + p = 5.6 x 10 -5 , scarred - cTnT + vs. Vimentin + p = 6.7 x 10 -4 ). b (i) Contraction profiles, (ii) contraction amplitude (a.u. absolute units), and (iii) peak-to-peak time (ms) of healthy and scarred cardiac spheroids at 3 days ( n = 3 biologically independent samples, mean +- s.d, two-sided student t test, (ii) p = 5.0 x 10 -4 ). c Quantification of calcium activation parameters from calcium mapping experiments in healthy and scarred spheroids at 3 days, including (i) calcium transient duration (ms), (ii) time-to-peak (ms), and (iii) calcium flux amplitude (F/Fo) ( n = 5 biologically independent samples, mean +- s.d, two-sided student t test, (i) p = 0.0014, (ii) p = 5.0 x 10 -6 , (iii) p = 2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 3D bioprinting cardiac microtissues for disease modeling applications. a (i) Schematic of 3D bioprinting of healthy and scarred cardiac microtissue rings and (ii) immunofluorescence staining for cTnT and vimentin in healthy and scarred cardiac microtissues after 5 days of fusion within the support hydrogel. Scalebar 100 um (insets 50 um). (iii) Immunofluorescence staining for alpha-actinin (green; sarcomeres) and cTnT (red; iPSC-CMs) and (iv) connexin-43 (green; gap junctions) and cTnT (red; iPSC-CMs), in healthy and scarred regions of microtissues after 5 days of fusion within the support hydrogel. Scalebar 10 um. b (i) Contraction profiles of healthy and scarred cardiac microtissues following removal from the support hydrogel after 5 days of culture. (ii) Contraction amplitude (a.u. absolute units) and (iii) Peak-to-peak time (ms) at 5 days ( n = 3 biologically independent samples, mean +- s.d, two-sided student t test, (ii) p = 0.061). c (i) Calcium mapping in healthy and scarred cardiac microtissues after 5 days culture, each image represents a 20 ms frame. Scalebar 100 um. (ii) Representative calcium traces from regions 1 and 2 (see methods) in healthy and scarred cardiac microtissues. Scalebars 0.5 DeltaF/F o (y), 500 ms (x). (iii) Activation maps of healthy and scarred cardiac microtissues, and activation delay (ms) (difference in activation time (ms) between regions 1 and 2) in healthy and scarred cardiac microtissues ( n = 3 biologically independent samples, m
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 Evaluating miRNA on the behavior of cardiac microtissues. a (i) Schematic of cholesterol modified miR302 (chol-miRNA 302 b/c) delivery to scarred cardiac spheroids for 0, 0-2, 0-4, and 0-7 days. (ii) Contraction amplitude (a.u) and (iii) peak-to-peak time (ms) within scarred spheroids measured after 2, 4, and 7 days for each treatment period ( n = 4, 6, 5, 5, 7, 5, 5, 5, 5 biologically independent samples (from left to right), mean +- s.d, one-way ANOVA, (ii) day 4: 0 vs. 0-4 days treatment p = 0.014, (ii) day 7: 0 vs. 0-7 days treatment p = 0.0045, (iii) day 4: 0 vs. 0-4 days treatment p = 1.6 x 10 -7 , (iii) day 7: 0 vs. 0-7 days treatment p = 4.1 x 10 -9 ). b (i) Immunofluorescence staining for cTnT (red; iPSC-CMs), vimentin (green; cardiac fibroblasts), and EdU (proliferation marker) in scarred spheroids at day 7 for each treatment condition. Top panel scalebar 50 um, and bottom panel scalebar 40 um. (ii) Quantification of cardiomyocyte proliferation (EdU + and cTnT + ) and (iii) fibroblast proliferation (EdU + and Vimentin + ) at day 7 for each treatment condition. ( n = 3, 4, 4, 4 biologically independent samples (from left to right), mean +- s.d, one-way ANOVA, 0 vs. 0-4 days treatment p = 0.044, 0 vs. 0-7 days treatment p = 0.026). Scalebar 50 um. c (i) Experimental outline where scarred microtissues are bioprinted in the support hydrogel as previously described, followed by 4 days treatment with miR302, and analysis compared to non-treated controls. (ii) Calci
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 The effect of MMP-9 downregulation on vimentin, N-cadherin and F-actin level. NT--non-transfected cells, T--cells transfected with MMP-9 siRNA, NT5 and T5 non-transfected and transfected cells treated with 5 mM cyclophosphamide for 24 h. ( A ) Immunofluorescent imaging of vimentin (red) and nuclei (blue), and relative fluorescence intensity of vimentin. ( B ) Immunofluorescent imaging of N-cadherin (red) and nuclei (blue), and relative fluorescence intensity of N-cadherin. ( C ), Immunofluorescent imaging of F-actin (green), and nuclei (blue), Bar = 50 mum. ( D ) The Western blot analysis of vimentin and N-cadherin and densitometric relative expression level of the Western blot experiments. Statistically significant differences were marked with '*' p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 ( A , B ) Cell EMT-related protein levels of E-cadherin, N-cadherin, Vimentin and beta-actin in AGS and ( C , D ) HGC-27 cells were investigated by Western blot. * p < 0.05, ** p < 0.01 and *** p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2 Pi3K pathway plays a major role in mesenchymal phenotype. (A) The correlation of EMT score with protein expression data of TCGA patients was calculated and plotted. The Yellow dots represent the positive correlation (>+0.2), and the green dots represent the negative correlation (
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry