Antibody data
- Antibody Data
- Antigen structure
- References [16]
- Comments [0]
- Validations
- Western blot [2]
- Immunohistochemistry [1]
- Flow cytometry [1]
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 35-8100 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- COX1 Monoclonal Antibody (COX 111)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- COX 111
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Endothelium-specific deletion of amyloid-β precursor protein exacerbates endothelial dysfunction induced by aging.
Mitochondrial AIF loss causes metabolic reprogramming, caspase-independent cell death blockade, embryonic lethality, and perinatal hydrocephalus.
Functional role of respiratory supercomplexes in mice: SCAF1 relevance and segmentation of the Q(pool).
Vascular phenotype of amyloid precursor protein-deficient mice.
Resveratrol Promotes Mitochondrial Biogenesis and Protects against Seizure-Induced Neuronal Cell Damage in the Hippocampus Following Status Epilepticus by Activation of the PGC-1α Signaling Pathway.
Mortaparib, a novel dual inhibitor of mortalin and PARP1, is a potential drug candidate for ovarian and cervical cancers.
Mechanism of super-assembly of respiratory complexes III and IV.
A cyclooxygenase-2-dependent prostaglandin E2 biosynthetic system in the Golgi apparatus.
Effect of ginger root on cyclooxygenase-1 and 15-hydroxyprostaglandin dehydrogenase expression in colonic mucosa of humans at normal and increased risk for colorectal cancer.
Roles of E-cadherin and cyclooxygenase enzymes in predicting different survival patterns of optimally cytoreduced serous ovarian cancer patients.
Modulation of CXCR3 ligand secretion by prostaglandin E2 and cyclooxygenase inhibitors in human breast cancer.
Asthmatics exhibit altered oxylipin profiles compared to healthy individuals after subway air exposure.
Amino acid-based ionic liquids: using XPS to probe the electronic environment via binding energies.
The Ras signaling inhibitor LOX-PP interacts with Hsp70 and c-Raf to reduce Erk activation and transformed phenotype of breast cancer cells.
Molecular typing of epithelial ovarian carcinomas using inflammatory markers.
Expression of protease-activated receptor-1 and -2 in orofacial granulomatosis.
d'Uscio LV, Katusic ZS
Aging 2021 Aug 12;13(15):19165-19185
Aging 2021 Aug 12;13(15):19165-19185
Mitochondrial AIF loss causes metabolic reprogramming, caspase-independent cell death blockade, embryonic lethality, and perinatal hydrocephalus.
Delavallée L, Mathiah N, Cabon L, Mazeraud A, Brunelle-Navas MN, Lerner LK, Tannoury M, Prola A, Moreno-Loshuertos R, Baritaud M, Vela L, Garbin K, Garnier D, Lemaire C, Langa-Vives F, Cohen-Salmon M, Fernández-Silva P, Chrétien F, Migeotte I, Susin SA
Molecular metabolism 2020 Oct;40:101027
Molecular metabolism 2020 Oct;40:101027
Functional role of respiratory supercomplexes in mice: SCAF1 relevance and segmentation of the Q(pool).
Calvo E, Cogliati S, Hernansanz-Agustín P, Loureiro-López M, Guarás A, Casuso RA, García-Marqués F, Acín-Pérez R, Martí-Mateos Y, Silla-Castro JC, Carro-Alvarellos M, Huertas JR, Vázquez J, Enríquez JA
Science advances 2020 Jun;6(26):eaba7509
Science advances 2020 Jun;6(26):eaba7509
Vascular phenotype of amyloid precursor protein-deficient mice.
d'Uscio LV, Katusic ZS
American journal of physiology. Heart and circulatory physiology 2019 Jun 1;316(6):H1297-H1308
American journal of physiology. Heart and circulatory physiology 2019 Jun 1;316(6):H1297-H1308
Resveratrol Promotes Mitochondrial Biogenesis and Protects against Seizure-Induced Neuronal Cell Damage in the Hippocampus Following Status Epilepticus by Activation of the PGC-1α Signaling Pathway.
Chuang YC, Chen SD, Hsu CY, Chen SF, Chen NC, Jou SB
International journal of molecular sciences 2019 Feb 25;20(4)
International journal of molecular sciences 2019 Feb 25;20(4)
Mortaparib, a novel dual inhibitor of mortalin and PARP1, is a potential drug candidate for ovarian and cervical cancers.
Putri JF, Bhargava P, Dhanjal JK, Yaguchi T, Sundar D, Kaul SC, Wadhwa R
Journal of experimental & clinical cancer research : CR 2019 Dec 19;38(1):499
Journal of experimental & clinical cancer research : CR 2019 Dec 19;38(1):499
Mechanism of super-assembly of respiratory complexes III and IV.
Cogliati S, Calvo E, Loureiro M, Guaras AM, Nieto-Arellano R, Garcia-Poyatos C, Ezkurdia I, Mercader N, Vázquez J, Enriquez JA
Nature 2016 Nov 24;539(7630):579-582
Nature 2016 Nov 24;539(7630):579-582
A cyclooxygenase-2-dependent prostaglandin E2 biosynthetic system in the Golgi apparatus.
Yuan C, Smith WL
The Journal of biological chemistry 2015 Feb 27;290(9):5606-20
The Journal of biological chemistry 2015 Feb 27;290(9):5606-20
Effect of ginger root on cyclooxygenase-1 and 15-hydroxyprostaglandin dehydrogenase expression in colonic mucosa of humans at normal and increased risk for colorectal cancer.
Jiang Y, Turgeon DK, Wright BD, Sidahmed E, Ruffin MT, Brenner DE, Sen A, Zick SM
European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP) 2013 Sep;22(5):455-60
European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP) 2013 Sep;22(5):455-60
Roles of E-cadherin and cyclooxygenase enzymes in predicting different survival patterns of optimally cytoreduced serous ovarian cancer patients.
Taşkin S, Dünder I, Erol E, Taşkin EA, Kiremitçi S, Öztuna D, Sertçelik A
Asian Pacific journal of cancer prevention : APJCP 2012;13(11):5715-9
Asian Pacific journal of cancer prevention : APJCP 2012;13(11):5715-9
Modulation of CXCR3 ligand secretion by prostaglandin E2 and cyclooxygenase inhibitors in human breast cancer.
Bronger H, Kraeft S, Schwarz-Boeger U, Cerny C, Stöckel A, Avril S, Kiechle M, Schmitt M
Breast cancer research : BCR 2012 Feb 14;14(1):R30
Breast cancer research : BCR 2012 Feb 14;14(1):R30
Asthmatics exhibit altered oxylipin profiles compared to healthy individuals after subway air exposure.
Lundström SL, Levänen B, Nording M, Klepczynska-Nyström A, Sköld M, Haeggström JZ, Grunewald J, Svartengren M, Hammock BD, Larsson BM, Eklund A, Wheelock ÅM, Wheelock CE
PloS one 2011;6(8):e23864
PloS one 2011;6(8):e23864
Amino acid-based ionic liquids: using XPS to probe the electronic environment via binding energies.
Hurisso BB, Lovelock KR, Licence P
Physical chemistry chemical physics : PCCP 2011 Oct 21;13(39):17737-48
Physical chemistry chemical physics : PCCP 2011 Oct 21;13(39):17737-48
The Ras signaling inhibitor LOX-PP interacts with Hsp70 and c-Raf to reduce Erk activation and transformed phenotype of breast cancer cells.
Sato S, Trackman PC, Mäki JM, Myllyharju J, Kirsch KH, Sonenshein GE
Molecular and cellular biology 2011 Jul;31(13):2683-95
Molecular and cellular biology 2011 Jul;31(13):2683-95
Molecular typing of epithelial ovarian carcinomas using inflammatory markers.
Ali-Fehmi R, Semaan A, Sethi S, Arabi H, Bandyopadhyay S, Hussein YR, Diamond MP, Saed G, Morris RT, Munkarah AR
Cancer 2011 Jan 15;117(2):301-9
Cancer 2011 Jan 15;117(2):301-9
Expression of protease-activated receptor-1 and -2 in orofacial granulomatosis.
Ketabchi S, Massi D, Ficarra G, Rubino I, Franchi A, Paglierani M, Simoni A, Capodiferro S, Favia G, Maiorano E, Tarantini F, Cirino G, Santucci M
Oral diseases 2007 Jul;13(4):419-25
Oral diseases 2007 Jul;13(4):419-25
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
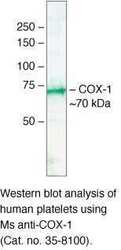
- Experimental details
- Western blot analysis of human platelets using Mouse anti-COX-1 monoclonal antibody (clone COX 111) (Product # 35-8100).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of Mouse Heart (Lane 1), NIH/3T3 (Lane 2), HT-29 (Lane 3), HCT 116 (Lane 4), C2C12 (Lane 5) and RAW 264.7 (Lane 6). The blots were probed with Anti-COX-1 Mouse Monoclonal Antibody (Product # 35-8100, 2-5 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Secondary Antibody, HRP conjugate (Product # 62-6520, 1:4000 dilution). A 64 kDa band corresponding to COX-1 was observed across cell lines tested except HCT 116 and RAW 264.7. A 56 kDa band was observed in Mouse Heart. Known quantity of protein samples were electrophoresed using Novex® NuPAGE®10 % Bis-Tris gel (Product # NP0301BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical staining of thyroid carcinoma tissue using COX-1 Monoclonal Antibody, Mouse (clone COX 111) (Product # 35-8100).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of COX-1 was done on NIH/3T3 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with COX-1 Mouse Monoclonal Antibody (358100, red histogram) or with mouse isotype control (yellow histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
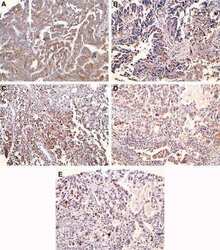
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Fluorescent immunocytochemistry was performed on cytospun BAL-cells using monoclonal antibodies against COX-1, COX-2, 15-LOX-1 and PPARgamma coupled with secondary antibodies labeled with fluorescent dyes Alexa488 (green) and Cy5 (red). Nuclear counter-staining was performed using DAPI (blue). Representative micrographs from a healthy and an asthmatic individual are shown for both the control air (A) and subway air (B) exposures. Semi-quantitative evaluation of staining intensities was performed using ImageJ software [24] .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Correlation between cyclooxygenase and CXCL9 expression in human breast cancer tissue samples . Homogenates were prepared from deep-frozen breast cancer tissue samples and analyzed for COX-1 and COX-2 expression by western blot analysis. Panel a shows representative immunoblots with high (first lane) and low (second lane) expression of both enzymes. For both cyclooxygenase isoenzymes samples were divided into a low and a high expressing group (see text for further details), and CXCL9 concentration was determined in all samples by ELISA. There was a trend towards lower CXCL9 expression in the high COX-1 expressing group ( b ; n low = 22, n high = 21), but a significantly reduced CXCL9 concentration in highly COX-2 expressing breast cancers ( c ; n low = 12, n high = 17). COX-2 overexpressing breast cancers display only half of the average CXCL9 concentration found in low-expressing cancers (56.5% +- 13.9%). (b and c) CXCL9/total protein ratio is presented as arbitrary units with 1.0 set as the arithmetic mean of the low expressing group. Horizontal lines in b and c represent the arithmetic mean.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Protein expressions of COX1 ( A ) and COX2 ( B ) in the aortas of wild-type (WT) littermates and eAPP -/- mice. Western blot results are the relative densitometry compared with beta-actin protein (n=10 per group for COX1 and n=8 per group for COX2). All results are representing box plots with whiskers showing the median, 25 th to 75 th percentiles, and min-max range. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Mortaparib-treated HeLa cells undergo apoptosis. Flow cytometric analysis showing increase in apoptotic cells in dose dependent manner following Mortaprib treatment ( a ). Western blot analysis showing molecular markers of apoptosis in control and Mortaparib-treated cells ( b ). Decrease in pro-caspase, BCl2, COX I, COX IV, Caspase-7, - 9, - 10 in Mortaparib-treated cells was recorded. Cleaved caspase-3 showed increase. Immunostaining showed decrease in COX I, COX IV, Procaspase 3 and BCl2 proteins in Mortaparib-treated cells (Scale bar = 20 muM). c . Caspase-3 activity, as evaluated using a fluoremetric assay, showed increase in dose-dependent manner ( d ). The quantitative data represents mean +- SD obtained from, at least, three independent experiments; p -values were calculated using Student's t -test. * < 0.05, ** < 0.01 and *** < 0.001 represent significant, very significant and very very significant, respectively
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA