Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- ABIN2482370 - Provider product page

- Provider
- antibodies-online
- Product name
- anti-Heat Shock Protein 70 (HSP70) antibody (Streptavidin)
- Antibody type
- Polyclonal
- Description
- PEG Purified
- Reactivity
- Human, Mouse, Rat
- Host
- Chicken/Avian
- Isotype
- IgY
- Vial size
- 100 μg
- Storage
- -20°C
Submitted references Characterization of a novel type of endogenous activator of soluble guanylyl cyclase.
Chaperone-mediated protein folding.
Molecular evolution of the HSP70 multigene family.
Uncoating protein (hsc70) binds a conformationally labile domain of clathrin light chain LCa to stimulate ATP hydrolysis.
Polypeptide chain binding proteins: catalysts of protein folding and related processes in cells.
Balashova N, Chang FJ, Lamothe M, Sun Q, Beuve A
The Journal of biological chemistry 2005 Jan 21;280(3):2186-96
The Journal of biological chemistry 2005 Jan 21;280(3):2186-96
Chaperone-mediated protein folding.
Fink AL
Physiological reviews 1999 Apr;79(2):425-49
Physiological reviews 1999 Apr;79(2):425-49
Molecular evolution of the HSP70 multigene family.
Boorstein WR, Ziegelhoffer T, Craig EA
Journal of molecular evolution 1994 Jan;38(1):1-17
Journal of molecular evolution 1994 Jan;38(1):1-17
Uncoating protein (hsc70) binds a conformationally labile domain of clathrin light chain LCa to stimulate ATP hydrolysis.
DeLuca-Flaherty C, McKay DB, Parham P, Hill BL
Cell 1990 Sep 7;62(5):875-87
Cell 1990 Sep 7;62(5):875-87
Polypeptide chain binding proteins: catalysts of protein folding and related processes in cells.
Rothman JE
Cell 1989 Nov 17;59(4):591-601
Cell 1989 Nov 17;59(4):591-601
No comments: Submit comment
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
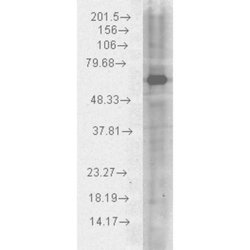
- Experimental details
- WB
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry