Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 16-0329-85 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD32 Monoclonal Antibody (6C4 (CD32)), Functional Grade, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This 6C4 monoclonal antibody reacts with human CD32 (also known as Fc gammaRII). This antibody recognizes two isoforms of the receptor, Fc gammaRIIA and Fc gammaRIIB. These 40-kDa polymorphic transmembrane glycoproteins are expressed on B cells, granulocytes, monocytes, macrophages, and platelets. Moreover, these receptors are detected on natural killer cells. CD32 enables interaction between Fc gammaRII-expressing cells and opsonized antigen or IgG-containing immune complexes. Both receptors exhibit low affinity towards IgG and play a role in inflammation and autoimmune disease. This clone has been reported to inhibit Ig binding in a rosette blocking assay. Applications Tested: This 6C4 (CD32) antibody has been tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at less than or equal to 1 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Storage and handling: Use in a sterile environment. Filtration: 0.2 µm post-manufacturing filtered. Purity: Greater than 90%, as determined by SDS-PAGE. Endotoxin Level: Less than 0.001 ng/µg antibody, as determined by LAL assay. Aggregation: Less than 10%, as determined by HPLC.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 6C4 (CD32)
- Vial size
- 500 µg
- Concentration
- 1 mg/mL
- Storage
- 4° C
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 APC (Product # 17-0199-42) and 0.5 µg of Mouse IgG1 K Isotype Control Functional Grade Purified (Product # 16-4714-82) (left) or 0.5 µg of Anti-Human CD32 Functional Grade Purified (right) followed by F (ab')2 Anti-Mouse IgG PE (Product # 12-4010-82).Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
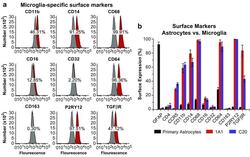
- Experimental details
- Fig. 6 Surface expression of key markers of microglia. a Flow cytometry analysis was used to measure the surface expression of CD11b, CD14, CD68, CD16, CD32, CD64, CD163, P2RY12, and TGFbetaR on the representative immortalized cell line hmuglia 1A1. In each experiment, 100,000 cells were resuspended in 1 mL of cold PBS in the presence of 0.5 mug of the antibody or isotype control for 20 min on ice. Appropriate secondary antibodies were used in the absence of fluorophore-conjugated primary antibody, and the cell-antibody complexes were centrifuged and resuspended in PBS. In each FACS profile, the gray distributions represent the proportion of cells bound to the isotype control, whereas the red distributions represent the proportions of cells bound by the target antibody. b Quantification of the abovementioned markers as well as GFAP, CD4, and CCR5 on the surface of primary human astrocytes and immortalized microglia, as indicated
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
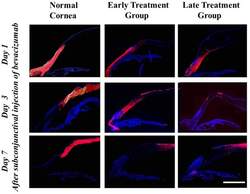
- Experimental details
- Figure 1 Immunohistochemical staining of bevacizumab after subconjunctival injection as detected by a Cy3-conjugated IgG antibody. Time-dependent diffusion of bevacizumab into the corneal stroma was observed. Normal: eyes without induction of corneal NV. Bevacizumab molecule staining was observed in the entire thickness of corneal stroma adjacent to the injection site in all groups 1 day after the injection. The distribution of bevacizumab staining moved to the more central cornea on day 3 in all groups. On day 7, bevacizumab staining decreased dramatically in the peripheral cornea but was detected in the central cornea in all groups. Purple red: bevacizumab molecule staining; blue: DAPI staining of cell nuclei. Scale bar: 4 mm.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry