Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Immunohistochemistry [6]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 50-9003-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor™ 660, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibodies AE1 and AE3 recognize many of the acidic and basic cytokeratin family members. Cytokeratins are intermediate filament proteins comprising one component of the cytoskeleton. There are two large families of cytokeratins, acidic and basic, but all contain the same basic domains (i.e. an alpha-helical core with an N- and C-terminal domain). The proteins are expressed in epithelial cells, but are developmentally regulated. Many tumors also express these proteins and their expression can help identify the origin of a neoplasm.
- Antibody clone number
- AE1/AE3
- Concentration
- 0.2 mg/mL
Submitted references Functional cooperation between co-amplified genes promotes aggressive phenotypes of HER2-positive breast cancer.
The Extracellular Small Leucine-Rich Proteoglycan Biglycan Is a Key Player in Gastric Cancer Aggressiveness.
Prognostic impact of CD73 expression and its relationship to PD-L1 in patients with radically treated pancreatic cancer.
Cysteine-Rich Angiogenic Inducer 61: Pro-Survival Function and Role as a Biomarker for Disseminating Breast Cancer Cells.
PD-L1 Expression on Circulating Tumour Cells May Be Predictive of Response to Regorafenib in Patients Diagnosed with Chemorefractory Metastatic Colorectal Cancer.
Screening Circulating Tumor Cells as a Noninvasive Cancer Test in 3388 Individuals from High-Risk Groups (ICELLATE2).
Neither epithelial nor mesenchymal circulating tumor cells isolated from breast cancer patients are tumorigenic in NOD-scid Il2rg(null) mice.
Flow cytometric determination of stem/progenitor content in epithelial tissues: an example from nonsmall lung cancer and normal lung.
KIT (CD117) expression in a subset of non-small cell lung carcinoma (NSCLC) patients.
Yang Y, Leonard M, Luo Z, Yeo S, Bick G, Hao M, Cai C, Charif M, Wang J, Guan JL, Lower EE, Zhang X
Cell reports 2021 Mar 9;34(10):108822
Cell reports 2021 Mar 9;34(10):108822
The Extracellular Small Leucine-Rich Proteoglycan Biglycan Is a Key Player in Gastric Cancer Aggressiveness.
Pinto F, Santos-Ferreira L, Pinto MT, Gomes C, Reis CA
Cancers 2021 Mar 16;13(6)
Cancers 2021 Mar 16;13(6)
Prognostic impact of CD73 expression and its relationship to PD-L1 in patients with radically treated pancreatic cancer.
Tahkola K, Ahtiainen M, Kellokumpu I, Mecklin JP, Laukkarinen J, Laakkonen J, Kenessey I, Jalkanen S, Salmi M, Böhm J
Virchows Archiv : an international journal of pathology 2021 Feb;478(2):209-217
Virchows Archiv : an international journal of pathology 2021 Feb;478(2):209-217
Cysteine-Rich Angiogenic Inducer 61: Pro-Survival Function and Role as a Biomarker for Disseminating Breast Cancer Cells.
Bartkowiak K, Heidrich I, Kwiatkowski M, Gorges TM, Andreas A, Geffken M, Verpoort K, Müller V, Schlüter H, Pantel K
Cancers 2021 Feb 2;13(3)
Cancers 2021 Feb 2;13(3)
PD-L1 Expression on Circulating Tumour Cells May Be Predictive of Response to Regorafenib in Patients Diagnosed with Chemorefractory Metastatic Colorectal Cancer.
Raimondi L, Raimondi FM, Di Benedetto L, Cimino G, Spinelli GP
International journal of molecular sciences 2020 Sep 20;21(18)
International journal of molecular sciences 2020 Sep 20;21(18)
Screening Circulating Tumor Cells as a Noninvasive Cancer Test in 3388 Individuals from High-Risk Groups (ICELLATE2).
Castro J, Sanchez L, Nuñez MT, Lu M, Castro T, Sharifi HR, Ericsson C
Disease markers 2018;2018:4653109
Disease markers 2018;2018:4653109
Neither epithelial nor mesenchymal circulating tumor cells isolated from breast cancer patients are tumorigenic in NOD-scid Il2rg(null) mice.
Donnenberg VS, Huber A, Basse P, Rubin JP, Donnenberg AD
NPJ breast cancer 2016;2:16004
NPJ breast cancer 2016;2:16004
Flow cytometric determination of stem/progenitor content in epithelial tissues: an example from nonsmall lung cancer and normal lung.
Donnenberg VS, Landreneau RJ, Pfeifer ME, Donnenberg AD
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2013 Jan;83(1):141-9
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2013 Jan;83(1):141-9
KIT (CD117) expression in a subset of non-small cell lung carcinoma (NSCLC) patients.
Donnenberg AD, Zimmerlin L, Landreneau RJ, Luketich JD, Donnenberg VS
PloS one 2012;7(12):e52885
PloS one 2012;7(12):e52885
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Multiplexed fluorescent western blot was performed using Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor 660, eBioscience™ (Product # 50-9003-82). Whole cell extracts of A-431 (Lane 1), MCF-7 (Lane 2), Caco-2 (Lane 3), Raji (Lane 4) as seen in Fig (a), A-431 (Lane 1), MOLT-4 (Lane 2) and Jurkat (Lane 3) as seen in Fig (b) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP03222BOX). Resolved proteins were transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot were probed with Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor 660, eBioscience™ (Product # 50-9003-82, 1 µg/mL) and HSP70 Polyclonal Antibody (Product # PA5-28003, 1:4000 dilution). Secondary antibody Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ Plus 800 (Product # A32808, 1:10000 dilution) was used for detection of HSP70. Fluorescent detection was performed using iBright FL1500 (Product # A44115).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
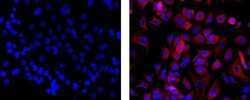
- Experimental details
- Immunocytochemistry of fixed and permeabilized MCF7 cells using 5 µg/mL of Mouse IgG1 K Isotype Control eFluor® 660 (left) or 5 µg/mL of Anti-Pan Cytokeratin (AE1/AE3) eFluor® 660 (right). Nuclei are stained with DAPI.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
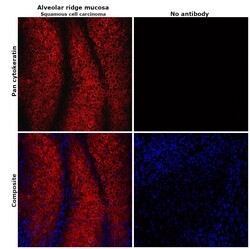
- Experimental details
- Immunohistochemical analysis of pan cytokeratin was performed using formalin-fixed paraffin-embedded alveolar ridge mucosa (squamous cell carcinoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
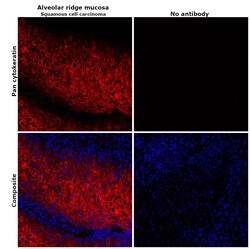
- Experimental details
- Immunohistochemical analysis of pan cytokeratin was performed using formalin-fixed paraffin-embedded alveolar ridge mucosa (squamous cell carcinoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
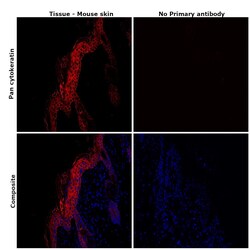
- Experimental details
- Immunohistochemical analysis of Pan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor™ 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
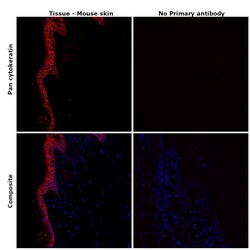
- Experimental details
- Immunohistochemical analysis ofpan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor™ 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis ofpan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor™ 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis ofpan Cytokeratin was performed using formalin-fixed paraffin-embedded mouse skin tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without Pan Cytokeratin Monoclonal Antibody (AE1/AE3), eFluor™ 660, eBioscience™ (Product # 50-9003-82) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
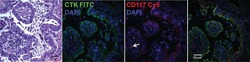
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
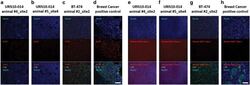
- Experimental details
- Figure 2 Immunofluorescent staining for human-specific Ki-67, human-specific cytokeratin, and murine major histocompatibility complex (MHC) Class I. Nuclei were stained with DAPI. Observed neoplasms in the URN10-014 group were negative for human Ki-67+ and human-specific cytokeratin (columns a , b ), but stained positively for murine-specific MHC Class I ( e , f ). A BT474 xenograft ( c , g ) and a human metastatic breast cancer control ( d , h ) were positive for human cytokeratin and Ki-67, but negative for murine MHC Class I antigen. Scale bar (white)=100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
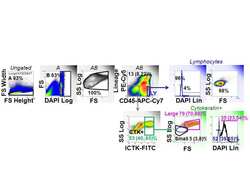
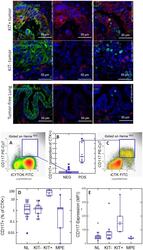
- Figure 1 CD117 expression in normal lung (NL) and NSCLC. Photomicrographs: Expression of CD117 and Ki-67 in NSC lung cancer and normal lung. The left columns show sections stained with CD117 (red), cytokeratin (green) and DAPI (blue). Sections in the center column show CD117 (red) and DAPI (blue) only, in order to reveal CD117 staining obscured by bright cytokeratin expression. Sections in the right column shows CD117 (red), the proliferation marker Ki67 (green) and DAPI (blue). Tumors were classified as KIT+ or negative on the basis of CD117 immunofluorescent staining of FFPE. In KIT+ tumors (top photomicrographs) CD117 (red stain, center and right panels) was expressed in virtually all cytokeratin+ tumor cells (green stain, left panels). Ki-67+ proliferating cells (green stain, right panels) were frequently seen among CD117+ tumor cells. In KIT negative tumors (center row of photomicrographs), only solitary CD117+ mast cells were detected (red stain, center and right panels). Proliferating Ki-67+ cells were frequent among cytokeratin+ CD117 negative tumor cells. Normal tumor-adjacent lung also appeared to lack CD117 expression among cytokeratin+ airway cells (bottom photomicrographs). Proliferating Ki-67+ cells were infrequent and confined to the basal layer of airway epithelium. When all NSCLC tumors are considered together, flow cytometry revealed bimodal CD117 expression (center panels A-C). Cells were gated on hematopoietic lineage negative singlet events with DNA conte
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
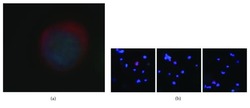
- Figure 1 (a) A circulating tumor cell prepared from a 7.5 ml blood sample from a 79-year-old male with no previous history of cancer. The cell is stained for cytokeratin (red) and for the cell nucleus (blue), typical of epithelial cells. Epithelial cells should not normally be present in the blood. The cell was negative for CD45, that is, not an immune cell. The cell nucleus has a large size typical of transcriptionally active cells, such as cancer cells, and the rounded shape of a cell in suspension, rather than the angular shape and cell sheath context of a normal solid tissue epithelium cell. (b) Three additional examples of circulating tumor cells stained for cytokeratin (red) and for the cell nucleus (blue). The lower magnification also shows the residual leucocytes surrounding the circulating tumor cells (blue nuclei, with no cytokeratin (red). The samples were enriched about 7500-fold for CTCs, with about 10,000 DAPI and CD45-positive leucocytes left in the sample after enrichment.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
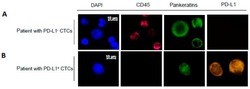
- Figure 2 Representative images of CTCs detected and subjected to immunostaining with DAPI, CD45, Pankeratins and PD-L1. Example images of CTCs from a patient with PD-L1 - CTCs ( A ) and PD-L1 + CTCs ( B ) are shown. The scale bar of 10 mum was applied to all pictures. ( C ) Number of CTCs and PD-L1 status isolated from blood samples from 38 patients (P1 through P38) with detectable PD-L1 status. ""P1"" stands for patient 1. Red bar represents the number of CK(+)/PD-L1(+) CTCs. Blue bar represents the number of CK(+)/PD-L1(-) CTCs.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
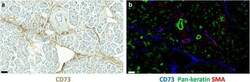
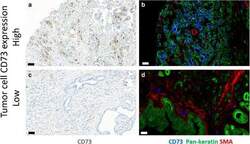
- Fig. 1 CD73 expression in normal pancreas. a Immunoperoxidase staining of normal pancreas for CD73 (brown). b Multicolour immunofluorescence staining of a consecutive section of normal pancreas for CD73 (blue), pan-cytokeratin (green) and alpha-smooth muscle actin (red). Bars, 50 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 2 CD73 expression in adenocarcinoma of pancreas. Representative immunoperoxidase stainings (a and c) for CD73 (brown) and multicolour immunofluorescence stainings (b, d) for CD73 (blue), pan-cytokeratin (green) and alpha-smooth muscle actin (red). Bars, 50 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
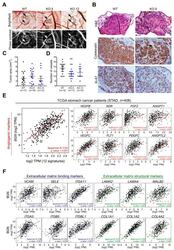
- Figure 6 Effect of biglycan in in vivo angiogenesis. ( A ) Representative images of tumors formed in the in vivo CAM by MKN74 cell models (WT and biglycan KO clones-KO.5 and KO.12). Phase contrast was used to better visualize tumor foci and vessels. MKN74 form multiple tumor foci (arrows/circles). Magnification at 20x. ( B ) Histological images of the formed tumors, cytokeratin staining confirming the presence of human epithelial tumors in the CAM. Ki-67 expression analysis was used to assess tumor aggressiveness. Histologically, tumors formed by KO cells present a less cohesive-like tumor mass with increased extracellular matrix stiffness. Hematoxilin & Eosin (H&E) staining images at 100X magnification, cytokeration and Ki-67 images at 200x magnification. ( C ) Quantification of the tumor area (mm 2 ) with no significant differences in the tumors being derived from the different cell lines. ( D ) Number of new vessels (less than 20 mum in diameter) formed towards the inoculation site. biglycan KO inoculated cells showed less capacity to form new vessels when compared to WT biglycan-positive tumors. ( E ) In silico gene analysis in GC tissues samples (TCGA, n = 408) showing that BGN was strongly positively correlated with angiogenic markers ( VEGFB, VEGFC, KDR, FLT1, FGF2, PDGFC, ANGPT1, ANGPT2, ANGPTL2, ANGPTL1, ANGPTL4, and ANGPTL7 ) . ( F ) In silico analysis demonstrating that BGN is positively correlated with ECM binding ( VCAM1, SELE, ITGA11, ITGA5, ITGB5, ITGBL1 ) and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
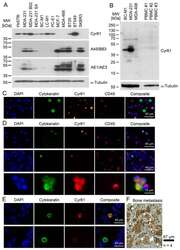
- Figure 5 Cyr61 detection in breast cancer cells. ( A ) Comparison of the cytoplasmic Cyr61 levels with cytokeratin levels, as analysed by the pan-cytokeratin antibody cocktails A45/BB3 and AE1/AE3 by Western blot analysis. ( B ) A comparison of the Cyr61 levels in the peripheral blood mononuclear cells (PBMC) of healthy women with the Cyr61 levels in breast cancer cell lines. ( C ) Cyr61 detection in BC-M1 and MDA-MB-468 spiked into blood samples from healthy women by immunocytochemical double staining. ( D ) Cyr61 detection in CTC from the peripheral blood of breast cancer patients (details: Table 1 ). ( E ) Detection of Cyr61 in the DTC from the bone marrow of breast cancer patients. The upper row shows a Cyr61-positive DTC, and the bottom row shows a Cyr61-negative DTC. ( F ) An immunohistochemical Cyr61 detection in the bone metastases of breast cancer patients. ( C - E ) The composite images are overlays of the Cytokeratin, Cyr61, Dapi and CD45 (if applied) signals, n biol : 3 ( A , C , E ), 4 ( F ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
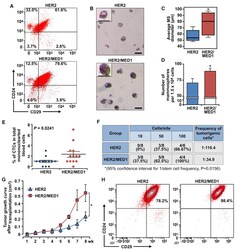
- Figure 4. MED1 overexpression promotes MMTV-HER2 CSC formation (A) FACS analyses of MMTV-HER2 and MMTV-HER2/MMTV-MED1 CSCs using antibodies against cell surface markers Lin, CD24, and CD29. (B) Mammosphere assays using FACS-sorted tumor cells in (A). Scale bar: 100 mum. (C and D) Average diameters (C) and numbers (D) of mammospheres formed in (B). (E) Statistics of flow cytometry analysis of CD45 - CK18 + EpCAM hi circulating tumor cells (CTCs) in mononuclear blood cells from MMTV-HER2 and MMTV-HER2/MMTV-MED1 tumor-bearing mice (n = 13). (F) Limiting dilution analyses of tumor-initiating cells in MMTV-HER2 and MMTV-HER2/MMTV-MED1 bulk tumors. (G) Growth curves of orthotopic MMTV-HER2 and MMTV-HER2/MMTV-MED1 tumor xenografts (n = 6). (H) FACS analyses of the grafted tumors using cell surface markers Lin (CD31, CD45, and Ter119), CD24, and CD29. The values are obtained from three independent experiments and shown as mean +- SD. *p < 0.05.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot