Antibody data
- Antibody Data
- Antigen structure
- References [6]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Immunohistochemistry [4]
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12542 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Skeletal Muscle Actin Monoclonal Antibody (5C5.F8.C7 (alpha-Sr-1))
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- MA5-12542 targets Actin Skeletal Muscle in IF and IHC (P) applications and shows reactivity with Amphibian, Bovine, Guinea Pig, Human, Ovine, Rabbit, and Rat samples.
- Antibody clone number
- 5C5.F8.C7 (alpha-Sr-1)
- Concentration
- 0.2 mg/mL
Submitted references In Vivo Analysis of Trypanosoma cruzi Persistence Foci at Single-Cell Resolution.
Differential roles of α-, β-, and γ-actin in axon growth and collateral branch formation in motoneurons.
Epithelial and muscular regionalization of the human developing anorectum.
Caveolin-1 expression is associated with plaque formation in hypercholesterolemic rabbits.
Spindle cell lipoma in an intramuscular lipoma.
Clear cell sarcoma of the stomach.
Ward AI, Lewis MD, Khan AA, McCann CJ, Francisco AF, Jayawardhana S, Taylor MC, Kelly JM
mBio 2020 Aug 4;11(4)
mBio 2020 Aug 4;11(4)
Differential roles of α-, β-, and γ-actin in axon growth and collateral branch formation in motoneurons.
Moradi M, Sivadasan R, Saal L, Lüningschrör P, Dombert B, Rathod RJ, Dieterich DC, Blum R, Sendtner M
The Journal of cell biology 2017 Mar 6;216(3):793-814
The Journal of cell biology 2017 Mar 6;216(3):793-814
Epithelial and muscular regionalization of the human developing anorectum.
Fritsch H, Aigner F, Ludwikowski B, Reinstadler-Zankl S, Illig R, Urbas D, Schwarzer C, Longato S
Anatomical record (Hoboken, N.J. : 2007) 2007 Nov;290(11):1449-58
Anatomical record (Hoboken, N.J. : 2007) 2007 Nov;290(11):1449-58
Caveolin-1 expression is associated with plaque formation in hypercholesterolemic rabbits.
Lin WW, Lin YC, Chang TY, Tsai SH, Ho HC, Chen YT, Yang VC
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2006 Aug;54(8):897-904
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2006 Aug;54(8):897-904
Spindle cell lipoma in an intramuscular lipoma.
Usta U, Türkmen E, Mizrak B, Yildiz D, Güzel Z
Pathology international 2004 Sep;54(9):734-9
Pathology international 2004 Sep;54(9):734-9
Clear cell sarcoma of the stomach.
Pauwels P, Debiec-Rychter M, Sciot R, Vlasveld T, den Butter B, Hagemeijer A, Hogendoorn PC
Histopathology 2002 Dec;41(6):526-30
Histopathology 2002 Dec;41(6):526-30
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Actin Skeletal Muscle was done on 70% confluent log phase L6 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Actin Skeletal Muscle (5C5.F8.C7 (alpha-Sr-1)) Mouse Monoclonal Antibody (Product # MA5-12542) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L)/IgM (L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Formalin-fixed, paraffin-embedded human skeletal muscle stained with Skeletal Muscle Actin antibody using peroxidase-conjugate and DAB chromogen. Note cytoplasmic staining of skeletal muscle cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
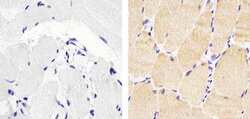
- Experimental details
- Immunohistochemistry analysis of Actin Skeletal Muscle showing staining in the cytoplasm of paraffin-embedded human skeletal muscle tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Actin Skeletal Muscle Mouse Monoclonal Antibody (Product # MA5-12542) diluted in 3% BSA-PBS at a dilution of 1:100 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Actin Skeletal Muscle showing staining in the cytoplasm of paraffin-embedded rat skeletal muscle tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Actin Skeletal Muscle Mouse Monoclonal Antibody (Product # MA5-12542) diluted in 3% BSA-PBS at a dilution of 1:100 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Actin Skeletal Muscle showing staining in the cytoplasm of paraffin-embedded mouse skeletal muscle tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Actin Skeletal Muscle Mouse Monoclonal Antibody (Product # MA5-12542) diluted in 3% BSA-PBS at a dilution of 1:100 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
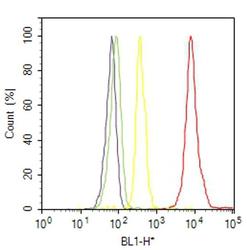
- Experimental details
- Flow cytometry analysis of Actin was done on C2C12 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Actin Skeletal Muscle Mouse Monoclonal Antibody (MA512542, red histogram) or with mouse isotype control (yellow histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Subcellular localization of endogenous Actalpha, Actbeta, and Actgamma mRNAs in cultured motoneurons. (A) In situ hybridization signals for Actalpha (A), Actbeta (B), and Actgamma transcripts (C) were detected in soma, axons, and axonal growth cones of cultured motoneurons. Cells were immunostained against Tau to determine cell boundaries. (D-F) Quantification of FISH signals shows that Actbeta and Actgamma are detectable at similar levels in the soma, but Actbeta is more abundant in the axon and axonal growth cone (***, P < 0.0001 by one-way ANOVA with Dunn's post-test for n = 6). Shown are mean +- SEM. (G) In triplex assays, cells were incubated simultaneously with probes detecting transcripts of Actalpha (white), Actbeta (green), and Actgamma (blue). Differential localization of actin mRNAs in soma, axons, and growth cones indicates that these mRNAs are present in different RNP granules. White circles and arrowheads in A and G indicate Actalpha mRNA punctae. Bars, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Actalpha protein localizes to axonal branch points, whereas Actbeta and Actgamma are highly enriched in axonal filopodia and axonal growth cones. (A-C) Immunoblots of total lysates obtained from actin isoform-specific knockdown and control motoneurons probed with specific Actalpha (A), Actbeta (B), and Actgamma (C) antibodies. Calnexin was used as loading control. All three isoforms are detected in cultured motoneurons. (D) shRNA-mediated knockdown leads to a 60% reduction in Actalpha (**, P < 0.004 for n = 4), a 95% reduction in Actbeta (**, P < 0.002 for n = 10), and a 85% reduction in Actgamma (**, P < 0.005 for n = 5) protein levels (one-tailed Mann-Whitney test). Shown are mean +- SEM. (E-G) Motoneurons were stained against Tau and Actalpha (E), Actbeta (F), and Actgamma (G). Actalpha protein is highly enriched in axonal branch points and neurites (E). Actbeta (F) and Actgamma proteins (G) are very abundant in the soma and localize to axonal filopodia and axonal growth cone filopodia. (H-J) Motoneurons were extracted using Triton X-100 to remove G-actin. Cells were fixed and stained with phalloidin and Actalpha (H), Actbeta (I), and Actgamma (J) antibodies. (H) Colocalization of Actalpha with phalloidin shows that this isoform incorporates into F-actin in the axon and particularly in the axonal branch points. (I) Filamentous Actbeta is present predominantly in the axonal growth cone filopodia. (J) Actgamma colocalizes with phalloidin mostly in the soma and axon
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry