Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-9109-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-ERK1/2 (Thr202, Tyr204) Monoclonal Antibody (MILAN8R), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This MILAN8R monoclonal antibody recognizes human and mouse extracellular signal-regulated kinases 1 and 2 (also known as ERK1/2, p44/p42, or MAPK3/1) when phosphorylated on T202/Y204. ERK1/2 belong to a family of conserved serine/threonine protein kinases known as mitogen-activated protein kinases (MAPKs) that are involved in many cellular programs such as proliferation, differentiation, motility, and survival. ERK1/2 signaling is activated in response to numerous extracellular stimuli including mitogens, growth factors, and cytokines. The primary activators of ERK1/2 are MEK1 and MEK2 which act by phosphorylating the activation loop residues T202/Y204 and T185/Y187 in ERK1 and ERK2, respectively. Several downstream targets of ERK1/2 have been identified, including p90RSK and the transcription factor Elk-1. ERK1/2 are negatively regulated by MAPK phosphatases, known as DUSPs or MKPs, as well as by chemical inhibitors of MEK including U0126 and PD98059. Disruption of the ERK pathway is common in many types of cancer. Specificity of this MILAN8R clone was determined by ELISA, flow cytometry, and western blotting. Applications Reported: This MILAN8R antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This MILAN8R antibody has been pre-titrated and tested by intracellular staining followed by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Staining Protocol: We recommend using Protocol C: Two-step protocol: Fixation/Methanol. Protocol A: Two-step protocol: intracellular (cytoplasmic) proteins and Protocol B: One-step protocol: intracellular (nuclear) proteins cannot be used. All Protocols can be found in the Flow Cytometry Protocols: "Staining Intracellular Antigens for Flow Cytometry Protocol" located in the Best Protocols Section under the Resources tab online. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- MILAN8R
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Suppression of 4.1R enhances the potency of NKG2D-CAR T cells against pancreatic carcinoma via activating ERK signaling pathway.
Berberine modulates hyper-inflammation in mouse macrophages stimulated with polyinosinic-polycytidylic acid via calcium-CHOP/STAT pathway.
Cancer cell-intrinsic expression of MHC II in lung cancer cell lines is actively restricted by MEK/ERK signaling and epigenetic mechanisms.
PRMT5 Modulates Splicing for Genome Integrity and Preserves Proteostasis of Hematopoietic Stem Cells.
Cas9-mediated excision of proximal DNaseI/H3K4me3 signatures confers robust silencing of microRNA and long non-coding RNA genes.
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Heterogeneity of leukemia-initiating capacity of chronic myelogenous leukemia stem cells.
Germline and somatic FGFR1 abnormalities in dysembryoplastic neuroepithelial tumors.
The Bacterial Enzyme IdeS Cleaves the IgG-Type of B Cell Receptor (BCR), Abolishes BCR-Mediated Cell Signaling, and Inhibits Memory B Cell Activation.
Gao Y, Lin H, Guo D, Cheng S, Zhou Y, Zhang L, Yao J, Farooq MA, Ajmal I, Duan Y, He C, Tao L, Wu S, Liu M, Jiang W
Oncogenesis 2021 Sep 21;10(9):62
Oncogenesis 2021 Sep 21;10(9):62
Berberine modulates hyper-inflammation in mouse macrophages stimulated with polyinosinic-polycytidylic acid via calcium-CHOP/STAT pathway.
Kim HJ, Kim YJ, Park W
Scientific reports 2021 May 28;11(1):11298
Scientific reports 2021 May 28;11(1):11298
Cancer cell-intrinsic expression of MHC II in lung cancer cell lines is actively restricted by MEK/ERK signaling and epigenetic mechanisms.
Neuwelt AJ, Kimball AK, Johnson AM, Arnold BW, Bullock BL, Kaspar RE, Kleczko EK, Kwak JW, Wu MH, Heasley LE, Doebele RC, Li HY, Nemenoff RA, Clambey ET
Journal for immunotherapy of cancer 2020 Apr;8(1)
Journal for immunotherapy of cancer 2020 Apr;8(1)
PRMT5 Modulates Splicing for Genome Integrity and Preserves Proteostasis of Hematopoietic Stem Cells.
Tan DQ, Li Y, Yang C, Li J, Tan SH, Chin DWL, Nakamura-Ishizu A, Yang H, Suda T
Cell reports 2019 Feb 26;26(9):2316-2328.e6
Cell reports 2019 Feb 26;26(9):2316-2328.e6
Cas9-mediated excision of proximal DNaseI/H3K4me3 signatures confers robust silencing of microRNA and long non-coding RNA genes.
Janga H, Aznaourova M, Boldt F, Damm K, Grünweller A, Schulte LN
PloS one 2018;13(2):e0193066
PloS one 2018;13(2):e0193066
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Chu F, Hu Y, Zhou Y, Guo M, Lu J, Zheng W, Xu H, Zhao J, Xu L
Clinical and experimental immunology 2018 Feb;191(2):166-179
Clinical and experimental immunology 2018 Feb;191(2):166-179
Heterogeneity of leukemia-initiating capacity of chronic myelogenous leukemia stem cells.
Zhang B, Li L, Ho Y, Li M, Marcucci G, Tong W, Bhatia R
The Journal of clinical investigation 2016 Mar 1;126(3):975-91
The Journal of clinical investigation 2016 Mar 1;126(3):975-91
Germline and somatic FGFR1 abnormalities in dysembryoplastic neuroepithelial tumors.
Rivera B, Gayden T, Carrot-Zhang J, Nadaf J, Boshari T, Faury D, Zeinieh M, Blanc R, Burk DL, Fahiminiya S, Bareke E, Schüller U, Monoranu CM, Sträter R, Kerl K, Niederstadt T, Kurlemann G, Ellezam B, Michalak Z, Thom M, Lockhart PJ, Leventer RJ, Ohm M, MacGregor D, Jones D, Karamchandani J, Greenwood CM, Berghuis AM, Bens S, Siebert R, Zakrzewska M, Liberski PP, Zakrzewski K, Sisodiya SM, Paulus W, Albrecht S, Hasselblatt M, Jabado N, Foulkes WD, Majewski J
Acta neuropathologica 2016 Jun;131(6):847-63
Acta neuropathologica 2016 Jun;131(6):847-63
The Bacterial Enzyme IdeS Cleaves the IgG-Type of B Cell Receptor (BCR), Abolishes BCR-Mediated Cell Signaling, and Inhibits Memory B Cell Activation.
Järnum S, Bockermann R, Runström A, Winstedt L, Kjellman C
Journal of immunology (Baltimore, Md. : 1950) 2015 Dec 15;195(12):5592-601
Journal of immunology (Baltimore, Md. : 1950) 2015 Dec 15;195(12):5592-601
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
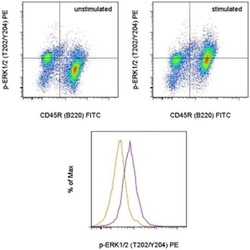
- Experimental details
- TOP: Mouse splenocytes were unstimulated (left) or stimulated with F (ab')2 Anti-Mouse IgM, u chain specific Functional Grade Purified (Product # 16-5092-85) and Anti-Mouse CD40 Functional Grade Purified (Product # 16-0401-82) (right). The cells were then intracellularly stained with Anti-Human/Mouse CD45R (B220) FITC (Product # 11-0452-82) and Anti-Human/Mouse phospho-ERK1/2 (T202/Y204) PE using the IC Fixation/Methanol Protocol. Cells in the lymphocyte gate were used for analysis. BOTTOM: Mouse splenocytes were unstimulated (orange histogram) or stimulated with F (ab')2 Anti-Mouse IgM, u chain specific Functional Grade Purified (Product # 16-5092-85) and Anti-Mouse CD40 Functional Grade Purified (Product # 16-0401-82) (purple histogram). The cells were then intracellularly stained with Anti-Human/Mouse CD45R (B220) FITC (Product # 11-0452-82) and Anti-Human/Mouse phospho-ERK1/2 (T202/Y204) PE using the IC Fixation/Methanol Protocol. B220+ cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Correlation between MHC II induction and basal phosphorylated ERK1/2 levels in murine NSCLC cells. Mouse NSCLC lines were cultured for 48 hours, and analyzed for basal levels of intracellular pERK1/2 by flow cytometric analysis. (A) Relative pERK1/2 levels in four murine NSCLC cell lines, including (B) representative histograms. Data are from two independent experiments, with n=3 replicates total. Statistical comparisons of pERK levels were focused on comparison of oncogene-matched cancer cell lines; pERK levels were not significantly different between CMT167 and EA1 cells. MFI is x10 6 . Graphs show mean+-SEM. Flow cytometry data analyzed singlets with MFI data visualized x10 6 values. LLC, Lewis lung carcinoma; MFI, median fluorescent intensity; NSCLC, non-small cell lung cancer.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
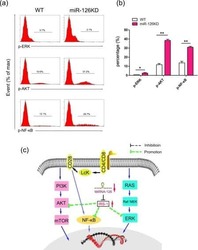
- Fig. 4 4.1R deficiency regulated the function of CAR T cells via ERK signaling pathway. a Mock T, NKG2D-CAR T, NC-NKG2D-CAR T, and KD2-NKG2D-CAR T were co-incubated with PANC28 at a 9:1 ratio for 16 h. The expression of p-ERK was detected by flow cytometry (left), and the percentage of p-ERK-positive T cells was statistically analyzed (right) ( n = 3). b Line plots displayed the cytotoxicity of NC-NKG2D-CAR T and KD2-NKG2D-CAR T against PANC28 at a different effector to target ( E : T ) ratios for 16 h in the absence and presence of 10 muM U0126. c NC-NKG2D-CAR T and KD2-NKG2D-CAR T were co-incubated with PANC28 at a different effector to target ( E : T ) ratios for 7 days in the absence and presence of 10 muM U0126. CFSE dilution was used as a measure of cell proliferation (left), and MFI was calculated (right) ( n = 3). NC-NKG2D-CAR T and KD2-NKG2D-CAR T were co-incubated with PANC28 at a different effector to target ( E : T ) ratios for 16 h in the absence and presence of 10 muM U0126. The expression of CD69 ( d ), Gzm B ( e ), and TIM-3 ( f ) was detected by flow cytometry (left). MFI and percentage were statistically analyzed and shown in column chart (middle and right) ( n = 3). Data were representative of three independent experiments. ** P < 0.01, *** P < 0.001, NS not significant.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
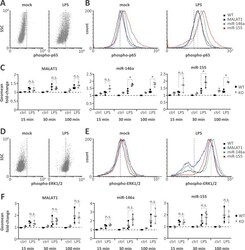
- Experimental details
- Fig 4 Elevated NFkappaB p65 but not ERK1/2 activity on miR-146a and miR-155 knockout. A) Representative FACS scatter plots showing a right-shift of 30 min LPS-stimulated (1 mug / ml) compared to mock-treated monocytes stained with phospho-p65 antibody (PE-channel). B) Representative histogram plots showing an increased right-shift of miR-146a and miR-155 deficient compared to control or MALAT1 deficient monocytes after 30 min LPS-stimulation (1 mug / ml) and staining with a phospho-p65 antibody (PE-channel). C) Fold change in phospho-p65 signal in monocytes stimulated with LPS (1 mug / ml) for 15, 30 or 100 min compared to mock-treatment (ctrl) in wild-type (WT) or the indicated ncRNA knockout (KO) cells. All fold-changes are relative to the respective WT mock control. D-F) Same as A-C) but with phospho-ERK1/2 staining (APC-channel). Statistical significance was determined by a one-way ANOVA test with multiple comparisons (* p
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry