Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Western blot [2]
- Other assay [17]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-988 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- GAPDH Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-988 detects rat GAPDH in human HeLa cell lysates and mouse and rat tissues.. PA1-988 has been used successfully in Western blotting. By Western blot this antibody specifically detects a ~36 kDa protein representing GAPDH. PA1-988 immunizing peptide corresponds to amino acid residues 4-18 from human GAPDH.
- Reactivity
- Human, Mouse, Rat, Canine
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
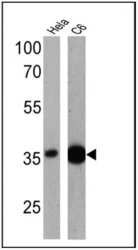
- Experimental details
- Western blot analysis of GAPDH was performed by loading 25 µg of Hela (lane 1) and C6 (lane 2) cell lysates onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a GAPDH polyclonal antibody (Product # PA1-988) at a dilution of 1:1000 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~36 kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of A549 (Lane 1), COS-7 (Lane 2), MDCK (Lane 3), HT-29 (Lane 4), C2C12 (Lane 5), PC-12 (Lane 6), U-2 OS (Lane 7) and tissue extract (30 µg lysate) of Rat Testis (Lane 8), Mouse Liver (Lane 9). The blot was probed with Anti-GAPDH Polyclonal Antibody (Product # PA1-988, 1:500 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 37 kDa band corresponding to GAPDH was observed across the cell lines and tissues tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 10% Bis-Tris gel (Product # NP0302BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5% skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 4 Differential protein levels and phenotypes are exhibited among MCF10A cells following selection through IH. (A) Analysis of protein levels was performed using the Reverse Phase Protein Array (RPPA) on the parental MCF10A line, passaged control clone 7, IH-selected clone 4, and IH-selected clone 9. Each clone was submitted in duplicate. Hierarchical clustering of significant (FDR
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Representative western blots showing the effects of grape pomace extract (GPE) on the expression of (A) gamma-glutamylcysteine synthetase (GCS), (B) catalase (CAT), (C) superoxide dismutase (SOD) and (D) heme oxygenase 1 (HO-1) in C2C12 muscle cells. The results of densitometric quantification for all enzymes are also shown. The cells were incubated with GPE at 2.5 and 10 u g/ml for 3, 6, 12, 18 and 24 h. The expression of GAPDH was used as a loading control for normalization. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Representative western blots showing the effects of grape pomace extract (GPE) on the expression of (A) gamma-glutamylcysteine synthetase (GCS), (B) catalase (CAT), (C) superoxide dismutase (SOD) and (D) heme oxygenase 1 (HO-1) in EA.hy926 endothelial cells. The results of densitometric quantification for all enzymes are also shown. The cells were incubated with GPE at 0.068 and 0.250 u g/ml for 3, 6, 12, 18 and 24 h. The expression of GAPDH was used as a loading control for normalization. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Gene therapy with AAV vectors encoding for a secretable GAA variant rapidly rescues skeletal muscle defects in Gaa KO DBA mice. Fig. 4 (a) Study diagram: Gaa KO DBA mice were treated by intravenous injection of AAV9 vectors encoding for a codon-optimized secretable human GAA (secGAA) under the control of the tandem liver-muscle LiMP promoter (AAV-secGAA, dose: 4 x 10 10 vg/mouse, ~2 x 10 13 vg/kg); littermate untreated (no Tx) Gaa KO DBA and Gaa WT DBA were used as controls. Vectors were delivered at postnatal day 2 (P2), analyses were performed 4 months (4M) later; SkM (skeletal muscle), SC (spinal cord). Biochemical (b-g) and downstream analysis of RNA sequencing (RNA-seq) data (h-i) of skeletal muscle (SkM) at the end of the study. (b) Analyses of GAA enzyme activity in triceps muscle. (c) Analyses of glycogen storage in triceps muscle. (d) Western blot analyses of triceps lysates with anti-human GAA, anti-Parkin, and anti-p62 antibodies, anti-Gapdh antibody was used as loading control. The molecular weight marker (kDa) is depicted. The lanes were run on the same gel but were non-contiguous. Western blot quantification is shown in panels e (GAA), f (p62) and g (Parkin). (b-g) Gaa WT DBA ( n = 4), Gaa KO DBA ( n = 5), Gaa KO DBA +AAV ( n = 4). Data are depicted as average+-SD. (h) Gene expression clusters. Expression profiles for 2573 DEGs with abs (logFC) > 0.5, detected in contrast untreated Gaa KO DBA vs. Gaa WT DBA , were constructed by recovering averaged expres
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Characteristics of iNs. (A) iNs exhibited bipolar neuronal morphologies and expressed tuj1 (red) and MAP2 (green) at different phases (day 7, 14 and 30). (B) iNs were stained for NeuN, GABA, vGLUT1 and GFAP (all green) on day 30. Scale bar, 50 um. (C) Reverse transcription-quantitative PCR analysis revealed that in comparison with HFFs, there was a significant increase in gene expression levels of MAP2, DCX, NeuroD1 and ASCL1 in iNs, whereas human fibroblast-specific genes were downregulated in iNs on day 30. Data are presented as the mean +- SD. (D) Western blot analysis of the protein expression levels of Tuj1, NeuN and GAPDH in HFFs and iNs. (E) Graphs show the semi-quantification of the western blot analysis. n=3 independent experiments. Data are presented as the mean +- SD. Unpaired Student's t-tests were used to compare data. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 9 SPK-3006 drives vector dose-dependent expression of GAA in liver and tissue uptake in non-human primates. A Circulating GAA antigen levels 4 weeks of post-injection of C57BL/6 mice with AAV-GAA or SPK-3006 at 2 x 10 12 vg/kg ( n = 5 per group). B GAA activity in plasma of non-human primates treated with SPK-3006 ( n = 4 per group). C , E Western blot analysis of GAA antigen levels in spinal cord ( C ) and brain ( E ). D , F Quantification of lysosomal GAA antigen in spinal cord ( D ) and brain ( F ) (2 x 10 12 and 6 x 10 12 vg/kg, n = 4 per group; 2 x 10 13 , n = 3). Data shown as average +- SD. Statistical analysis: A Two-sided Mann-Whitney t -test; B Two-way ANOVA with Tukey post-hoc test; D , F One-way ANOVA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Phosphorylation levels of LRRK2 at Ser1292 (pSer1292) are elevated in G2019S knock-in (KI) mice. Striatal pSer1292 and total LRRK2 levels were measured by Western blotting in 12-month-old G2019S KI mice and age-matched WT controls. Representative blots (left) and quantification (right) are shown. Data are expressed as pSer1292 LRRK2/total LRRK2 and are means +- SEM of 7 animals per group. Statistical analysis was performed with the Student t -test, two tailed for unpaired data. ** p < 0.01, different from WT
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 Autophagic flux studies in GSDIII mouse tissues. a. Augmented glycogen content in both GDE KO mice of 14 and 20 months . b. SQSTM1/p62 protein by immunostaining of total protein lysates of quadriceps tissues showing variable levels of SQSTM1/p62 proteinold mice. c. p62/SQSTM1 protein by immunostaining of total protein lysates of quadriceps tissues in 20 months GSDIII mice showing slightly decreased level of p62
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 ( A ). Western Blot analysis of NACHO expression. GH3 and GH4C1 cells express endogenous NACHO, but mouse macrophage-like RAW264.7 cells, primary mouse macrophages, and HEK-293 do not. ( B ) In vitro 125 I-alphaBGT binding assessments. Primary cultured macrophages and GH3 cells transfected with rat chrna7 plasmid, but not RAW264.7 mouse macrophage-derived cells, showed 125 I-alphaBGT binding.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot