Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-918 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Calmodulin Monoclonal Antibody (6D4)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA3-918 detects calmodulin from bovine, chicken, Chlamydomonas, Dictyostelium, and rat samples. This antibody does not detect parvalbumin, tropinin, S-100, or myosin light chain kinase (MLCK). MA3-918 has been successfully used in Western blot, immunocytochemistry and ELISA procedures. By Western blot, this antibody detects a 17 kDa protein representing calmodulin from Dictyostelium cell lysate. Addition of EGTA to buffers completely inhibits antibody staining. Immunohistochemical staining of calmodulin in Dictyostelium cells with MA3-918 results in staining of the contractile vacuoles. The MA3-918 antigen is calmodulin purified from Dictyostelium discoideum.
- Reactivity
- Human, Rat, Bacteria, Bovine, Chicken/Avian
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 6D4
- Vial size
- 100 µL
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of purified calmodulin using Product # MA3-917 and Product # MA3-918.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
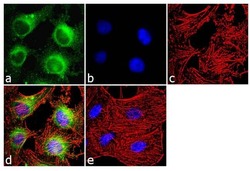
- Experimental details
- Immunofluorescence analysis of Calmodulin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Calmodulin (6D4) Mouse Monoclonal Antibody (Product # MA3-918) at 1:250 dilution in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
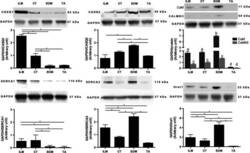
- Experimental details
- Figure 2 Protein levels of sarcoplasmic reticulum Ca 2+ -handling proteins in intrinsic laryngeal muscles (ILM), cricothyroid (CT) muscle, extraocular muscles (EOM), and tibialis anterior (TA) muscle. Western blot analysis showing relative abundance of indicated proteins: calsequestrins 1 and 2 (CASQ1 and CASQ2), Sercas 1 and 2 (SERCA1 and SERCA2), calmodulin (CaM), calmodulin kinase II (CaMKII), and Orai1. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a control for protein loading, Western blot transfer and nonspecific changes in protein levels. The molecular weight, expressed in kDa, for each protein is indicated. Quantifications from three independent muscle samples, each sample containing muscles pooled from three or four different rats. Asterisks and different letter combination indicate statistical significance (* P < 0.05 and ab, ac, ad, bc, or bd P < 0.05, respectively). In ILM, CASQ 1 was more abundant than CASQ2 compared with TA. SERCA1 was less than SERCA2 in ILM and higher compared with TA. CT and the other ILM showed similar levels of the proteins studied.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
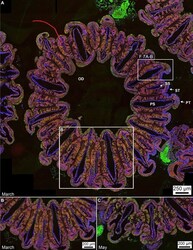
- Experimental details
- Figure 6 Immunohistochemical fluorescence microscopy of the distributions and abundance of Symbiodiniaceae, chromatophores carbonic anhydrase, calmodulin and mucus within tagged polyp tissues of O. faveolata . ( A ) Horizontal cross-section of decalcified polyp tissues of O. faveolata collected in March 2008 of superimposed emissions from mucus (blue fluorescence), carbonic anhydrase (green fluorescence), and calmodulin (red fluorescence). ( B ) Enlargement of the box shown in A. ( C ) Horizontal cross-section of a decalcified polyp of O. faveolata collected in May 2008 of superimposed mucus (blue fluorescence), carbonic anhydrase (green fluorescence), and calmodulin (red fluorescence). Enlargement from the box shown within a full polyp shown in Supplementary Fig. S7 . Labels include: PS--primary septum; SS--secondary septum; PT--primary tentacle; ST--secondary tentacle; and OD--oral disk.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
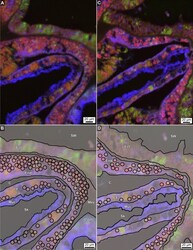
- Experimental details
- Figure 7 Enlargement of immunohistochemical fluorescence microscopy of the distributions and abundance of carbonic anhydrase, calmodulin and mucus within tagged polyp tissues of O. faveolata . ( A ) Original enlarged image (from box 7A-B shown in Fig. 6 A) of a horizontal cross-section of decalcified polyp tissues of O. faveolata collected in March 2008 exhibiting superimposed emissions from mucus (blue fluorescence), carbonic anhydrase (green fluorescence), and calmodulin (red fluorescence). ( B ) Line tracing of A indicating individual Symbiodiniaceae. ( C ) A second enlargement (from area shown in Supplementary Figs. S5 , S6 ) image of a horizontal cross-section of decalcified polyp tissues of O. faveolata collected in May 2008 exhibiting superimposed emissions from mucus (blue fluorescence), carbonic anhydrase (green fluorescence), and calmodulin (red fluorescence). ( D ) Line tracing of C indicating individual Symbiodiniaceae are found in both the oral endoderm and the calicoblastic epithelium. Labels include: End--endoderm; Mes--mesoglea; Ect--ectoderm; Muc--mucus; PS--primary septum; SW--seawater at the time the polyps were collected; and Sk--skeleton. Tissue structure terminology follows nomenclature in Allemand et al and Peters .
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA