35-9200
antibody from Invitrogen Antibodies
Targeting: PRLR
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [33]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Flow cytometry [1]
- Other assay [33]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 35-9200 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Prolactin Receptor Monoclonal Antibody (1A2B1)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- This antibody is predicted to react with mouse and rat and reacts with the long form of the human prolactin receptor (hPRLr) protein at ~85-90 kDa, and may also identify the human prolactin receptor intermediate and delta S1 isoforms by western blotting. On immunoprecipitation, this antibody strongly recognizes only the long form of hPRLr. During development reactivity was confirmed with human T-47D breast carcinoma cells.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 1A2B1
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references The short isoform of PRLR suppresses the pentose phosphate pathway and nucleotide synthesis through the NEK9-Hippo axis in pancreatic cancer.
The human intermediate prolactin receptor is a mammary proto-oncogene.
RAB6 GTPase regulates mammary secretory function by controlling the activation of STAT5.
Bispecific Antibodies and Antibody-Drug Conjugates (ADCs) Bridging HER2 and Prolactin Receptor Improve Efficacy of HER2 ADCs.
Role of Prolactin Receptors in Lymphangioleiomyomatosis.
Gain-of-function Prolactin Receptor Variants Are Not Associated With Breast Cancer and Multiple Fibroadenoma Risk.
Stimulation of prolactin receptor induces STAT-5 phosphorylation and cellular invasion in glioblastoma multiforme.
The Role of Prolactin in Bone Metastasis and Breast Cancer Cell-Mediated Osteoclast Differentiation.
Prolactin signaling through focal adhesion complexes is amplified by stiff extracellular matrices in breast cancer cells.
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
The prolactin receptor is expressed in rheumatoid arthritis and psoriatic arthritis synovial tissue and contributes to macrophage activation.
Dense collagen-I matrices enhance pro-tumorigenic estrogen-prolactin crosstalk in MCF-7 and T47D breast cancer cells.
Blockade of estrogen-stimulated proliferation by a constitutively-active prolactin receptor having lower expression in invasive ductal carcinoma.
Prolactin receptor expression and breast cancer: relationships with tumor characteristics among pre- and post-menopausal women in a population-based case-control study from Poland.
Reevaluation of the proposed autocrine proliferative function of prolactin in breast cancer.
Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells.
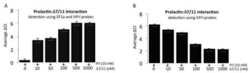
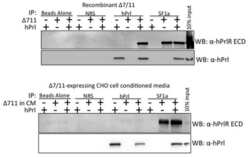
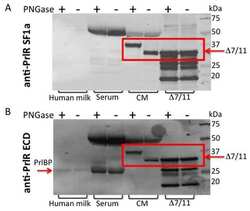
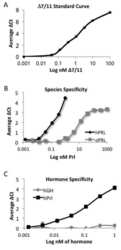
Characterization of Δ7/11, a functional prolactin-binding protein.
Functional consequences of prolactin signalling in endothelial cells: a potential link with angiogenesis in pathophysiology?
Prolactin increases survival and migration of ovarian cancer cells: importance of prolactin receptor type and therapeutic potential of S179D and G129R receptor antagonists.
Functional impact of manipulation on the relative orientation of human prolactin receptor domains.
The prolactin receptor is expressed in macrophages within human carotid atherosclerotic plaques: a role for prolactin in atherogenesis?
Identification of gain-of-function variants of the human prolactin receptor.
Prolactin and oestrogen synergistically regulate gene expression and proliferation of breast cancer cells.
Azaspiracid-1 inhibits endocytosis of plasma membrane proteins in epithelial cells.
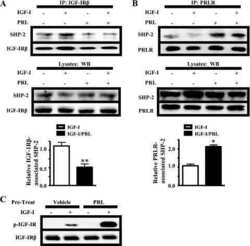
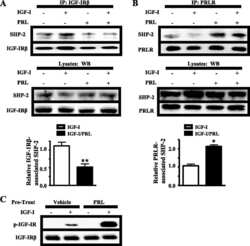
Prolactin enhances insulin-like growth factor I receptor phosphorylation by decreasing its association with the tyrosine phosphatase SHP-2 in MCF-7 breast cancer cells.
Re-evaluation of the prolactin receptor expression in human breast cancer.
Polyubiquitination of prolactin receptor stimulates its internalization, postinternalization sorting, and degradation via the lysosomal pathway.
Identification of a gain-of-function mutation of the prolactin receptor in women with benign breast tumors.
Quantification of PRL/Stat5 signaling with a novel pGL4-CISH reporter.
A molecular mimic demonstrates that phosphorylated human prolactin is a potent anti-angiogenic hormone.
Proteasomes mediate prolactin-induced receptor down-regulation and fragment generation in breast cancer cells.
Endogenous human prolactin and not exogenous human prolactin induces estrogen receptor alpha and prolactin receptor expression and increases estrogen responsiveness in breast cancer cells.
Inhibition of prolactin (PRL)-induced proliferative signals in breast cancer cells by a molecular mimic of phosphorylated PRL, S179D-PRL.
Nie H, Huang PQ, Jiang SH, Yang Q, Hu LP, Yang XM, Li J, Wang YH, Li Q, Zhang YF, Zhu L, Zhang YL, Yu Y, Xiao GG, Sun YW, Ji J, Zhang ZG
Theranostics 2021;11(8):3898-3915
Theranostics 2021;11(8):3898-3915
The human intermediate prolactin receptor is a mammary proto-oncogene.
Grible JM, Zot P, Olex AL, Hedrick SE, Harrell JC, Woock AE, Idowu MO, Clevenger CV
NPJ breast cancer 2021 Mar 26;7(1):37
NPJ breast cancer 2021 Mar 26;7(1):37
RAB6 GTPase regulates mammary secretory function by controlling the activation of STAT5.
Cayre S, Faraldo MM, Bardin S, Miserey-Lenkei S, Deugnier MA, Goud B
Development (Cambridge, England) 2020 Oct 8;147(19)
Development (Cambridge, England) 2020 Oct 8;147(19)
Bispecific Antibodies and Antibody-Drug Conjugates (ADCs) Bridging HER2 and Prolactin Receptor Improve Efficacy of HER2 ADCs.
Andreev J, Thambi N, Perez Bay AE, Delfino F, Martin J, Kelly MP, Kirshner JR, Rafique A, Kunz A, Nittoli T, MacDonald D, Daly C, Olson W, Thurston G
Molecular cancer therapeutics 2017 Apr;16(4):681-693
Molecular cancer therapeutics 2017 Apr;16(4):681-693
Role of Prolactin Receptors in Lymphangioleiomyomatosis.
Alkharusi A, Lesma E, Ancona S, Chiaramonte E, Nyström T, Gorio A, Norstedt G
PloS one 2016;11(1):e0146653
PloS one 2016;11(1):e0146653
Gain-of-function Prolactin Receptor Variants Are Not Associated With Breast Cancer and Multiple Fibroadenoma Risk.
Chakhtoura Z, Laki F, Bernadet M, Cherifi I, Chiche A, Pigat N, Bernichtein S, Courtillot C, Boutillon F, Bièche I, Vacher S, Tanguy ML, Bissery A, Grouthier V, Camparo P, Foretz M, Do Cruzeiro M, Pierre R, Rakotozafy F, Tichet J, Tejedor I, Guidotti JE, Sigal-Zafrani B, Goffin V, Touraine P
The Journal of clinical endocrinology and metabolism 2016 Nov;101(11):4449-4460
The Journal of clinical endocrinology and metabolism 2016 Nov;101(11):4449-4460
Stimulation of prolactin receptor induces STAT-5 phosphorylation and cellular invasion in glioblastoma multiforme.
Alkharusi A, Yu S, Landázuri N, Zadjali F, Davodi B, Nyström T, Gräslund T, Rahbar A, Norstedt G
Oncotarget 2016 Nov 29;7(48):79572-79583
Oncotarget 2016 Nov 29;7(48):79572-79583
The Role of Prolactin in Bone Metastasis and Breast Cancer Cell-Mediated Osteoclast Differentiation.
Sutherland A, Forsyth A, Cong Y, Grant L, Juan TH, Lee JK, Klimowicz A, Petrillo SK, Hu J, Chan A, Boutillon F, Goffin V, Egan C, Tang PA, Cai L, Morris D, Magliocco A, Shemanko CS
Journal of the National Cancer Institute 2016 Mar;108(3)
Journal of the National Cancer Institute 2016 Mar;108(3)
Prolactin signaling through focal adhesion complexes is amplified by stiff extracellular matrices in breast cancer cells.
Barcus CE, Keely PJ, Eliceiri KW, Schuler LA
Oncotarget 2016 Jul 26;7(30):48093-48106
Oncotarget 2016 Jul 26;7(30):48093-48106
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
Legorreta-Haquet MV, Chávez-Rueda K, Chávez-Sánchez L, Cervera-Castillo H, Zenteno-Galindo E, Barile-Fabris L, Burgos-Vargas R, Álvarez-Hernández E, Blanco-Favela F
Medicine 2016 Feb;95(5):e2384
Medicine 2016 Feb;95(5):e2384
The prolactin receptor is expressed in rheumatoid arthritis and psoriatic arthritis synovial tissue and contributes to macrophage activation.
Tang MW, Reedquist KA, Garcia S, Fernandez BM, Codullo V, Vieira-Sousa E, Goffin V, Reuwer AQ, Twickler MT, Gerlag DM, Tak PP
Rheumatology (Oxford, England) 2016 Dec;55(12):2248-2259
Rheumatology (Oxford, England) 2016 Dec;55(12):2248-2259
Dense collagen-I matrices enhance pro-tumorigenic estrogen-prolactin crosstalk in MCF-7 and T47D breast cancer cells.
Barcus CE, Holt EC, Keely PJ, Eliceiri KW, Schuler LA
PloS one 2015;10(1):e0116891
PloS one 2015;10(1):e0116891
Blockade of estrogen-stimulated proliferation by a constitutively-active prolactin receptor having lower expression in invasive ductal carcinoma.
Huang KT, Tan D, Chen KE, Walker AM
Cancer letters 2015 Mar 28;358(2):152-160
Cancer letters 2015 Mar 28;358(2):152-160
Prolactin receptor expression and breast cancer: relationships with tumor characteristics among pre- and post-menopausal women in a population-based case-control study from Poland.
Faupel-Badger JM, Duggan MA, Sherman ME, Garcia-Closas M, Yang XR, Lissowska J, Brinton LA, Peplonska B, Vonderhaar BK, Figueroa JD
Hormones & cancer 2014 Feb;5(1):42-50
Hormones & cancer 2014 Feb;5(1):42-50
Reevaluation of the proposed autocrine proliferative function of prolactin in breast cancer.
Nitze LM, Galsgaard ED, Din N, Lund VL, Rasmussen BB, Berchtold MW, Christensen L, Panina S
Breast cancer research and treatment 2013 Nov;142(1):31-44
Breast cancer research and treatment 2013 Nov;142(1):31-44
Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells.
Barcus CE, Keely PJ, Eliceiri KW, Schuler LA
The Journal of biological chemistry 2013 May 3;288(18):12722-32
The Journal of biological chemistry 2013 May 3;288(18):12722-32
Characterization of Δ7/11, a functional prolactin-binding protein.
Fleming JM, Ginsburg E, McAndrew CW, Heger CD, Cheston L, Rodriguez-Canales J, Vonderhaar BK, Goldsmith P
Journal of molecular endocrinology 2013 Feb;50(1):79-90
Journal of molecular endocrinology 2013 Feb;50(1):79-90
Functional consequences of prolactin signalling in endothelial cells: a potential link with angiogenesis in pathophysiology?
Reuwer AQ, Nowak-Sliwinska P, Mans LA, van der Loos CM, von der Thüsen JH, Twickler MT, Spek CA, Goffin V, Griffioen AW, Borensztajn KS
Journal of cellular and molecular medicine 2012 Sep;16(9):2035-48
Journal of cellular and molecular medicine 2012 Sep;16(9):2035-48
Prolactin increases survival and migration of ovarian cancer cells: importance of prolactin receptor type and therapeutic potential of S179D and G129R receptor antagonists.
Tan D, Chen KE, Khoo T, Walker AM
Cancer letters 2011 Nov 1;310(1):101-8
Cancer letters 2011 Nov 1;310(1):101-8
Functional impact of manipulation on the relative orientation of human prolactin receptor domains.
Liu W, Brooks CL
Biochemistry 2011 Jun 14;50(23):5333-44
Biochemistry 2011 Jun 14;50(23):5333-44
The prolactin receptor is expressed in macrophages within human carotid atherosclerotic plaques: a role for prolactin in atherogenesis?
Reuwer AQ, van Eijk M, Houttuijn-Bloemendaal FM, van der Loos CM, Claessen N, Teeling P, Kastelein JJ, Hamann J, Goffin V, von der Thüsen JH, Twickler MT, Aten J
The Journal of endocrinology 2011 Feb;208(2):107-17
The Journal of endocrinology 2011 Feb;208(2):107-17
Identification of gain-of-function variants of the human prolactin receptor.
Goffin V, Bogorad RL, Touraine P
Methods in enzymology 2010;484:329-55
Methods in enzymology 2010;484:329-55
Prolactin and oestrogen synergistically regulate gene expression and proliferation of breast cancer cells.
Rasmussen LM, Frederiksen KS, Din N, Galsgaard E, Christensen L, Berchtold MW, Panina S
Endocrine-related cancer 2010 Sep;17(3):809-22
Endocrine-related cancer 2010 Sep;17(3):809-22
Azaspiracid-1 inhibits endocytosis of plasma membrane proteins in epithelial cells.
Bellocci M, Sala GL, Callegari F, Rossini GP
Toxicological sciences : an official journal of the Society of Toxicology 2010 Sep;117(1):109-21
Toxicological sciences : an official journal of the Society of Toxicology 2010 Sep;117(1):109-21
Prolactin enhances insulin-like growth factor I receptor phosphorylation by decreasing its association with the tyrosine phosphatase SHP-2 in MCF-7 breast cancer cells.
Carver KC, Piazza TM, Schuler LA
The Journal of biological chemistry 2010 Mar 12;285(11):8003-12
The Journal of biological chemistry 2010 Mar 12;285(11):8003-12
Re-evaluation of the prolactin receptor expression in human breast cancer.
Galsgaard ED, Rasmussen BB, Folkesson CG, Rasmussen LM, Berchtold MW, Christensen L, Panina S
The Journal of endocrinology 2009 Apr;201(1):115-28
The Journal of endocrinology 2009 Apr;201(1):115-28
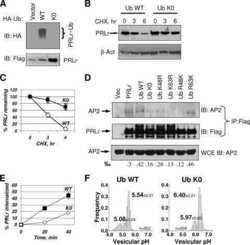
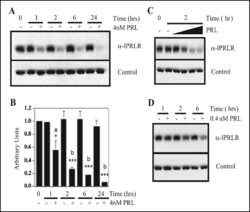
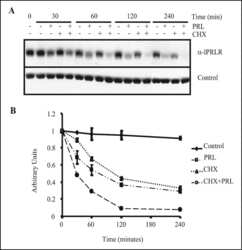
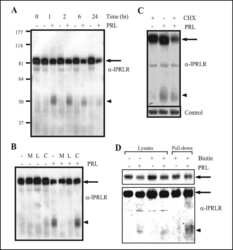
Polyubiquitination of prolactin receptor stimulates its internalization, postinternalization sorting, and degradation via the lysosomal pathway.
Varghese B, Barriere H, Carbone CJ, Banerjee A, Swaminathan G, Plotnikov A, Xu P, Peng J, Goffin V, Lukacs GL, Fuchs SY
Molecular and cellular biology 2008 Sep;28(17):5275-87
Molecular and cellular biology 2008 Sep;28(17):5275-87
Identification of a gain-of-function mutation of the prolactin receptor in women with benign breast tumors.
Bogorad RL, Courtillot C, Mestayer C, Bernichtein S, Harutyunyan L, Jomain JB, Bachelot A, Kuttenn F, Kelly PA, Goffin V, Touraine P, Benign Breast Diseases Study Group
Proceedings of the National Academy of Sciences of the United States of America 2008 Sep 23;105(38):14533-8
Proceedings of the National Academy of Sciences of the United States of America 2008 Sep 23;105(38):14533-8
Quantification of PRL/Stat5 signaling with a novel pGL4-CISH reporter.
Fang F, Antico G, Zheng J, Clevenger CV
BMC biotechnology 2008 Feb 6;8:11
BMC biotechnology 2008 Feb 6;8:11
A molecular mimic demonstrates that phosphorylated human prolactin is a potent anti-angiogenic hormone.
Ueda E, Ozerdem U, Chen YH, Yao M, Huang KT, Sun H, Martins-Green M, Bartolini P, Walker AM
Endocrine-related cancer 2006 Mar;13(1):95-111
Endocrine-related cancer 2006 Mar;13(1):95-111
Proteasomes mediate prolactin-induced receptor down-regulation and fragment generation in breast cancer cells.
Lu JC, Piazza TM, Schuler LA
The Journal of biological chemistry 2005 Oct 7;280(40):33909-16
The Journal of biological chemistry 2005 Oct 7;280(40):33909-16
Endogenous human prolactin and not exogenous human prolactin induces estrogen receptor alpha and prolactin receptor expression and increases estrogen responsiveness in breast cancer cells.
Gutzman JH, Miller KK, Schuler LA
The Journal of steroid biochemistry and molecular biology 2004 Jan;88(1):69-77
The Journal of steroid biochemistry and molecular biology 2004 Jan;88(1):69-77
Inhibition of prolactin (PRL)-induced proliferative signals in breast cancer cells by a molecular mimic of phosphorylated PRL, S179D-PRL.
Schroeder MD, Brockman JL, Walker AM, Schuler LA
Endocrinology 2003 Dec;144(12):5300-7
Endocrinology 2003 Dec;144(12):5300-7
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
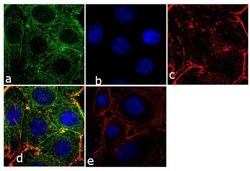
- Immunofluorescence analysis of Prolactin Receptor was performed using 70% confluent log phase SW40 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Prolactin Receptor (ECD) (1A2B1) Monoclonal Antibody (Product # 35-9200) at 2µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjµgate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
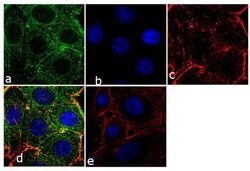
- Immunofluorescence analysis of Prolactin Receptor was performed using 70% confluent log phase SW40 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Prolactin Receptor (ECD) (1A2B1) Monoclonal Antibody (Product # 35-9200) at 2µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjµgate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
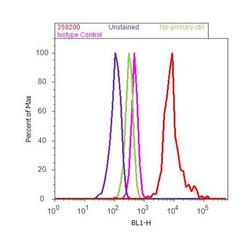
- Flow cytometry analysis of Prolactin Receptor (ECD) was done on MCF7 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Prolactin Receptor (ECD) Mouse Monoclonal Antibody (Product # 35-9200, red histogram) or with mouse isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (Product # A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control..
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
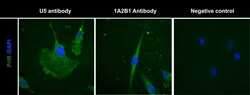
- Fig 3 Detection of PrlR in human LAM cells using immunofluorescence. Staining LAM/TSC cells using different PrlR antibodies detect a significant immunofluorescence signal in LAM/TSC cells. The antibodies employed were U5 (Thermoscientific) and A12B1 (Invitrogen).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
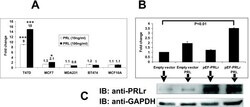
- Figure 3 Expression of pGL4-CISH to PRL and PRLr in different human breast cancer and epithelial cell lines. Cells were transfected with pGL4-CISH. After transfection, cells were arrested in minimal defined medium without FBS, the transfectants were stimulated with PRL for 24 hours prior to analysis of luminescence. A. Cells lines were transfected with 50 ng pGL4-CISH/1 x 10 5 cells. B. MCF7 cells were overexpressed with pEF-PRLr and treated with PRL (200 ng/ml). *, P < 0.05. ***, P < 0.001. C. The low inset is Western blot demonstrating PRLr overexpression level obtained with the various transfectants with the arrows indicating each lane of treatments. Error bars represent SEM; * denotes P < 0.05, and *** denotes P < 0.001 as compared with no PRL stimulation alone.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
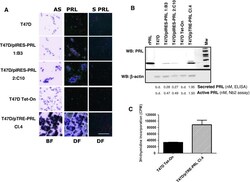
- Fig. 2 Establishment of T47D/PRL clones with high levels of ectopic PRL expression a ISH analysis of PRL mRNA expression in wild type and PRL-transfected cell lines. Bright field (BF) and dark field (DF) pictures are shown. Sections of paraffin-embedded cells were hybridized with an antisense cRNA probe (507 bp) complementary to the PRL mRNA (AS PRL). Signal is visualised under DF as white grains . BF represents the haematoxylin and eosin stained sections after hybridization with AS PRL probe to show morphology of the cells. Under BF ISH signal is visualised as black grains . Lack of detectable hybridization signal with sense probe (S PRL) indicates specificity of signals obtained with the PRL AS probe. Sections were developed after 2 weeks of exposure. Scale bars = 100 mum. b Analysis of the PRL protein expression. Cells were grown to 70-80 % confluence in 10 % FCS medium and then starved for 24 h in serum-free DMEM medium. 20 mug of cellular protein extracts were analysed by Western blotting using a PRL-specific antibody ( top panel ); beta-actin was used as protein loading control ( lower panel ). Supernatants were analysed for secreted and active PRL using ELISA and Nb2 proliferation, respectively. The PRL level is stated as nmoles/1 mio cells/24 h. b.d. below detection limit. c Transwell co-culture of T47D and T47D/PRL cells. T47D Tet-On cells were seeded in 24-well multi-dishes in 10 % CSS medium. After 24 h of plating, inserts with either T47D Tet-On or T47D/pTRE-PRL C
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
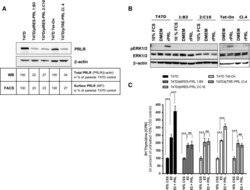
- Experimental details
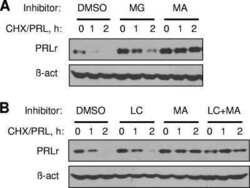
- Fig. 4 Ectopically expressed PRL down-regulates PRLR expression and responsiveness to exogenous rPRL stimulation a PRLR protein expression in T47D/PRL cells. Cells were harvested at 70-80 % confluence and 40 mug protein extracts were analysed by Western blotting using an anti-human PRLR antibody. beta-actin was used as protein loading control. The signals of PRLR were quantified and normalised by the beta-actin signals. The numbers represent a percentage of the respective control T47D cell line. Expression of PRLR on the surface of the cells was analysed by flow cytometry (FACS). Cells were stained with an anti-human PRLR mAb followed by anti-mouse IgG-APC. The median fluorescent intensities (MFI) are shown. b Cell signalling induced by rPRL. Cells were grown to 70-80 % confluence in 10 % FCS medium and starved in serum-free DMEM medium for 24 h prior to treatment with 20 nM rPRL for 30 min. 20 mug of protein extracts were analysed for activation of the ERK1/2 pathway by Western blotting using an anti-pERK1/2 antibody. Membranes were stripped and sequentially re-probed with antibodies detecting total ERK1/2 protein and beta-actin as loading controls. c Proliferation of T47D/PRL cells upon E2 and PRL exposure. Cells were seeded with a density of 1.2 x 10 4 cells/well in 96-well multi-dishes in 10 % CSS medium. Next day, medium was renewed and cells were cultured for three days in absence or presence of 1 nM 17beta-estradiol (E2) and 20 nM rPRL as indicated. Cell proliferation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
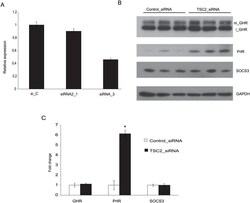
- Experimental details
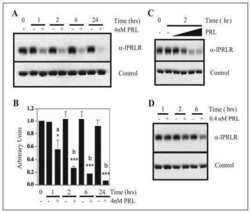
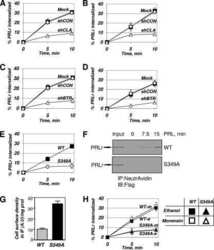
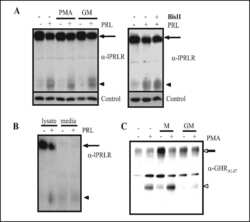
- Fig 1 Effect of TSC2 siRNA on GHR and PrlR expression. The mouse cell line CRL-2620 was transfected with two different TSC2 siRNAs (siRNA_1 and siRNA_3), or a control siRNA (si_C). (A) Quantitative RT-PCR was used to test the efficacy with which these siRNAs down-regulated TSC2 mRNA. The most pronounced effect (approximately 60% reduction) was seen with TSC2 siRNA_3, which was used for further experiments. (B) The figure shows a representative Western blot probed with antibodies directed to GHR, PrlR, SOCS3 and GAPDH; iGHR and mGHR, are the immature and mature forms of GHR, respectively. (C) Densitometric analyses of Western blots. Protein lysates were prepared individually from three separate culture dishes treated with TSC2 siRNA or control siRNA and subject to Western blot analysis; experiments were repeated three times and gave consistent results. * = P-value
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
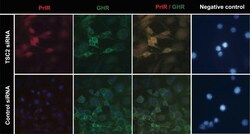
- Experimental details
- Fig 2 Immunofluorescence of CRL-2620 using PrlR and GHR antibodies. Cells were transfected with TSC2 or control siRNAs. After over-night incubation, the cells were washed 3 times with PBS and incubated with antibodies for PrlR and GHR. The figures show that PrlR immunostaining was markedly increased following TSC2 knock-down. A strong immunofluorescence signal was observed at intracellular locations. In the negative control panel, cells were incubated with mouse IgG and rabbit IgG antibodies. Fluorescence images were acquired at a magnification of 63x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
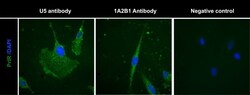
- Experimental details
- Fig 3 Detection of PrlR in human LAM cells using immunofluorescence. Staining LAM/TSC cells using different PrlR antibodies detect a significant immunofluorescence signal in LAM/TSC cells. The antibodies employed were U5 (Thermoscientific) and A12B1 (Invitrogen).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
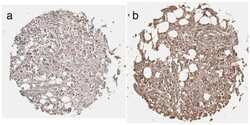
- Fig 4 Prolactin receptor immuno reactivity in human LAM lung lesions. Histological sections from LAM patients were analyzed. Human lung tissues were obtained from subjects who underwent lung resection for diagnostic reasons and immunohistochemical-staining using the PrlR antibody clone 1A2B1 (Invitrogen) was carried out. Arrows indicate the location of PrlR immuno reactivity in one microscope field of a representative image from LAM tissue; compared to positive cells for PrlR in control lung tissue. For negative control, primary antibody was omitted, Scale bars: 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
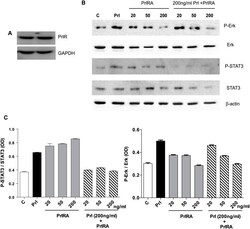
- Fig 5 Human LAM cells express PrlR, and prolactin stimulates phosphorylation of STAT3 and Erk. LAM/TSC cells were cultured in serum-free medium overnight. The cells were then exposed to different doses of PrlRA (20, 50, 200 ng/ml) for 15 minutes, and subsequently the cells were exposed to 200 ng/ml Prl for 60 minutes or PBS as a control. Then protein extracts were prepared for Western blot analysis, and probed with antibodies for P-STAT3, STAT3, P-Erk and Erk. (A) Western blot to analyze PrlR in LAM/TSC control cells; an antibody against human PrlR detected a protein band of 89-90 kD. (B) Western blot, using antibodies directed against P-STAT3, STAT3, P-Erk and Erk in LAM/TSC cells following treatment as described in Material and methods section. (C) Densitometric quantification of Western blot signals, in which the Y-axis depicts the ratio between phosphorylated STAT3 and Erk to the total protein.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
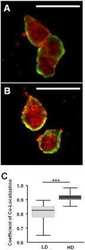
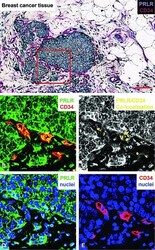
- Fig 6 Immunohistochemical staining of the prolactin receptor and CD34 in mammary carcinoma. (A) Staining of the prolactin receptor (turquoise) and CD34 (endothelial cells, purple), counterstaining with haematoxylin (blue). The pictures were digitally processed to unmix the different colours. (B) Fluorescence-like image of both prolactin receptor (green) and CD34 (red) in pseudo-colours. (C) Areas where prolactin receptor and CD34 co-localize, and endothelial expression of prolactin receptor. (D) and (E), respectively, show pseudo-fluorescent staining for the prolactin receptor (green), endothelial cells (red) and nuclei (blue). Scale bar = 0.075 mum in (A-E).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
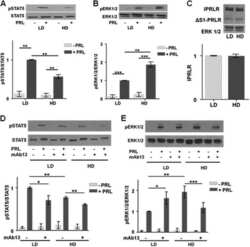
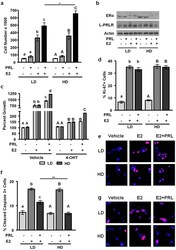
- Figure 4 High density/ stiff collagen matrices enhance E2 and PRL induced cell growth, and inhibit apoptosis in T47D cells. a) T47D cells were cultured in LD or HD collagen and treated +- 1nM E2, +- 4nM PRL for 7d. Cells were removed from gels and counted via hemocytometer. n = 4. b) T47D cells were cultured in LD or HD collagen and treated +- 1nM E2, +- 4nM PRL for 72h and lysates were immunoblotted with the indicated antibodies. c) T47D cells were cultured as in (a) , and treated +- 4-OHT 1h prior to hormone treatments and quantitation of cell number. n = 4. d,e,f,g) T47D cells were cultured as in (b) , fixed and stained for DAPI and Ki-67 (d,e) or cleaved caspase 3 (f,g) , and quantified as described in the Materials and Methods. n = 3. Representative images of DAPI/ Ki67 are shown in (e) and DAPI/ cleaved caspase 3 are shown in (g). Different letters represent significant differences (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
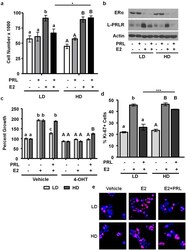
- Figure 3 E2 and PRL co-treatment inhibits MCF-7 cell growth and proliferation in low density/ compliant but not high density/ stiff collagen matrices. a) MCF-7 cells were cultured in LD or HD collagen and treated +- E2, +- PRL for 72h. Cells were removed from gels and counted. n = 3. b) MCF-7 cells were cultured and treated as in (a) and lysates were immunoblotted with the indicated antibodies. Representative blots shown. c) MCF-7 cells were cultured as in (a) and treated +- 4-OHT 1h prior to hormone treatment. n = 4. d,e) MCF-7 cells were cultured and treated as in (a) . Gels were fixed and stained for DAPI and Ki-67, and quantified as described in the Materials and Methods (d). n = 3. e) Representative images of DAPI/ Ki-67 are shown in (d). Different letters represent significant differences (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
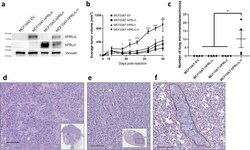
- Fig. 1 MCF10AT hPRLrL + I co-expression in vivo. a MCF10AT cells were stably transfected with either hPRLrL, hPRLrI, both isoforms together (at a 1:1 ratio), or empty vector (MCF10AT-hPRLrL, MCF10AT-hPRLrI, MCF10AT-hPRLrL+I, and MCF10AT-EV, respectively), and expression was confirmed by IB. b These cells were orthotopically xenografted into female NSG mice, and primary tumor growth was monitored longitudinally c Lungs were harvested and micrometastatic burden was counted manually. Representative images (scale bar, 100 um) of H&E stained d primary tumor, e axillary lymph node and f lung indicate primary tumors to be high grade adenocarcinoma with nodal infiltration and successful distant metastatic dissemination (outlined; representative image shows metastatic growth long a blood vessel). * p < 0.05, ** p < 0.01, *** p < 0.005. n = 4 mice/study arm. Data is shown as mean +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
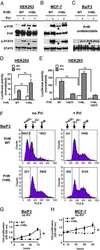
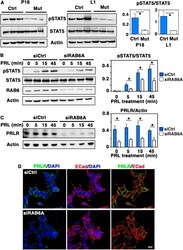
- Fig. 6. Decreased STAT5 activation in Rab6a -deficient mammary glands and RAB6A-depleted T47-D cells. (A) Western blots for pSTAT5, total STAT5 and actin performed on three distinct control and mutant mammary gland protein extracts at P18 and L1 (left). Quantification of pSTAT5/STAT5 amounts (mean+-s.e.m.) (right). * P =0.038 (P18), * P =0.041 (L1). (B) Western blots for pSTAT5, total STAT5, RAB6 and actin performed with T47-D cell lysates (left). SiCtrl and SiRAB6A cells were stimulated with PRL for 5, 15 and 45 min. Quantification of pSTAT5/STAT5 amount at each time point (right). Data are mean+-s.e.m. of four separate siRNA assays. *0.01< P
 Explore
Explore Validate
Validate Learn
Learn