MA1-81381
antibody from Invitrogen Antibodies
Targeting: S100A9
60B8AG, CAGB, CFAG, CGLB, LIAG, MAC387, MIF, MRP14, NIF, P14
Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Immunocytochemistry [6]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-81381 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Calprotectin Monoclonal Antibody (MAC387)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This clone recognizes the L1 or Calprotectin molecule, an intracytoplasmic antigen comprised of a 12 kDa alpha chain and a 14 kDa beta chain. Although originally described as binding to epitopes common to both the alpha and beta chains (Flavell et al. 1987) subsequent studies indicate that the antibody detects an epitope exclusively expressed on the beta chain (Goebeler et al. 1994) demonstrated by immunofluorescent and western blotting on both naturally expressing and transfected targets. In addition Mouse anti Human macrophages, clone MAC387 detects the beta chain in complex with the alpha. The antigen recognized by Mouse anti Human macrophages, clone MAC387 is expressed by granulocytes, monocytes and by tissue macrophages. Variable results have been reported for staining brain macrophages and microglia. The epitope recognized appears to be well conserved and the antibody is routinely used for the detection of myeloid cellds in a wide range of species. This product requires protein digestion pre-treatment of paraffin sections using trypsin or pronase prior to IHC staining. A suggested positive control is human spleen. For flow cytometry, use 10 µL of the suggested working dilution to label 1x10^6 cells in 100 µL.
- Reactivity
- Human, Rat, Bovine, Canine, Feline, Goat, Guinea Pig, Porcine, Rabbit
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- MAC387
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
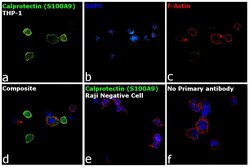
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-80155) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
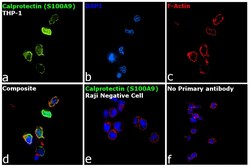
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-80446) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
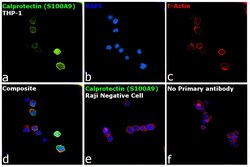
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-81381) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
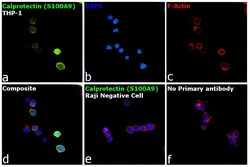
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-81381) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
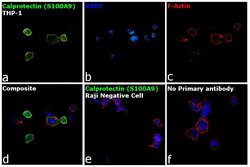
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-80155) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
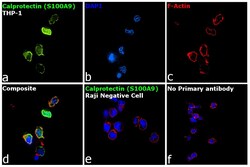
- Experimental details
- Immunofluorescence analysis of Calprotectin (S100A9) was performed using 70% confluent log phase THP-1 and Raji cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Calprotectin (S100A9) Monoclonal Antibody (MAC387) (Product # MA1-80446) at 1:250 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of THP-1 showing localization to plasma membrane, nucleus and cytoplasm. Panel e shows Raji cells with no expression of Calprotectin (S100A9). Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
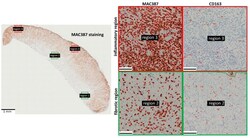
- Experimental details
- Figure 3 Immunohistochemistry showing the spatial organization of MAC387+ cells and CD163+ cells in horse EGT. The majority of the MAC387 staining was found in the inflammatory regions (indicated in red) near the wound surface. The CD163 staining was more uniformly distributed. Stained pixels were detected using the positive pixel count algorithm in QuPath and encircled in red. Scale bar inserts = 100 um. Red frame: inflammatory regions; green frame: fibrotic regions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
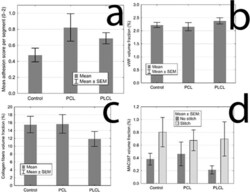
- Experimental details
- Figure 6 Statistical analysis of quantitative assessment of different parameters: (a ) The mean adhesion score for each group, the control group scored the lowest with no statistical significance; ( b ) The volume fractions of vWF positively stained area for each groups showing the level of vascularisation, the three groups show the same quality of scar in this aspect; ( c ) The volume fractions of collagen fibres for each group, the three groups show the same quality of scar in this aspect; ( d ) The volume fractions of MAC387 positive area for each group, showing the inflammatory cells infiltration, the presence of a stitch in the section proves to be the only statistically significant factor, the three groups show the same quality of scar in this aspect as well.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
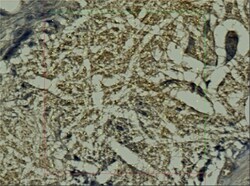
- Experimental details
- Figure 7 Histological section, PCL group, MAC387 staining: Detail of the marginal zone of the material applied, the empty spaces in the shape of the fibres (stereological grid).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
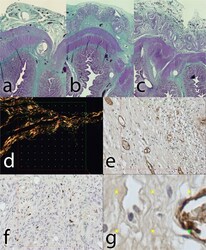
- Experimental details
- Figure 8 Histological staining of explanted anastomoses: (a ) Green trichrome: Control group; ( b ) Green trichrome: PCL group, the empty space on the site of application of the nanomaterial can be seen in the upper layer, surrounded by normal granulation tissue; (c ) Green trichrome: PLCL group, a much thinner empty area can be seen in the upper layer, also surrounded by normal granulation tissue; ( d ) PSR staining, collagen fibres stained yellow, stereological mesh; ( e ) vWF factor staining, the endothelial cells stained brown, stereological grid; ( f ) MAC 387 staining stereology, positive cells stained blue, stereological grid; ( g ) magnification of vWF staining stereology with a positive cross in the upper right corner.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunohistochemistry
Immunohistochemistry