Antibody data
- Antibody Data
- Antigen structure
- References [45]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [25]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-0168-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD16 Monoclonal Antibody (eBioCB16 (CB16)), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The eBioCB16 monoclonal antibody recognizes CD16 (Fc gammaRIII), the low-affinity receptor for IgG with an apparent molecular weight of 50-80 kDa. CD16 is represented by two similar genes, CD16A (Fc gammaRIIIA), which exists as a hetero-oligomeric polypeptide-anchored form in macrophages and NK cells and CD16B (Fc gammaRIIIB), which exist as a monomeric GPI-anchored form in neutrophils. Furthermore, there are two known polymorphisms of CD16B, NA-1 and NA-2. Individuals homozygous for NA-2 show a lower phagocytic capacity compared with NA-1. CD16 binds IgG in the form of immune complexes and shows preferential binding of IgG1 and IgG3 isotypes and minimal binding of IgG2 and IgG4. Upon IgG binding, both CD16 isoforms initiate signal transduction cascades that lead to a variety of responses including antibody-dependent cell-mediated cytotoxicity (ADCC), phagocytosis, degranulation and proliferation. Applications Reported: This eBioCB16 (CB16) antibody has been reported for use in flow cytometric analysis. Applications Tested: This eBioCB16 (CB16) antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 520 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- eBioCB16 (CB16)
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references SARS-CoV-2 Delta (B.1.617.2) variant replicates and induces syncytia formation in human induced pluripotent stem cell-derived macrophages.
Exhaustion‑like dysfunction of T and NKT cells in an X‑linked severe combined immunodeficiency patient with maternal engraftment by single‑cell analysis.
Prognostic Lnc-S100B-2 Affects Cell Apoptosis and Microenvironment of Colorectal Cancer through MLLT10 Signaling.
Gene-dosage effect of Pfkfb3 on monocyte/macrophage biology in atherosclerosis.
Increased Levels of VCAM-1 in Sera and VLA-4 Expression on Neutrophils in Dermatomyositis with Interstitial Lung Disease.
The Composition of Adipose-Derived Regenerative Cells Isolated from Lipoaspirate Using a Point of Care System Does Not Depend on the Subject's Individual Age, Sex, Body Mass Index and Ethnicity.
-------A type I IFN, prothrombotic hyperinflammatory neutrophil signature is distinct for COVID-19 ARDS--.
Associations between Hypertriglyceridemia and Circulating Neutrophil Subpopulation in Patients with Dyslipidemia.
High-Molecular-Weight Hyaluronic Acid Inhibits IL-1β-Induced Synovial Inflammation and Macrophage Polarization through the GRP78-NF-κB Signaling Pathway.
Broad and potent neutralizing human antibodies to tick-borne flaviviruses protect mice from disease.
Ex vivo expansion of dysfunctional regulatory T lymphocytes restores suppressive function in Parkinson's disease.
Heterogeneous disease-propagating stem cells in juvenile myelomonocytic leukemia.
Isolation of polymorphonuclear neutrophils and monocytes from a single sample of human peripheral blood.
CD14(+)CD16(-) monocytes are the main precursors of osteoclasts in rheumatoid arthritis via expressing Tyro3TK.
Reprogramming of bone marrow myeloid progenitor cells in patients with severe coronary artery disease.
Loss of testosterone impairs anti-tumor neutrophil function.
Human Mast Cell Proteome Reveals Unique Lineage, Putative Functions, and Structural Basis for Cell Ablation.
SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology.
A FACS-based approach to obtain viable eosinophils from human adipose tissue.
IL-36 Promotes Systemic IFN-I Responses in Severe Forms of Psoriasis.
Effector memory differentiation increases detection of replication-competent HIV-l in resting CD4+ T cells from virally suppressed individuals.
A child with perinatal HIV infection and long-term sustained virological control following antiretroviral treatment cessation.
Bioreactor-based mass production of human iPSC-derived macrophages enables immunotherapies against bacterial airway infections.
NF-κB/MAPK activation underlies ACVR1-mediated inflammation in human heterotopic ossification.
Molecular signatures associated with tumor-specific immune response in melanoma patients treated with dendritic cell-based immunotherapy.
Human lactoferrin attenuates the proinflammatory response of neonatal monocyte-derived macrophages.
Innate and adaptive immune dysregulation in critically ill ICU patients.
The Influence of Cytomegalovirus on Expression of HLA-G and its Ligand KIR2DL4 by Human Peripheral Blood Leucocyte Subsets.
Metformin-treated cancer cells modulate macrophage polarization through AMPK-NF-κB signaling.
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
A cluster of immunoresolvents links coagulation to innate host defense in human blood.
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Eomeshi NK Cells in Human Liver Are Long-Lived and Do Not Recirculate but Can Be Replenished from the Circulation.
A DNA vaccine encoding mutated HPV58 mE6E7-Fc-GPI fusion antigen and GM-CSF and B7.1.
Hyperreactive onchocerciasis is characterized by a combination of Th17-Th2 immune responses and reduced regulatory T cells.
NK cell immunesenescence is increased by psychological but not physical stress in older adults associated with raised cortisol and reduced perforin expression.
Retinoic acid regulates hematopoietic development from human pluripotent stem cells.
Depressive symptoms in hip fracture patients are associated with reduced monocyte superoxide production.
Decreased plasma levels of soluble CD18 link leukocyte infiltration with disease activity in spondyloarthritis.
Human natural killer cell maturation defect supports in vivo CD56(bright) to CD56(dim) lineage development.
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Changes in natural killer cell subsets in pediatric liver transplant recipients.
CpG and non-CpG oligodeoxynucleotides directly costimulate mouse and human CD4+ T cells through a TLR9- and MyD88-independent mechanism.
Systemic hypoxia enhances exercise-mediated bactericidal and subsequent apoptotic responses in human neutrophils.
Systemic hypoxia enhances bactericidal activities of human polymorphonuclear leuocytes.
Thaweerattanasinp T, Wanitchang A, Saenboonrueng J, Srisutthisamphan K, Wanasen N, Sungsuwan S, Jongkaewwattana A, Chailangkarn T
PeerJ 2023;11:e14918
PeerJ 2023;11:e14918
Exhaustion‑like dysfunction of T and NKT cells in an X‑linked severe combined immunodeficiency patient with maternal engraftment by single‑cell analysis.
Dong W, Li W, Zhang S, Zeng X, Qin Q, Fan H, Tang Z, Wu X, Lu G
International journal of molecular medicine 2023 Mar;51(3)
International journal of molecular medicine 2023 Mar;51(3)
Prognostic Lnc-S100B-2 Affects Cell Apoptosis and Microenvironment of Colorectal Cancer through MLLT10 Signaling.
Yi J, Peng F, Zhao J, Gong X
Journal of oncology 2022;2022:3565118
Journal of oncology 2022;2022:3565118
Gene-dosage effect of Pfkfb3 on monocyte/macrophage biology in atherosclerosis.
Guo S, Li A, Fu X, Li Z, Cao K, Song M, Huang S, Li Z, Yan J, Wang L, Dai X, Feng D, Wang Y, He J, Huo Y, Xu Y
British journal of pharmacology 2022 Nov;179(21):4974-4991
British journal of pharmacology 2022 Nov;179(21):4974-4991
Increased Levels of VCAM-1 in Sera and VLA-4 Expression on Neutrophils in Dermatomyositis with Interstitial Lung Disease.
Lin M, Yang C, Liu X, Zhao S, Tian B, Hou X, Xu J, Yang P
Immunological investigations 2022 May;51(4):980-992
Immunological investigations 2022 May;51(4):980-992
The Composition of Adipose-Derived Regenerative Cells Isolated from Lipoaspirate Using a Point of Care System Does Not Depend on the Subject's Individual Age, Sex, Body Mass Index and Ethnicity.
Schmitz C, Alt C, Azares AR, Pearce DA, Facile TR, Furia JP, Maffulli N, Huang C, Alt EU
Cells 2022 Dec 21;12(1)
Cells 2022 Dec 21;12(1)
-------A type I IFN, prothrombotic hyperinflammatory neutrophil signature is distinct for COVID-19 ARDS--.
Reyes L, A Sanchez-Garcia M, Morrison T, Howden AJM, Watts ER, Arienti S, Sadiku P, Coelho P, Mirchandani AS, Zhang A, Hope D, Clark SK, Singleton J, Johnston S, Grecian R, Poon A, McNamara S, Harper I, Fourman MH, Brenes AJ, Pathak S, Lloyd A, Blanco GR, von Kriegsheim A, Ghesquiere B, Vermaelen W, Cologna CT, Dhaliwal K, Hirani N, Dockrell DH, Whyte MKB, Griffith D, Cantrell DA, Walmsley SR
Wellcome open research 2021;6:38
Wellcome open research 2021;6:38
Associations between Hypertriglyceridemia and Circulating Neutrophil Subpopulation in Patients with Dyslipidemia.
Genkel V, Dolgushin I, Baturina I, Savochkina A, Kuznetsova A, Pykhova L, Shaposhnik I
International journal of inflammation 2021;2021:6695468
International journal of inflammation 2021;2021:6695468
High-Molecular-Weight Hyaluronic Acid Inhibits IL-1β-Induced Synovial Inflammation and Macrophage Polarization through the GRP78-NF-κB Signaling Pathway.
Lee CH, Chiang CF, Kuo FC, Su SC, Huang CL, Liu JS, Lu CH, Hsieh CH, Wang CC, Lee CH, Shen PH
International journal of molecular sciences 2021 Nov 3;22(21)
International journal of molecular sciences 2021 Nov 3;22(21)
Broad and potent neutralizing human antibodies to tick-borne flaviviruses protect mice from disease.
Agudelo M, Palus M, Keeffe JR, Bianchini F, Svoboda P, Salát J, Peace A, Gazumyan A, Cipolla M, Kapoor T, Guidetti F, Yao KH, Elsterová J, Teislerová D, Chrdle A, Hönig V, Oliveira T, West AP, Lee YE, Rice CM, MacDonald MR, Bjorkman PJ, Růžek D, Robbiani DF, Nussenzweig MC
The Journal of experimental medicine 2021 May 3;218(5)
The Journal of experimental medicine 2021 May 3;218(5)
Ex vivo expansion of dysfunctional regulatory T lymphocytes restores suppressive function in Parkinson's disease.
Thome AD, Atassi F, Wang J, Faridar A, Zhao W, Thonhoff JR, Beers DR, Lai EC, Appel SH
NPJ Parkinson's disease 2021 May 13;7(1):41
NPJ Parkinson's disease 2021 May 13;7(1):41
Heterogeneous disease-propagating stem cells in juvenile myelomonocytic leukemia.
Louka E, Povinelli B, Rodriguez-Meira A, Buck G, Wen WX, Wang G, Sousos N, Ashley N, Hamblin A, Booth CAG, Roy A, Elliott N, Iskander D, de la Fuente J, Fordham N, O'Byrne S, Inglott S, Norfo R, Salio M, Thongjuea S, Rao A, Roberts I, Mead AJ
The Journal of experimental medicine 2021 Feb 1;218(2)
The Journal of experimental medicine 2021 Feb 1;218(2)
Isolation of polymorphonuclear neutrophils and monocytes from a single sample of human peripheral blood.
Cui C, Schoenfelt KQ, Becker KM, Becker L
STAR protocols 2021 Dec 17;2(4):100845
STAR protocols 2021 Dec 17;2(4):100845
CD14(+)CD16(-) monocytes are the main precursors of osteoclasts in rheumatoid arthritis via expressing Tyro3TK.
Xue J, Xu L, Zhu H, Bai M, Li X, Zhao Z, Zhong H, Cheng G, Li X, Hu F, Su Y
Arthritis research & therapy 2020 Sep 21;22(1):221
Arthritis research & therapy 2020 Sep 21;22(1):221
Reprogramming of bone marrow myeloid progenitor cells in patients with severe coronary artery disease.
Noz MP, Bekkering S, Groh L, Nielen TM, Lamfers EJ, Schlitzer A, El Messaoudi S, van Royen N, Huys EH, Preijers FW, Smeets EM, Aarntzen EH, Zhang B, Li Y, Bremmers ME, van der Velden WJ, Dolstra H, Joosten LA, Gomes ME, Netea MG, Riksen NP
eLife 2020 Nov 10;9
eLife 2020 Nov 10;9
Loss of testosterone impairs anti-tumor neutrophil function.
Markman JL, Porritt RA, Wakita D, Lane ME, Martinon D, Noval Rivas M, Luu M, Posadas EM, Crother TR, Arditi M
Nature communications 2020 Mar 31;11(1):1613
Nature communications 2020 Mar 31;11(1):1613
Human Mast Cell Proteome Reveals Unique Lineage, Putative Functions, and Structural Basis for Cell Ablation.
Plum T, Wang X, Rettel M, Krijgsveld J, Feyerabend TB, Rodewald HR
Immunity 2020 Feb 18;52(2):404-416.e5
Immunity 2020 Feb 18;52(2):404-416.e5
SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology.
Veras FP, Pontelli MC, Silva CM, Toller-Kawahisa JE, de Lima M, Nascimento DC, Schneider AH, Caetité D, Tavares LA, Paiva IM, Rosales R, Colón D, Martins R, Castro IA, Almeida GM, Lopes MIF, Benatti MN, Bonjorno LP, Giannini MC, Luppino-Assad R, Almeida SL, Vilar F, Santana R, Bollela VR, Auxiliadora-Martins M, Borges M, Miranda CH, Pazin-Filho A, da Silva LLP, Cunha LD, Zamboni DS, Dal-Pizzol F, Leiria LO, Siyuan L, Batah S, Fabro A, Mauad T, Dolhnikoff M, Duarte-Neto A, Saldiva P, Cunha TM, Alves-Filho JC, Arruda E, Louzada-Junior P, Oliveira RD, Cunha FQ
The Journal of experimental medicine 2020 Dec 7;217(12)
The Journal of experimental medicine 2020 Dec 7;217(12)
A FACS-based approach to obtain viable eosinophils from human adipose tissue.
Hernandez JD, Tew BY, Li T, Gooden GC, Ghannam H, Masuda M, Madura J 2nd, Salhia B, Jacobsen EA, De Filippis E
Scientific reports 2020 Aug 6;10(1):13210
Scientific reports 2020 Aug 6;10(1):13210
IL-36 Promotes Systemic IFN-I Responses in Severe Forms of Psoriasis.
Catapano M, Vergnano M, Romano M, Mahil SK, Choon SE, Burden AD, Young HS, Carr IM, Lachmann HJ, Lombardi G, Smith CH, Ciccarelli FD, Barker JN, Capon F
The Journal of investigative dermatology 2020 Apr;140(4):816-826.e3
The Journal of investigative dermatology 2020 Apr;140(4):816-826.e3
Effector memory differentiation increases detection of replication-competent HIV-l in resting CD4+ T cells from virally suppressed individuals.
Wonderlich ER, Subramanian K, Cox B, Wiegand A, Lackman-Smith C, Bale MJ, Stone M, Hoh R, Kearney MF, Maldarelli F, Deeks SG, Busch MP, Ptak RG, Kulpa DA
PLoS pathogens 2019 Oct;15(10):e1008074
PLoS pathogens 2019 Oct;15(10):e1008074
A child with perinatal HIV infection and long-term sustained virological control following antiretroviral treatment cessation.
Violari A, Cotton MF, Kuhn L, Schramm DB, Paximadis M, Loubser S, Shalekoff S, Da Costa Dias B, Otwombe K, Liberty A, McIntyre J, Babiker A, Gibb D, Tiemessen CT
Nature communications 2019 Jan 24;10(1):412
Nature communications 2019 Jan 24;10(1):412
Bioreactor-based mass production of human iPSC-derived macrophages enables immunotherapies against bacterial airway infections.
Ackermann M, Kempf H, Hetzel M, Hesse C, Hashtchin AR, Brinkert K, Schott JW, Haake K, Kühnel MP, Glage S, Figueiredo C, Jonigk D, Sewald K, Schambach A, Wronski S, Moritz T, Martin U, Zweigerdt R, Munder A, Lachmann N
Nature communications 2018 Nov 30;9(1):5088
Nature communications 2018 Nov 30;9(1):5088
NF-κB/MAPK activation underlies ACVR1-mediated inflammation in human heterotopic ossification.
Barruet E, Morales BM, Cain CJ, Ton AN, Wentworth KL, Chan TV, Moody TA, Haks MC, Ottenhoff TH, Hellman J, Nakamura MC, Hsiao EC
JCI insight 2018 Nov 15;3(22)
JCI insight 2018 Nov 15;3(22)
Molecular signatures associated with tumor-specific immune response in melanoma patients treated with dendritic cell-based immunotherapy.
García-Salum T, Villablanca A, Matthäus F, Tittarelli A, Baeza M, Pereda C, Gleisner MA, González FE, López MN, Hoheisel JD, Norgauer J, Gebicke-Haerter PJ, Salazar-Onfray F
Oncotarget 2018 Mar 30;9(24):17014-17027
Oncotarget 2018 Mar 30;9(24):17014-17027
Human lactoferrin attenuates the proinflammatory response of neonatal monocyte-derived macrophages.
Wisgrill L, Wessely I, Spittler A, Förster-Waldl E, Berger A, Sadeghi K
Clinical and experimental immunology 2018 Jun;192(3):315-324
Clinical and experimental immunology 2018 Jun;192(3):315-324
Innate and adaptive immune dysregulation in critically ill ICU patients.
Duggal NA, Snelson C, Shaheen U, Pearce V, Lord JM
Scientific reports 2018 Jul 5;8(1):10186
Scientific reports 2018 Jul 5;8(1):10186
The Influence of Cytomegalovirus on Expression of HLA-G and its Ligand KIR2DL4 by Human Peripheral Blood Leucocyte Subsets.
Albayati Z, Alyami A, Alomar S, Middleton D, Bonnett L, Aleem S, Flanagan BF, Christmas SE
Scandinavian journal of immunology 2017 Nov;86(5):396-407
Scandinavian journal of immunology 2017 Nov;86(5):396-407
Metformin-treated cancer cells modulate macrophage polarization through AMPK-NF-κB signaling.
Chiang CF, Chao TT, Su YF, Hsu CC, Chien CY, Chiu KC, Shiah SG, Lee CH, Liu SY, Shieh YS
Oncotarget 2017 Mar 28;8(13):20706-20718
Oncotarget 2017 Mar 28;8(13):20706-20718
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
Lebedeva A, Vorobyeva D, Vagida M, Ivanova O, Felker E, Fitzgerald W, Danilova N, Gontarenko V, Shpektor A, Vasilieva E, Margolis L
Atherosclerosis 2017 Dec;267:90-98
Atherosclerosis 2017 Dec;267:90-98
A cluster of immunoresolvents links coagulation to innate host defense in human blood.
Norris PC, Libreros S, Chiang N, Serhan CN
Science signaling 2017 Aug 1;10(490)
Science signaling 2017 Aug 1;10(490)
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Sakthivel P, Grunewald J, Eklund A, Bruder D, Wahlström J
Clinical and experimental immunology 2016 Feb;183(2):294-306
Clinical and experimental immunology 2016 Feb;183(2):294-306
Eomeshi NK Cells in Human Liver Are Long-Lived and Do Not Recirculate but Can Be Replenished from the Circulation.
Cuff AO, Robertson FP, Stegmann KA, Pallett LJ, Maini MK, Davidson BR, Male V
Journal of immunology (Baltimore, Md. : 1950) 2016 Dec 1;197(11):4283-4291
Journal of immunology (Baltimore, Md. : 1950) 2016 Dec 1;197(11):4283-4291
A DNA vaccine encoding mutated HPV58 mE6E7-Fc-GPI fusion antigen and GM-CSF and B7.1.
Wang H, Yu J, Li L
OncoTargets and therapy 2015;8:3067-77
OncoTargets and therapy 2015;8:3067-77
Hyperreactive onchocerciasis is characterized by a combination of Th17-Th2 immune responses and reduced regulatory T cells.
Katawa G, Layland LE, Debrah AY, von Horn C, Batsa L, Kwarteng A, Arriens S, W Taylor D, Specht S, Hoerauf A, Adjobimey T
PLoS neglected tropical diseases 2015 Jan;9(1):e3414
PLoS neglected tropical diseases 2015 Jan;9(1):e3414
NK cell immunesenescence is increased by psychological but not physical stress in older adults associated with raised cortisol and reduced perforin expression.
Duggal NA, Upton J, Phillips AC, Hampson P, Lord JM
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9748
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9748
Retinoic acid regulates hematopoietic development from human pluripotent stem cells.
Rönn RE, Guibentif C, Moraghebi R, Chaves P, Saxena S, Garcia B, Woods NB
Stem cell reports 2015 Feb 10;4(2):269-81
Stem cell reports 2015 Feb 10;4(2):269-81
Depressive symptoms in hip fracture patients are associated with reduced monocyte superoxide production.
Duggal NA, Beswetherick A, Upton J, Hampson P, Phillips AC, Lord JM
Experimental gerontology 2014 Jun;54:27-34
Experimental gerontology 2014 Jun;54:27-34
Decreased plasma levels of soluble CD18 link leukocyte infiltration with disease activity in spondyloarthritis.
Kragstrup TW, Jalilian B, Hvid M, Kjærgaard A, Østgård R, Schiøttz-Christensen B, Jurik AG, Robinson WH, Vorup-Jensen T, Deleuran B
Arthritis research & therapy 2014 Feb 4;16(1):R42
Arthritis research & therapy 2014 Feb 4;16(1):R42
Human natural killer cell maturation defect supports in vivo CD56(bright) to CD56(dim) lineage development.
Domaica CI, Fuertes MB, Uriarte I, Girart MV, Sardañons J, Comas DI, Di Giovanni D, Gaillard MI, Bezrodnik L, Zwirner NW
PloS one 2012;7(12):e51677
PloS one 2012;7(12):e51677
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.
Riek AE, Oh J, Sprague JE, Timpson A, de las Fuentes L, Bernal-Mizrachi L, Schechtman KB, Bernal-Mizrachi C
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
The Journal of biological chemistry 2012 Nov 9;287(46):38482-94
Changes in natural killer cell subsets in pediatric liver transplant recipients.
Pham B, Piard-Ruster K, Silva R, Gallo A, Esquivel CO, Martinez OM, Krams SM
Pediatric transplantation 2012 Mar;16(2):176-82
Pediatric transplantation 2012 Mar;16(2):176-82
CpG and non-CpG oligodeoxynucleotides directly costimulate mouse and human CD4+ T cells through a TLR9- and MyD88-independent mechanism.
Landrigan A, Wong MT, Utz PJ
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 15;187(6):3033-43
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 15;187(6):3033-43
Systemic hypoxia enhances exercise-mediated bactericidal and subsequent apoptotic responses in human neutrophils.
Wang JS, Chiu YT
Journal of applied physiology (Bethesda, Md. : 1985) 2009 Oct;107(4):1213-22
Journal of applied physiology (Bethesda, Md. : 1985) 2009 Oct;107(4):1213-22
Systemic hypoxia enhances bactericidal activities of human polymorphonuclear leuocytes.
Wang JS, Liu HC
Clinical science (London, England : 1979) 2009 May 1;116(11):805-17
Clinical science (London, England : 1979) 2009 May 1;116(11):805-17
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
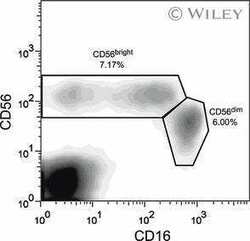
- Staining of normal human peripheral blood cells with Anti-Human CD56 (NCAM) PE (Product # 12-0567-42) and Mouse IgG1 K Isotype Control FITC (Product # 11-4714-42) (left) or Anti-Human CD16 FITC (right). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 The IL-36 receptor is preferentially expressed by plasmacytoid dendritic cell s. ( a-e ) Representative flow cytometry plots showing IL36R surface expression compared with fluorescence minus one control. ( a ) neutrophils (gated as CD14 + , CD15 + , CD16 + cells); ( b ) innate lymphoid cells (lineage - [CD3 - , CD4 - , CD19 - , CD20 - , CD56 - ], CD127 + ); ( c ) monocytes (CD3 - , CD20 - , CD19 - , CD56 - ) separated into classical (CD16 - , CD14 high ), intermediate (CD16 + , CD14 + ), and pro-inflammatory (CD16 high , CD14 - ) populations; ( d ) pDCs (lineage - , HLA-DR + , CD123 + , CD11c - ) and mDCs (lineage - , HLA-DR + , CD123 - , CD11c + ). ( e ) Histogram showing the percentage IL36R + cells in each leukocyte population. Data were obtained in at least three GPP cases and three sex-matched controls. Results are presented as mean +- SEM. No significant differences were observed between GPP cases and healthy donors. FMO, fluorescence minus 1; IL36R, IL-36 receptor; GPP, generalized pustular psoriasis; mDC, myeloid dendritic cell; Mo, monocytes; pDC, plasmacytoid dendritic cell; SEM, standard error of the mean.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure S2. Sorting strategy and antibody sequence analysis. (A) Sorting strategy. Forward and side scatter (FSC and SSC, respectively) were used to gate on single lymphocytes. Dump channel included CD3, CD8, CD14, CD16, and a viability dye. CD20 + B cells that failed to bind OVA (OVA - ) but did bind to the TBEV EDIII bait coupled with both PE and AF647 fluorophores were purified. (B) For each donor, the number of V gene somatic nucleotide mutations is shown on the left and the amino acid length of the CDR3 is shown on the right. (C) As in B, but for all donors combined. For B and C, horizontal red lines indicate the mean. (D) Distribution of hydrophobicity GRAVY scores at the IGH CDR3 of antibodies from all donors combined and compared with human repertoire (). P < 0.0001 was determined using the Wilcoxon nonparametric test. Related to Fig. 2 , Table S2 , and Table S3 .
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
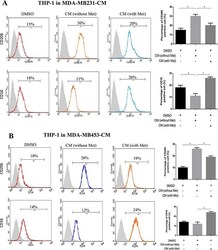
- Figure 1 Metformin treated cancer cells polarized macrophage toward M1 phenotype THP-1 cells were stimulated with PMA (200 nM) for 24 h, then incubated with breast cancer (MDA-MB231/MDA-MB453) conditioned medium (CM) with or without metformin (60 muM) for 6 h, followed by flow cytometry analysis to quantify the amount of CD206, an M2 macrophage marker, and CD16, an M1 marker ( A, B ). Data are expressed as mean +- SD, * p < 0.05. DMSO: control; Met: metformin. Representative flow data shown are from experiments independently performed at least three times.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure S1. Immunopathological characteristics of COVID-19 patients. (A) Doublet cells were excluded by forward scatter height (FSC-H) and forward scatter area (FSC-A) gating for all flow cytometry analysis. Viable cells were identified using fixable viability stain and side scatter area (SSC-A) gating. Neutrophils were identified as cells stained for CD15 + CD16 + among CD14 - CD19 - cells. (B) Flow cytometry analyses of living cells from the whole blood of healthy controls (H.Control) or COVID-19 patients. (C) Frequency and absolute numbers of CD15 + CD16 + neutrophils gated on CD14 - CD19 - live cells from whole blood from healthy controls ( n = 7) or COVID-19 patients ( n = 7). (D) CT of the chest of one patient who died from COVID-19. Images from apical to basal segments (I to IV) show multiple consolidations with air bronchograms in a peripheral and peribronchovascular distribution, more evident in the lower lobes, associated with ground-glass opacities. (E) Representative pulmonary histological findings in 10 cases, autopsied by ultrasound-guided, minimally invasive autopsy. I: The area with interstitial and alveolar neutrophilic pneumonia with diffuse alveolar damage and hyaline membranes in the alveolar space (black arrows). Septal vessel with margination of leukocytes and an intraluminal early fibrin thrombus (green star). II: Area with neutrophilic pneumonia (red arrow), septal thickening, epithelial desquamation, and squamous metaplasia (black arrows). I and II: H&
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
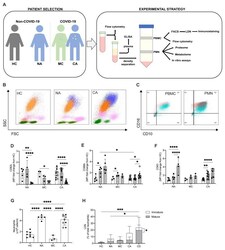
- Figure 2. Circulating neutrophil populations are expanded in COVID-19 and non-COVID-19 ARDS. ( A ) Patient classification (healthy control, HC, non-COVID-19 ARDS, NA, moderate COVID-19, MC, and COVID-19 ARDS, CA), neutrophil isolation, and workflow depicted. ( B ) Representative side scatter (SSC) vs. forward scatter (FSC) plots of stained whole blood from HC, NA and CA displaying lymphocyte (green), monocyte (pink), mature (CD16+, orange) and immature (CD16-, blue) neutrophil populations. ( C ) Representative CD16 vs. CD10 dot plots of stained polymorphonuclear (PMN) and peripheral blood mononuclear cell (PBMC) layers isolated by Percoll gradients from HC (grey), NA (blue) or CA (pink) patients, with quadrant2 (Q2) delimiting the CD16+CD10+ (mature neutrophils) area. ( D - F ) Surface expression of neutrophil activation markers expressed as a fold change of geometric mean fluorescence intensity (MFI) from normal density neutrophils (NDN) respective to the disease state as determined by flow cytometry analysis of mature NDN (CD66b+CD16+, open bars), mature low density neutrophils (LDN) (CD66b+CD16+, horizontal striped bars) and immature LDN (CD66b+CD16-, vertical striped bars) from NA (n = 5), MC (n = 3), or COVID-19 (n = 11; open circles, dexamethasone treated patients) patients. Data are mean +- SD. *p < 0.05, determined by repeated two-way ANOVA and Sidak's post hoc-testing. ( G ) Total neutrophil counts of HC (n = 8), NA (n = 5), MC (n=3) and CA (n = 11) performed by haem
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Effects of Lnc-S100B-2 and MLLT10 on the tumor microenvironment. (a) The expressions of CD3 were detected by IF assay. (b) The percent of CD3 (-) CD16 (+) cells was detected by flow cytometry. (c) The percent of CD11b (+) cells was analyzed by flow cytometry. (d, e) The expression E-cadherin and vimentin was analyzed by IF assay. (f-l) The expressions of E-cadherin, N-cadherin, vimentin, beta -catenin, snail, and slug were detected by qRT-PCR and Western blot. * P < 0.05 versus NC group, # P < 0.05 versus sh-MLLT10 group, & P < 0.05 versus sh-Lnc-S100B-2 group, and one-way ANOVA.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
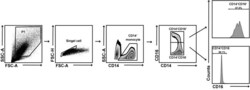
- Figure 4 Flow cytometry analysis of PBMCs Flow cytometry gating strategy to analyze PBMCs. CD14 and CD16 were used to distinguish low-density neutrophils (LDNs), non-classical monocytes, intermediate monocytes, and classical monocytes. Gray = negative control; Red = labeled sample.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 IL-1beta-treated primary synoviocytes affected M1/M2 polarization. The primary synoviocytes were treated with IL-1beta (10 ng/mL) for 24 h and then replaced with fresh medium; 24 h later, the CM was collected to treat THP-1 cells. ( a ) Quantitative PCR analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( b ) ELISA analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( c , d ) Flow cytometry analysis of the expression of CD206, a marker of M2, and the expression of CD16, a marker of M1. Data are expressed as the mean +- SD, * p < 0.05. IgG as a negative control. The data presented are representative of three independent experiments.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Knockdown GRP78 in synoviocytes regulated macrophage polarization to promote the M2 phenotype, using knockdown GRP78 in IL-1beta (10 ng/mL) treated synoviocytes. Then, the CM was collected from synoviocytes to treat THP-1 cells. ( a ) Quantitative PCR analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( b ) ELISA analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( c , d ) Flow cytometry analysis of the expression of CD206, a marker of M2, and the expression of CD16, a marker of M1. Data are expressed as mean +- SD, * p < 0.05. IgG as a negative control. The data presented are representative of three independent experiments.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
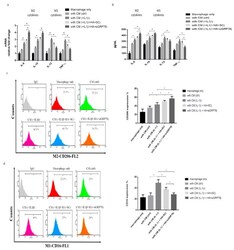
- Figure 6 HMW-HA treatment in synoviocytes affected macrophage polarization toward the M2 phenotype. HMW-HA treated in knockdown GRP78 synoviocytes, and the supernatant was then collected from synoviocytes as CM and then used to treat THP-1. ( a ) Quantitative PCR analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( b ) ELISA analysis of IL-8, IL-10, IL-12, and TNF-alpha. ( c , d ) Flow cytometry analysis of the expression of CD206, a marker of M2, and the expression of CD16, a marker of M1. Data are expressed as a mean +- SD, * p < 0.05. IgG as a negative control. The data presented are representative of three independent experiments.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Retinoic Acid Inhibition Increases the Differentiation Capacity of Hematopoietic Progenitors toward Myeloid and Lymphoid Lineage (A) CFU potential of 500 sorted progenitors (CD45/CD43 + CD34 + ) from DEAB-treated cultures (green) and DMSO control (gray) from four independent experiments (n = 4). CFU potential of progenitors similarly sorted from cord blood (CD45/CD43 + CD34 + ) is shown in red. (B) Lymphoid/myeloid differentiation assay performed on iPSC-derived hematopoietic cells. FACS plots and bar graphs showing non-myeloid (CD33/CD14 - ), CD19 - , CD56 + , and CD56 + CD16 + NK fraction from wells seeded with 50 HSC-like cells (CD45/43 + CD34 hi CD38 lo CD90 + CD45RA - ) (17 individual wells) cultured on OP9 stroma for 4 weeks. Left bar graph shows the cluster size of the NK fraction. Right bar graph shows the frequency of this population in the viable non-stroma fraction. See also Figure S2 . (C) FACS plots showing non-myeloid, CD7 + , CD45RA + multipotent early lymphoid progenitors from 3,500 sorted CD34 + cells cultured on OP9DL1 stroma for 3 weeks from one out of two independent experiments (n = 2). FACS plots are gated on the viable non-stroma fraction. Bar graph show the total output of differentiated cells from progenitors generated with DMSO, DEAB, or RA. FACS gates are based on FMO controls. Data represent mean +- SEM. Asterisks indicate significant differences ( * p < 0.05).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Construction of the PVAX1-HPV58 mE6E7FcGB plasmid and expression of the target proteins assessed by immunofluorescence staining. Notes: ( A ) Diagram of the construction of the recombinant DNA vaccine PVAX1-HPV58 mE6E7FcGB. HPV58 mE6E7 was cloned into the plasmid PCI-sig-Fc-GPI, and the fused cassette sig-HPV58 mE6E7-Fc-GPI was subsequently excised from PCI-sig-HPV58 mE6E7-Fc-GPI plasmid and inserted into the upstream of IRES in PVAX1-IRES-GM/B7.1 vector to create PVAX1-HPV58 mE6E7FcGB. ( B ) The transfected 293T cells were concurrently incubated with a rabbit anti-human IgG-FITC antibody (green) and a mouse anti-human B7-1-PE antibody (red). Immunofluorescence revealed the expression of sig-HPV58 mE6E7-Fc-GPI (a, green, x50) and GM-CSF/B7.1 (b, red, x50) at the cells membrane. Abbreviations: IgG-FITC, immunoglobulin G-conjugated fluorescein isothiocyanate; IRES, internal ribosome entry site; HPV, human papillomavirus.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 HIV-specific responses and immune response capability of the case at 9.5 years of age. a Detection of HIV-specific antibodies at 9.5 years of age by western blot. The case antibody profile is compared with controls that are a high positive, low positive and HIV-negative. HIV proteins corresponding to bands in the blots are shown in the left grey-shaded block; the case profile was positive for the core proteins indicated in pink. b Quantitation of HIV-specific antibodies by multiplex bead array for all isotypes and subclasses (indicated on the left side--IgG1, IgG2, IgG3, IgG4, IgM, IgA1, IgA2) against gp41, Gag, RT, gp120, Nef, Tat, Vpu, peptide Vpu9 and V1V2 scaffold antigens (indicated at the top). Results are expressed as mean fluorescence intensities (MFI)), the colour key shows ranges of MFI according to colour intensity (the darker the more HIV-specific antibody detected). A result is considered positive if above the cut-off (mean +- 3 SD) determined from eight adult uninfected controls. Vpu9 amino acid sequence: STMVDMGHLRLLDVNDL. c Proportions of natural killer (NK) cells that respond to anti-CD16 antibody, and CD4+ and CD8+ T cells that respond to staphylococcal enterotoxin B (SEB) in a whole blood intracellular cytokine (ICC) assay that measures induction of interferon-gamma (IFN-gamma) and interleukin-2 (IL-2). HIV-uninfected adult reference values for comparison ( n = 21; median % and range)--natural killer (NK) anti-CD16%: 37.92 (12-67.6), CD4 SEB%:
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Analysis of neutrophils from control or androgen deprivation therapy (ADT) treated patients. a Percent of circulating neutrophils from control or ADT treated prostate cancer patients that have a mature, segmented appearance morphologically. b Percent Cd11b + CD66b + CD16 lo neutrophils. c Percent myeloid derived suppressor cell (MDSC)-like neutrophils defined as HLA-DR - CD14 - CD33 + CD15 + . d Percent fold change in migration (percent crystal violet positive area of transwell stimulated with a chemoattractant divided by percent area without chemoattractant). e Percent of unstimulated neutrophils producing DHR123. f Percent of neutrophils producing DHR123 following PMA stimulation. g Percent myeloperoxidase + (MPO + ) neutrophils. h Percent of plated netosising neutrophils at 1 and 3 h following purification. Data in a - h are mean +- s.e.m., * p < 0.05, ** p < 0.01, *** p < 0.001 by multivariable linear regression using ADT as its main predictor and adjusting for presence of prostatectomy, age, race, and current status; control prostate cancer patients ( n = 8 in a , c , g and n = 7 in b , d , e , f , h ) and prostate cancer pati e nts receiving ADT ( n = 22 in a , c , g and n = 21 in b , d , e , f , h ).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 Gating strategy for flow cytometry sorting of human CD14 + CD16 + and CD14 + CD16 - monocytes. Peripheral blood mononuclear cells from RA and HC were stained with FITC-conjugated anti-CD14 antibody and APC-conjugated anti-CD16 antibody. CD14 + CD16 + and CD14 + CD16 - monocytes were analyzed and sorted by flow cytometric; the purity of sorted CD14 + CD16 + and CD14 + CD16 - monocytes used for experiments was ~ 90%
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 CD14 + CD16 - monocytes are the main osteoclast precursors in RA. Purified CD14 + CD16 + and CD14 + CD16 - monocytes from RA ( n = 5) and HC ( n = 5) were cultured with rhM-CSF (30 ng/ml) and rhRANKL (50 ng/ml) for osteoclast differentiation. The cells were detected for tartrate-resistant acid phosphatase (TRAP) staining on day 17, and the TRAP-positive multinuclear cells were osteoclasts. The representative charts and the statistical results were shown. a CD14 + CD16 + versus CD14 + CD16 - monocytes in HC (* P = 0.026). b RA versus HC for CD14 + CD16 - monocytes (* P = 0.019). c RA versus HC for CD14 + CD16 + monocytes. * P < 0.05; ns, not significant (Student''s t test, a - c )
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
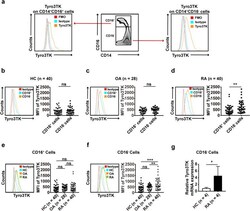
- Fig. 3 The expression of Tyro3TK on CD14 + CD16 - monocytes is increased in RA. a Gating strategy for identifying the expression of Tyro3TK on CD14 + CD16 + and CD14 + CD16 - monocytes. Accordingly, the expression of Tyro3TK on CD14 + CD16 + and CD14 + CD16 - monocytes in HC ( n = 40) ( b ), OA ( n = 28) ( c ), and RA patients ( n = 40, ** P = 0.008) ( d ) were analyzed and presented as the mean fluorescence intensity (MFI). e The expression of Tyro3TK on CD14 + CD16 + monocytes were compared between HC, OA, and RA patients. f The expression of Tyro3TK on CD14 + CD16 - monocytes were compared between HC, OA, and RA patients (** P = 0.004, *** P < 0.001). g Flow cytometry-sorted CD14 + CD16 - monocytes from RA ( n = 4) and HC ( n = 4) were set to detect the mRNA expression of Tyro3TK by qPCR (* P = 0.029). * P < 0.05, ** P < 0.01, *** P < 0.001; ns, not significant (Mann-Whitney U test, b , d , and g ; Student's t test, c ; Kruskal-Wallis test followed by Dunn's post-test for multiple comparisons, e - f )
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
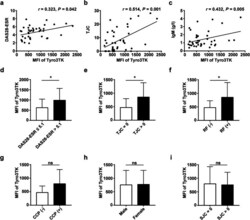
- Experimental details
- Fig. 4 Correlation analysis of Tyro3TK on CD14 + CD16 - monocytes with RA patient clinical manifestations. The associations of Tyro3TK on CD14 + CD16 - monocytes with RA patient DAS28-ESR ( r = 0.323, * P = 0.042) ( a ), tender joint counts (TJC) ( r = 0.514, ** P = 0.001) ( b ), and IgM ( r = 0.432, ** P = 0.005) ( c ) were analyzed. The expression of Tyro3TK on CD14 + CD16 - monocytes were also compared between the different RA patient groups: d RA with high disease activity (DAS28-ESR > 5.1) and non-high disease activity (DAS28-ESR
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
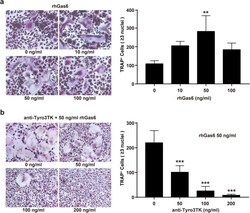
- Experimental details
- Fig. 5 Tyro3TK promotes CD14 + CD16 - monocyte-mediated osteoclastogenesis in RA. Purified CD14 + CD16 - monocytes from RA patients ( n = 8) were cultured with rhM-CSF (30 ng/ml) and rhRANKL (50 ng/ml) under different conditions for osteoclast differentiation. Seventeen days later, the cells were harvested for TRAP staining. The representative charts and the statistical results were shown. a Different concentrations of rhGas6 (0 ng/ml, 10 ng/ml, 50 ng/ml, and 100 ng/ml) were supplemented for osteoclast differentiation ( n = 3 per group, ** P = 0.006). b Different concentrations of anti-Tyro3TK antibody (0 ng/ml, 50 ng/ml, 100 ng/ml, and 200 ng/ml), and 50 ng/ml rhGas6 was supplemented for osteoclast differentiation ( n = 5 per group, *** P < 0.001). ** P < 0.01, *** P < 0.001 (one-way ANOVA test followed by Dunn''s post-test for multiple comparisons, a , b )
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry