Antibody data
- Antibody Data
- Antigen structure
- References [2]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-1037-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD103 (Integrin alpha E) Monoclonal Antibody (Ber-ACT8), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The Ber-ACT8 monoclonal antibody reacts with human CD103, the alpha E integrin. CD103 non-covalently associates with integrin beta7. CD103 is expressed mainly on intraepithelial lymphocytes and a small subset of peripheral lymphocytes. CD103 is also expressed by hairy cell leukemia (HCL) and by some chronic B cell lymphocytic leukemias. In vitro stimulation of human T cells with mitogens induces upregulation of CD103. Epithelial cell antigen (E-cadherin) binds to CD103 and mediates homing of lymphocytes to the intestinal epithelium. Cross-blocking studies suggest that B-Ly7 and Ber-ACT8 see different epitopes. Applications Reported: This Ber-ACT8 antibody has been reported for use in flow cytometric analysis. Applications Tested: This Ber-ACT8 antibody has been pre-titrated and tested by flow cytometric analysis of 2-3 day PHA-activated normal human peripheral blood cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 520 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- Ber-ACT8
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Neoadjuvant anti-OX40 (MEDI6469) therapy in patients with head and neck squamous cell carcinoma activates and expands antigen-specific tumor-infiltrating T cells.
Ber-ACT8: new monoclonal antibody to the mucosa lymphocyte antigen.
Duhen R, Ballesteros-Merino C, Frye AK, Tran E, Rajamanickam V, Chang SC, Koguchi Y, Bifulco CB, Bernard B, Leidner RS, Curti BD, Fox BA, Urba WJ, Bell RB, Weinberg AD
Nature communications 2021 Feb 16;12(1):1047
Nature communications 2021 Feb 16;12(1):1047
Ber-ACT8: new monoclonal antibody to the mucosa lymphocyte antigen.
Kruschwitz M, Fritzsche G, Schwarting R, Micklem K, Mason DY, Falini B, Stein H
Journal of clinical pathology 1991 Aug;44(8):636-45
Journal of clinical pathology 1991 Aug;44(8):636-45
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of 3-day cultured (left) or 3-day PHA-stimulated (right) human peripheral blood cells with Anti-Human CD8a APC (Product # 17-0088-42) and Anti-Human CD103 (Integrin alpha E) FITC. Total viable cells were used for analysis.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
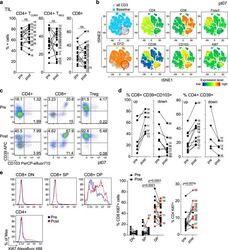
- Fig. 2 Changes in TIL composition after OX40 administration. TIL from a pretreatment biopsy and a surgical specimen after OX40 therapy were assessed for lymphocyte composition and activation markers. The gating strategy is outlined in Supplementary Fig. 3 . a Percentages of CD4+ Tconv cells, CD4+ Treg cells, and CD8+ T cells in each patient before and after OX40 administration, N = 17 patients. b tSNE analysis of the pre and post specimens from patient HNOX07, gated on CD3+ cells. Blue represents the baseline sample, orange the day of surgery sample and gray is the concatenated file. The red circle indicates the population of cells expressing both CD103 and CD39. tSNE analysis was performed on N = 4 patients, one representative patient is shown here, two more patients are shown in Supplementary Fig. 4 . c Flow cytometric analysis of the expression of CD103 and CD39 in CD4+ Tconv cells, CD8+ cells, and CD4+ Treg cells in one immune-responding head and neck squamous cell carcinoma (HNSCC) patient pre- and post OX40 therapy. d Summary of the flow cytometric analysis in (c), left panel depicts CD8+ CD103+ CD39+ T cells and the right panel depicts CD4+ CD39+ T cells; patients with an increase are shown on the left, patients with a decrease are on the right. e Expression of Ki-67 was assessed among memory CD4+ TIL and CD8+ TIL subsets (DN, SP, and DP) in biopsy (pre) and DOS (post) tissue ( N = 17 patients). Blue histograms indicate pre, red indicate post tissues. The left graph sh
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry