Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [3]
- Immunohistochemistry [2]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-773 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- TIMP1 Monoclonal Antibody (F31 P2 A5)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- MA1-773 contains 100 µg of in vitro produced, protein A purified antibody (1 mg/mL) in PBS containing 1 mg/mL BSA and 0.05% sodium azide. MA1-773 detects TIMP-1 from human samples. MA1-773 has been successfully used in Western blot, immunofluorescence, and immunohistochemistry procedures. By Western blot, MA1-773 detects a ~30 kDa band representing TIMP-1 from K562 cell lysate. It is recommended to block with 3% BSA. The MA1-773 immunogen is a synthetic peptide corresponding to residues F(72) Q A L G D A A D I R(82) of mouse TIMP-1.This sequence is 100% conserved in some non-human primate TIMP-1.
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- F31 P2 A5
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Surface-bound matrix metalloproteinase-8 on macrophages: Contributions to macrophage pericellular proteolysis and migration through tissue barriers.
Protective Effects of Kuding Tea (Ilex kudingcha C. J. Tseng) Polyphenols on UVB-Induced Skin Aging in SKH1 Hairless Mice.
Lactobacillus casei Strain Shirota Enhances the In Vitro Antiproliferative Effect of Geniposide in Human Oral Squamous Carcinoma HSC-3 Cells.
Histone lysine dimethyl-demethylase KDM3A controls pathological cardiac hypertrophy and fibrosis.
Diminished bone regeneration after debridement of posttraumatic osteomyelitis is accompanied by altered cytokine levels, elevated B cell activity, and increased osteoclast activity.
Articular cartilage degradation is prevented by tanshinone IIA through inhibiting apoptosis and the expression of inflammatory cytokines.
Intranasal Curcumin Inhibits Pulmonary Fibrosis by Modulating Matrix Metalloproteinase-9 (MMP-9) in Ovalbumin-Induced Chronic Asthma.
Inhibition of transforming growth factor beta signaling by halofuginone as a modality for pancreas fibrosis prevention.
Wang X, Zhang D, Fucci QA, Dollery CM, Owen CA
Physiological reports 2021 Mar;9(5):e14778
Physiological reports 2021 Mar;9(5):e14778
Protective Effects of Kuding Tea (Ilex kudingcha C. J. Tseng) Polyphenols on UVB-Induced Skin Aging in SKH1 Hairless Mice.
Yi R, Zhang J, Sun P, Qian Y, Zhao X
Molecules (Basel, Switzerland) 2019 Mar 13;24(6)
Molecules (Basel, Switzerland) 2019 Mar 13;24(6)
Lactobacillus casei Strain Shirota Enhances the In Vitro Antiproliferative Effect of Geniposide in Human Oral Squamous Carcinoma HSC-3 Cells.
Qian Y, Song JL, Sun P, Yi R, Liu H, Feng X, Park KY, Zhao X
Molecules (Basel, Switzerland) 2018 May 3;23(5)
Molecules (Basel, Switzerland) 2018 May 3;23(5)
Histone lysine dimethyl-demethylase KDM3A controls pathological cardiac hypertrophy and fibrosis.
Zhang QJ, Tran TAT, Wang M, Ranek MJ, Kokkonen-Simon KM, Gao J, Luo X, Tan W, Kyrychenko V, Liao L, Xu J, Hill JA, Olson EN, Kass DA, Martinez ED, Liu ZP
Nature communications 2018 Dec 7;9(1):5230
Nature communications 2018 Dec 7;9(1):5230
Diminished bone regeneration after debridement of posttraumatic osteomyelitis is accompanied by altered cytokine levels, elevated B cell activity, and increased osteoclast activity.
Wagner JM, Jaurich H, Wallner C, Abraham S, Becerikli M, Dadras M, Harati K, Duhan V, Khairnar V, Lehnhardt M, Behr B
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2017 Nov;35(11):2425-2434
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2017 Nov;35(11):2425-2434
Articular cartilage degradation is prevented by tanshinone IIA through inhibiting apoptosis and the expression of inflammatory cytokines.
Jia PT, Zhang XL, Zuo HN, Lu X, Li L
Molecular medicine reports 2017 Nov;16(5):6285-6289
Molecular medicine reports 2017 Nov;16(5):6285-6289
Intranasal Curcumin Inhibits Pulmonary Fibrosis by Modulating Matrix Metalloproteinase-9 (MMP-9) in Ovalbumin-Induced Chronic Asthma.
Chauhan PS, Dash D, Singh R
Inflammation 2017 Feb;40(1):248-258
Inflammation 2017 Feb;40(1):248-258
Inhibition of transforming growth factor beta signaling by halofuginone as a modality for pancreas fibrosis prevention.
Zion O, Genin O, Kawada N, Yoshizato K, Roffe S, Nagler A, Iovanna JL, Halevy O, Pines M
Pancreas 2009 May;38(4):427-35
Pancreas 2009 May;38(4):427-35
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of A431 (Lane 1), HeLa (Lane 2), Jurkat (Lane 3), HT29 (Lane 4) and COS-7 (Lane 5). The blots were probed with Anti-TIMP1 Mouse Monoclonal Antibody (Product # MA1-773, 1:250-1:1000 dilution) and detected by chemiluminescence Goat anti-Mouse IgG (H+L) Secondary Antibody, HRP conjugate (Product # 62-6520, 1:4000 dilution). expected molecular weight is 20 kDa, a 35 kDa band was observed across cell lines tested which may be due to glycosylated TIMP1.Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane by Pierce™ Power Blotter System (22834).The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
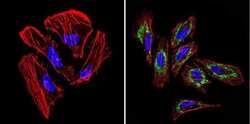
- Experimental details
- Immunofluorescent analysis of TIMP1 (green) in Hela cells. Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes at room temperature and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a TIMP1 Monclonal Antibody (F31 P2 A5) (Product # MA1-773) at a dilution of 1:100 and incubated overnight in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody for 45 minutes at room temperature in the dark. F-actin (red) was stained with a fluorescent phalloidin and nuclei (blue) were stained with DAPI. Images were taken at a 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of TIMP1 (green) in murine cells. Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes at room temperature and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a TIMP1 Monclonal Antibody (F31 P2 A5) (Product # MA1-773) at a dilution of 1:20 and incubated overnight in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody for 45 minutes at room temperature in the dark. F-actin (red) was stained with a fluorescent phalloidin and nuclei (blue) were stained with DAPI. Images were taken at a 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of TIMP1 was done on 70% confluent log phase HepG2 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with TIMP1 (F31 P2 A5) Mouse Monoclonal Antibody (Product # MA1-773) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
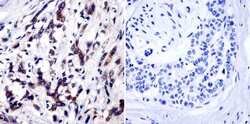
- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human breast carcinoma tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature and probed with a TIMP1 monoclonal antibody (Product # MA1-773) at a dilution of 1:20 or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
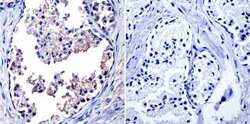
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human prostate tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature and probed with a TIMP1 monoclonal antibody (Product # MA1-773) at a dilution of 1:20 or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 7 FIGURE Timp-1 is localized on the surface of activated macrophages, but does not serve as the receptor for Mmp-8 on macrophages. Quiescent WT peritoneal macrophages were incubated without (Unstim) or with 10 ug/mL bacterial lipopolysaccharide (LPS) for 18 h, fixed, and then immunostained with a red fluorophore and an antibody to Timp-1 or a non-immune primary antibody (murine IgG; Ms IgG). Images shown in (a) are representative of 3 different cell preparations. In (b) surface Timp-1 staining on unstimulated (Unstim) versus LPS-activated wild-type (WT) macrophages was quantified, as described in Methods. Data are mean + SEM; n = 500 cells in 3 separate experiments. In (c-d), WT and Timp-1 -/- peritoneal macrophages were activated with LPS 10 ug/mL LPS for 18 h, fixed, and then either immunostained for surface-bound Mmp-8 (c) or surface type I collagenase activity was measured on equal numbers of cells (5 x 10 6 cells/assay) using type I collagen conjugated to quenched FITC as the substrate and fluorimetry (d), as described in Methods. In (c-d), data are mean + SEM; n = 3 different cell preparations
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot