Antibody data
- Antibody Data
- Antigen structure
- References [45]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [2]
- Immunohistochemistry [2]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-13688 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- TIMP1 Monoclonal Antibody (102D1)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- MA5-13688 targets TIMP-1 in IHC (P) and WB applications and shows reactivity with Human samples. The MA5-13688 immunogen is recombinant human TIMP-1.
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 102D1
- Vial size
- 500 µL
- Concentration
- 0.2 mg/mL
- Storage
- 4° C
Submitted references Substrate Stiffness-Driven Membrane Tension Modulates Vesicular Trafficking via Caveolin-1.
MAPKAP Kinase-2 Drives Expression of Angiogenic Factors by Tumor-Associated Macrophages in a Model of Inflammation-Induced Colon Cancer.
Pulsed focused ultrasound enhances the therapeutic effect of mesenchymal stromal cell-derived extracellular vesicles in acute kidney injury.
Cancer Burden Is Controlled by Mural Cell-β3-Integrin Regulated Crosstalk with Tumor Cells.
EphrinA1-Fc Attenuates Ventricular Remodeling and Dysfunction in Chronically Nonreperfused WT but not EphA2-R-M mice.
The Activity of Matrix Metalloproteinases (MMP-2, MMP-9) and Their Tissue Inhibitors (TIMP-1, TIMP-3) in the Cerebral Cortex and Hippocampus in Experimental Acanthamoebiasis.
The Predictive and Prognostic Role of Topoisomerase IIα and Tissue Inhibitor of Metalloproteinases 1 Expression in Locally Advanced Breast Carcinoma of Egyptian Patients Treated With Anthracycline-based Neoadjuvant Chemotherapy.
Expression and Clinical Significance of Metalloproteases and Their Inhibitors by Endothelial Cells From Invasive Breast Carcinomas.
Molecular events underlying interleukin-6 independence in a subclone of the CMA-03 multiple myeloma cell line.
Expression of matrix metalloproteinases and their inhibitors in different immunohistochemical-based molecular subtypes of breast cancer.
Correlation of bacterial coinfection versus matrix metalloproteinase 9 and tissue inhibitor of metalloproteinase 1 expression in aortic aneurysm and atherosclerosis.
Immunoexpression of matrix metalloproteinases and their inhibitors in different areas of oral squamous cell carcinoma.
Comparative study of the immunohistochemical expression of tissue inhibitors of metalloproteinases 1 and 2 between clearly invasive carcinomas and "in situ" trophoblast invasion.
Expression of metalloproteases and their inhibitors by tumor and stromal cells in ductal carcinoma in situ of the breast and their relationship with microinvasive events.
Study of matrix metalloproteinases and their inhibitors in prostate cancer.
Expression of metalloproteases and their inhibitors in different histological types of breast cancer.
Immunohistochemical study of matrix metalloproteinases and their inhibitors in pure and mixed invasive and in situ ductal carcinomas of the breast.
Expression of metalloproteases and their inhibitors in primary tumors and in local recurrences after mastectomy for breast cancer.
Hypoxia promotes metastasis in human gastric cancer by up-regulating the 67-kDa laminin receptor.
Immunohistochemical expression of matrix metalloproteinases in photodamaged skin by photodynamic therapy.
Comparative analysis and clinical value of the expression of metalloproteases and their inhibitors by intratumor stromal fibroblasts and those at the invasive front of breast carcinomas.
Menstrual activity of matrix metalloproteinases is decreased in endometrium regenerating after thermal ablation.
A mouse to human search for plasma proteome changes associated with pancreatic tumor development.
Gelatinase B expression as a prognostic factor in patients with stage II/III rectal carcinoma treated by postoperative adjuvant therapy.
Serous and mucinous ovarian tumors express different profiles of MMP-2, -7, -9, MT1-MMP, and TIMP-1 and -2.
Overexpression of matrix metalloproteinases and their inhibitors in mononuclear inflammatory cells in breast cancer correlates with metastasis-relapse.
Multinucleate giant cells release functionally unopposed matrix metalloproteinase-9 in vitro and in vivo.
Matrix metalloproteinases and their inhibitors in the chamber angle of normal eyes and patients with primary open-angle glaucoma and exfoliation glaucoma.
Usefulness of MMP-9/TIMP-1 in predicting tumor recurrence in patients undergoing curative surgical resection for gastric carcinoma.
Study of matrix metalloproteinases and their inhibitors in breast cancer.
Matrix metalloproteinase-9 expression correlated with tumor response in patients with locally advanced rectal cancer undergoing preoperative chemoradiotherapy.
Modulation of extracellular matrix components by metalloproteinases and their tissue inhibitors during degeneration and regeneration of rat sural nerve.
In vitro modeling of human pancreatic duct epithelial cell transformation defines gene expression changes induced by K-ras oncogenic activation in pancreatic carcinogenesis.
Tissue inhibitor of metalloproteinase-1 protects human breast epithelial cells from extrinsic cell death: a potential oncogenic activity of tissue inhibitor of metalloproteinase-1.
Tissue inhibitor of matrix metalloproteinase-1 overexpression in M1 myeloblasts impairs IL-6-induced differentiation.
Upregulation of the tissue inhibitor of metalloproteinase-1 protein is associated with progression of human non-small-cell lung cancer.
Tissue inhibitor of metalloproteinase-1 protects human breast epithelial cells against intrinsic apoptotic cell death via the focal adhesion kinase/phosphatidylinositol 3-kinase and MAPK signaling pathway.
Unopposed matrix metalloproteinase-9 expression in human tuberculous granuloma and the role of TNF-alpha-dependent monocyte networks.
Unopposed matrix metalloproteinase-9 expression in human tuberculous granuloma and the role of TNF-alpha-dependent monocyte networks.
Imbalance between expression of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 in invasiveness and metastasis of human gastric carcinoma.
Imbalance between expression of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 in invasiveness and metastasis of human gastric carcinoma.
The in vitro effects of dehydroepiandrosterone on human osteoarthritic chondrocytes.
The in vitro effects of dehydroepiandrosterone on human osteoarthritic chondrocytes.
Abnormal expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in brain arteriovenous malformations.
Systemic arterial expression of matrix metalloproteinases 2 and 9 in acute Kawasaki disease.
Lachowski D, Matellan C, Gopal S, Cortes E, Robinson BK, Saiani A, Miller AF, Stevens MM, Del Río Hernández AE
ACS nano 2022 Mar 22;16(3):4322-4337
ACS nano 2022 Mar 22;16(3):4322-4337
MAPKAP Kinase-2 Drives Expression of Angiogenic Factors by Tumor-Associated Macrophages in a Model of Inflammation-Induced Colon Cancer.
Suarez-Lopez L, Kong YW, Sriram G, Patterson JC, Rosenberg S, Morandell S, Haigis KM, Yaffe MB
Frontiers in immunology 2020;11:607891
Frontiers in immunology 2020;11:607891
Pulsed focused ultrasound enhances the therapeutic effect of mesenchymal stromal cell-derived extracellular vesicles in acute kidney injury.
Ullah M, Liu DD, Rai S, Razavi M, Concepcion W, Thakor AS
Stem cell research & therapy 2020 Sep 14;11(1):398
Stem cell research & therapy 2020 Sep 14;11(1):398
Cancer Burden Is Controlled by Mural Cell-β3-Integrin Regulated Crosstalk with Tumor Cells.
Wong PP, Muñoz-Félix JM, Hijazi M, Kim H, Robinson SD, De Luxán-Delgado B, Rodríguez-Hernández I, Maiques O, Meng YM, Meng Q, Bodrug N, Dukinfield MS, Reynolds LE, Elia G, Clear A, Harwood C, Wang Y, Campbell JJ, Singh R, Zhang P, Schall TJ, Matchett KP, Henderson NC, Szlosarek PW, Dreger SA, Smith S, Jones JL, Gribben JG, Cutillas PR, Meier P, Sanz-Moreno V, Hodivala-Dilke KM
Cell 2020 Jun 11;181(6):1346-1363.e21
Cell 2020 Jun 11;181(6):1346-1363.e21
EphrinA1-Fc Attenuates Ventricular Remodeling and Dysfunction in Chronically Nonreperfused WT but not EphA2-R-M mice.
Whitehurst KS, Chan VA, Estes HK, Valsaraj S, Kent S, Sharma UM, Chase RC, Bhuiyan M, Virag JAI
International journal of molecular sciences 2020 Aug 13;21(16)
International journal of molecular sciences 2020 Aug 13;21(16)
The Activity of Matrix Metalloproteinases (MMP-2, MMP-9) and Their Tissue Inhibitors (TIMP-1, TIMP-3) in the Cerebral Cortex and Hippocampus in Experimental Acanthamoebiasis.
Łanocha-Arendarczyk N, Baranowska-Bosiacka I, Gutowska I, Kolasa-Wołosiuk A, Kot K, Łanocha A, Metryka E, Wiszniewska B, Chlubek D, Kosik-Bogacka D
International journal of molecular sciences 2018 Dec 19;19(12)
International journal of molecular sciences 2018 Dec 19;19(12)
The Predictive and Prognostic Role of Topoisomerase IIα and Tissue Inhibitor of Metalloproteinases 1 Expression in Locally Advanced Breast Carcinoma of Egyptian Patients Treated With Anthracycline-based Neoadjuvant Chemotherapy.
El Rebey HS, Aiad HA, Abulkheir IL, Asaad NY, El-Wahed MM, Abulkasem FM, Mahmoud SF
Applied immunohistochemistry & molecular morphology : AIMM 2016 Mar;24(3):167-78
Applied immunohistochemistry & molecular morphology : AIMM 2016 Mar;24(3):167-78
Expression and Clinical Significance of Metalloproteases and Their Inhibitors by Endothelial Cells From Invasive Breast Carcinomas.
Cid S, Eiro N, González LO, Beridze N, Vazquez J, Vizoso FJ
Clinical breast cancer 2016 Aug;16(4):e83-91
Clinical breast cancer 2016 Aug;16(4):e83-91
Molecular events underlying interleukin-6 independence in a subclone of the CMA-03 multiple myeloma cell line.
Verdelli D, Nobili L, Todoerti K, Mosca L, Fabris S, D'Anca M, Pellegrino E, Piva R, Inghirami G, Capelli C, Introna M, Baldini L, Chiaramonte R, Lombardi L, Neri A
Genes, chromosomes & cancer 2014 Feb;53(2):154-67
Genes, chromosomes & cancer 2014 Feb;53(2):154-67
Expression of matrix metalloproteinases and their inhibitors in different immunohistochemical-based molecular subtypes of breast cancer.
Kim GE, Lee JS, Choi YD, Lee KH, Lee JH, Nam JH, Choi C, Kim SS, Park MH, Yoon JH, Kweon SS
BMC cancer 2014 Dec 16;14:959
BMC cancer 2014 Dec 16;14:959
Correlation of bacterial coinfection versus matrix metalloproteinase 9 and tissue inhibitor of metalloproteinase 1 expression in aortic aneurysm and atherosclerosis.
Roggério A, Sambiase NV, Palomino SA, de Castro MA, da Silva ES, Stolf NG, de Lourdes Higuchi M
Annals of vascular surgery 2013 Oct;27(7):964-71
Annals of vascular surgery 2013 Oct;27(7):964-71
Immunoexpression of matrix metalloproteinases and their inhibitors in different areas of oral squamous cell carcinoma.
Suarez-Roa ML, Asbun-Bojalil J, Ruiz-Godoy LM, Meneses-García AA
Australian dental journal 2012 Sep;57(3):300-7
Australian dental journal 2012 Sep;57(3):300-7
Comparative study of the immunohistochemical expression of tissue inhibitors of metalloproteinases 1 and 2 between clearly invasive carcinomas and "in situ" trophoblast invasion.
Dimo B, Ioannidis I, Karameris A, Vilaras G, Tzoumakari P, Nonni A, Patsouris E, Lazaris AC
Medical oncology (Northwood, London, England) 2012 Sep;29(3):2270-5
Medical oncology (Northwood, London, England) 2012 Sep;29(3):2270-5
Expression of metalloproteases and their inhibitors by tumor and stromal cells in ductal carcinoma in situ of the breast and their relationship with microinvasive events.
González LO, González-Reyes S, Junquera S, Marín L, González L, Del Casar JM, González JM, Vizoso F
Journal of cancer research and clinical oncology 2010 Sep;136(9):1313-21
Journal of cancer research and clinical oncology 2010 Sep;136(9):1313-21
Study of matrix metalloproteinases and their inhibitors in prostate cancer.
Escaff S, Fernández JM, González LO, Suárez A, González-Reyes S, González JM, Vizoso FJ
British journal of cancer 2010 Mar 2;102(5):922-9
British journal of cancer 2010 Mar 2;102(5):922-9
Expression of metalloproteases and their inhibitors in different histological types of breast cancer.
Del Casar JM, González-Reyes S, González LO, González JM, Junquera S, Bongera M, García MF, Andicoechea A, Serra C, Vizoso FJ
Journal of cancer research and clinical oncology 2010 Jun;136(6):811-9
Journal of cancer research and clinical oncology 2010 Jun;136(6):811-9
Immunohistochemical study of matrix metalloproteinases and their inhibitors in pure and mixed invasive and in situ ductal carcinomas of the breast.
Gonzalez LO, Junquera S, del Casar JM, González L, Marín L, González-Reyes S, Andicoechea A, González-Fernández R, González JM, Pérez-Fernández R, Vizoso FJ
Human pathology 2010 Jul;41(7):980-9
Human pathology 2010 Jul;41(7):980-9
Expression of metalloproteases and their inhibitors in primary tumors and in local recurrences after mastectomy for breast cancer.
del Casar JM, Carreño G, González LO, Junquera S, González-Reyes S, González JM, Bongera M, Merino AM, Vizoso FJ
Journal of cancer research and clinical oncology 2010 Jul;136(7):1049-58
Journal of cancer research and clinical oncology 2010 Jul;136(7):1049-58
Hypoxia promotes metastasis in human gastric cancer by up-regulating the 67-kDa laminin receptor.
Liu L, Sun L, Zhao P, Yao L, Jin H, Liang S, Wang Y, Zhang D, Pang Y, Shi Y, Chai N, Zhang H, Zhang H
Cancer science 2010 Jul;101(7):1653-60
Cancer science 2010 Jul;101(7):1653-60
Immunohistochemical expression of matrix metalloproteinases in photodamaged skin by photodynamic therapy.
Almeida Issa MC, Piñeiro-Maceira J, Farias RE, Pureza M, Raggio Luiz R, Manela-Azulay M
The British journal of dermatology 2009 Sep;161(3):647-53
The British journal of dermatology 2009 Sep;161(3):647-53
Comparative analysis and clinical value of the expression of metalloproteases and their inhibitors by intratumor stromal fibroblasts and those at the invasive front of breast carcinomas.
Del Casar JM, González LO, Alvarez E, Junquera S, Marín L, González L, Bongera M, Vázquez J, Vizoso FJ
Breast cancer research and treatment 2009 Jul;116(1):39-52
Breast cancer research and treatment 2009 Jul;116(1):39-52
Menstrual activity of matrix metalloproteinases is decreased in endometrium regenerating after thermal ablation.
Brun JL, Galant C, Delvaux D, Lemoine P, Henriet P, Courtoy PJ, Marbaix E
Human reproduction (Oxford, England) 2009 Feb;24(2):333-40
Human reproduction (Oxford, England) 2009 Feb;24(2):333-40
A mouse to human search for plasma proteome changes associated with pancreatic tumor development.
Faca VM, Song KS, Wang H, Zhang Q, Krasnoselsky AL, Newcomb LF, Plentz RR, Gurumurthy S, Redston MS, Pitteri SJ, Pereira-Faca SR, Ireton RC, Katayama H, Glukhova V, Phanstiel D, Brenner DE, Anderson MA, Misek D, Scholler N, Urban ND, Barnett MJ, Edelstein C, Goodman GE, Thornquist MD, McIntosh MW, DePinho RA, Bardeesy N, Hanash SM
PLoS medicine 2008 Jun 10;5(6):e123
PLoS medicine 2008 Jun 10;5(6):e123
Gelatinase B expression as a prognostic factor in patients with stage II/III rectal carcinoma treated by postoperative adjuvant therapy.
Unsal D, Akyurek N, Uner A, Erpolat OP, Han U, Akmansu M, Mentes BB, Dursun A
American journal of clinical oncology 2008 Feb;31(1):55-63
American journal of clinical oncology 2008 Feb;31(1):55-63
Serous and mucinous ovarian tumors express different profiles of MMP-2, -7, -9, MT1-MMP, and TIMP-1 and -2.
Brun JL, Cortez A, Commo F, Uzan S, Rouzier R, Daraï E
International journal of oncology 2008 Dec;33(6):1239-46
International journal of oncology 2008 Dec;33(6):1239-46
Overexpression of matrix metalloproteinases and their inhibitors in mononuclear inflammatory cells in breast cancer correlates with metastasis-relapse.
González LO, Pidal I, Junquera S, Corte MD, Vázquez J, Rodríguez JC, Lamelas ML, Merino AM, García-Muñiz JL, Vizoso FJ
British journal of cancer 2007 Oct 8;97(7):957-63
British journal of cancer 2007 Oct 8;97(7):957-63
Multinucleate giant cells release functionally unopposed matrix metalloproteinase-9 in vitro and in vivo.
Zhu XW, Price NM, Gilman RH, Recarvarren S, Friedland JS
The Journal of infectious diseases 2007 Oct 1;196(7):1076-9
The Journal of infectious diseases 2007 Oct 1;196(7):1076-9
Matrix metalloproteinases and their inhibitors in the chamber angle of normal eyes and patients with primary open-angle glaucoma and exfoliation glaucoma.
Rönkkö S, Rekonen P, Kaarniranta K, Puustjärvi T, Teräsvirta M, Uusitalo H
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 2007 May;245(5):697-704
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 2007 May;245(5):697-704
Usefulness of MMP-9/TIMP-1 in predicting tumor recurrence in patients undergoing curative surgical resection for gastric carcinoma.
Seo YS, Park JJ, Kim JH, Kim JY, Yeon JE, Kim JS, Byun KS, Bak YT
Digestive diseases and sciences 2007 Mar;52(3):753-9
Digestive diseases and sciences 2007 Mar;52(3):753-9
Study of matrix metalloproteinases and their inhibitors in breast cancer.
Vizoso FJ, González LO, Corte MD, Rodríguez JC, Vázquez J, Lamelas ML, Junquera S, Merino AM, García-Muñiz JL
British journal of cancer 2007 Mar 26;96(6):903-11
British journal of cancer 2007 Mar 26;96(6):903-11
Matrix metalloproteinase-9 expression correlated with tumor response in patients with locally advanced rectal cancer undergoing preoperative chemoradiotherapy.
Unsal Kilic D, Uner A, Akyurek N, Erpolat P, Dursun A, Pak Y
International journal of radiation oncology, biology, physics 2007 Jan 1;67(1):196-203
International journal of radiation oncology, biology, physics 2007 Jan 1;67(1):196-203
Modulation of extracellular matrix components by metalloproteinases and their tissue inhibitors during degeneration and regeneration of rat sural nerve.
Gantus MA, Nasciutti LE, Cruz CM, Persechini PM, Martinez AM
Brain research 2006 Nov 29;1122(1):36-46
Brain research 2006 Nov 29;1122(1):36-46
In vitro modeling of human pancreatic duct epithelial cell transformation defines gene expression changes induced by K-ras oncogenic activation in pancreatic carcinogenesis.
Qian J, Niu J, Li M, Chiao PJ, Tsao MS
Cancer research 2005 Jun 15;65(12):5045-53
Cancer research 2005 Jun 15;65(12):5045-53
Tissue inhibitor of metalloproteinase-1 protects human breast epithelial cells from extrinsic cell death: a potential oncogenic activity of tissue inhibitor of metalloproteinase-1.
Liu XW, Taube ME, Jung KK, Dong Z, Lee YJ, Roshy S, Sloane BF, Fridman R, Kim HR
Cancer research 2005 Feb 1;65(3):898-906
Cancer research 2005 Feb 1;65(3):898-906
Tissue inhibitor of matrix metalloproteinase-1 overexpression in M1 myeloblasts impairs IL-6-induced differentiation.
Haviernik P, Lahoda C, Bradley HL, Hawley TS, Ramezani A, Hawley RG, Stetler-Stevenson M, Stetler-Stevenson WG, Bunting KD
Oncogene 2004 Dec 9;23(57):9212-9
Oncogene 2004 Dec 9;23(57):9212-9
Upregulation of the tissue inhibitor of metalloproteinase-1 protein is associated with progression of human non-small-cell lung cancer.
Aljada IS, Ramnath N, Donohue K, Harvey S, Brooks JJ, Wiseman SM, Khoury T, Loewen G, Slocum HK, Anderson TM, Bepler G, Tan D
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2004 Aug 15;22(16):3218-29
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2004 Aug 15;22(16):3218-29
Tissue inhibitor of metalloproteinase-1 protects human breast epithelial cells against intrinsic apoptotic cell death via the focal adhesion kinase/phosphatidylinositol 3-kinase and MAPK signaling pathway.
Liu XW, Bernardo MM, Fridman R, Kim HR
The Journal of biological chemistry 2003 Oct 10;278(41):40364-72
The Journal of biological chemistry 2003 Oct 10;278(41):40364-72
Unopposed matrix metalloproteinase-9 expression in human tuberculous granuloma and the role of TNF-alpha-dependent monocyte networks.
Price NM, Gilman RH, Uddin J, Recavarren S, Friedland JS
Journal of immunology (Baltimore, Md. : 1950) 2003 Nov 15;171(10):5579-86
Journal of immunology (Baltimore, Md. : 1950) 2003 Nov 15;171(10):5579-86
Unopposed matrix metalloproteinase-9 expression in human tuberculous granuloma and the role of TNF-alpha-dependent monocyte networks.
Price NM, Gilman RH, Uddin J, Recavarren S, Friedland JS
Journal of immunology (Baltimore, Md. : 1950) 2003 Nov 15;171(10):5579-86
Journal of immunology (Baltimore, Md. : 1950) 2003 Nov 15;171(10):5579-86
Imbalance between expression of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 in invasiveness and metastasis of human gastric carcinoma.
Zhang S, Li L, Lin JY, Lin H
World journal of gastroenterology 2003 May;9(5):899-904
World journal of gastroenterology 2003 May;9(5):899-904
Imbalance between expression of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 in invasiveness and metastasis of human gastric carcinoma.
Zhang S, Li L, Lin JY, Lin H
World journal of gastroenterology 2003 May;9(5):899-904
World journal of gastroenterology 2003 May;9(5):899-904
The in vitro effects of dehydroepiandrosterone on human osteoarthritic chondrocytes.
Jo H, Park JS, Kim EM, Jung MY, Lee SH, Seong SC, Park SC, Kim HJ, Lee MC
Osteoarthritis and cartilage 2003 Aug;11(8):585-94
Osteoarthritis and cartilage 2003 Aug;11(8):585-94
The in vitro effects of dehydroepiandrosterone on human osteoarthritic chondrocytes.
Jo H, Park JS, Kim EM, Jung MY, Lee SH, Seong SC, Park SC, Kim HJ, Lee MC
Osteoarthritis and cartilage 2003 Aug;11(8):585-94
Osteoarthritis and cartilage 2003 Aug;11(8):585-94
Abnormal expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in brain arteriovenous malformations.
Hashimoto T, Wen G, Lawton MT, Boudreau NJ, Bollen AW, Yang GY, Barbaro NM, Higashida RT, Dowd CF, Halbach VV, Young WL, University of California, San Francisco BAVM Study Group
Stroke 2003 Apr;34(4):925-31
Stroke 2003 Apr;34(4):925-31
Systemic arterial expression of matrix metalloproteinases 2 and 9 in acute Kawasaki disease.
Gavin PJ, Crawford SE, Shulman ST, Garcia FL, Rowley AH
Arteriosclerosis, thrombosis, and vascular biology 2003 Apr 1;23(4):576-81
Arteriosclerosis, thrombosis, and vascular biology 2003 Apr 1;23(4):576-81
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on 30 µg lysate of Mouse Liver (Lane 1), membrane enriched extract MDA-MB-231 (Lane 2) and conditioned media of MDA-MB -231 (Lane 3). The blots were probed with Anti-TIMP-1 Mouse Monoclonal Antibody (Product # MA5-13688, 1-2 µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Secondary Antibody, HRP conjugate (Product # 62-6520, 1:4000 dilution). A ~ 23 kDa band corresponding to TIMP-1 was observed across cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 10 % Bis-Tris gel (Product # NP0302BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-TIMP1 Monoclonal Antibody (102D1) (Product # MA5-13688) and a 28kDa band corresponding to TIMP1 was observed in MDA-MB-231 but was absent in Daudi which is reported to be negative. An additional band at ~40kDa was observed in Daudi. Whole cell extracts (30 µg lysate) of MDA-MB-231 (Lane 1), MDA-MB-231 treated with Protein Transport Inhibitor (PTI; 1x for 4 hours) (Lane 2), Daudi (Lane 3) and Daudi treated with Protein Transport Inhibitor (1x for 4 hours) (Lane 4) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:100 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
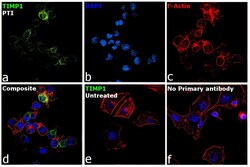
- Experimental details
- Immunofluorescence analysis of TIMP1 was performed using 70% confluent log phase MDA-MB-231 cells treated with Protein Transport Inhibitor (PTI; 1x for 4 hours). The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with TIMP1 Monoclonal Antibody (Product # MA5-13688) at 3µg/mL dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing increased TIMP1 expression and localization to golgi and cytoplasm upon treatment with PTI. Panel e shows untreated cells with lower expression of TIMP1. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
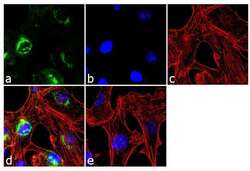
- Experimental details
- Immunofluorescence analysis of TIMP1 was performed using 70% confluent log phase MDA-MB-231 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with TIMP-1 (102D1) Mouse Monoclonal Antibody (Product # MA5-13688) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
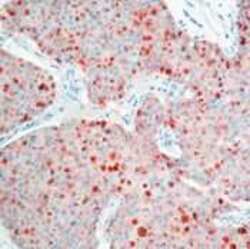
- Experimental details
- Formalin-fixed, paraffin-embedded human breast carcinoma stained with TIMP-1 antibody using peroxidase-conjugate and AEC chromogen. Note cytoplasmic staining of tumor cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
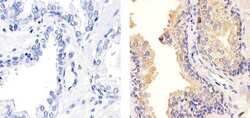
- Experimental details
- Immunohistochemistry analysis of TIMP-1 (102D1) showing staining in the cytoplasm of paraffin-embedded human prostate tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a TIMP-1 Antibody (102D1) Mouse Monoclonal Antibody (Product # MA5-13688) diluted in 3% BSA-PBS at a dilution of 1:100 for 1 hour at 37°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
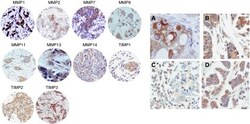
- Experimental details
- Figure 1 Left: examples of TMAs with immunostaining for each protein. 200 x Right: ( A ) immunohistochemical staining of MMP2 in epithelial cells, ( B ) TIMP3 in epithelial cells and fibroblastic cells, ( C ) TIMP3 in inflammatory mononuclear cells, and ( D ) TIMP2 in epithelial cells, fibroblast and inflammatory mononuclear cells. 400 x .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
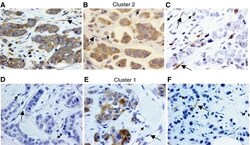
- Experimental details
- Figure 1 Left: examples of tissue with immunostaining for the more significant proteins in patients with tumours belong to poor prognostic group (cluster 2 group: A , B and C ) and better prognostic group (cluster 1 group: D , E and F ; magnification x 400). ( A ) Immunohistochemical staining of matrix metalloproteinase (MMP)-11 in inflammatory mononuclear cells, tumour cells and fibroblastic cells. ( B ) Immunohistochemical staining of MMP-9 in inflammatory mononuclear cells, tumour cells and fibroblastic cell negatives. ( C ) Immunohistochemical staining of tissular inhibitors of metalloproteinase (TIMP)-1 in inflammatory mononuclear cells and in fibroblastic cells, tumour cell negatives. ( D ) No immunohistochemical staining of MMP-9 in inflammatory mononuclear cells, tumour cells and fibroblastic cells. ( E ) Immunohistochemical staining of MMP-11 in tumour cells, inflammatory mononuclear cells and fibroblastic cell negatives. ( F ) Immunohistochemical staining of TIMP-1 in fibroblastic cells, inflammatory mononuclear cells and tumour cell negatives. In all cases, inflammatory mononuclear cells are indicated with small arrows, and fibroblastic cells with large arrows.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
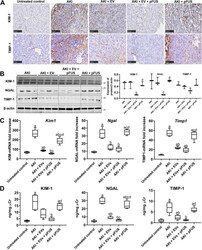
- Experimental details
- Fig. 3 Molecular markers of kidney injury. a Immunohistochemical staining showing expression of injury markers KIM-1 and TIMP-1. Scale bar represents 100 mum. b Western blot for KIM-1, NGAL, TIMP-1, and beta-actin (left), and their respective quantification (right). c Quantitative real-time PCR for Kim1 , Ngal , and Timp1 in the kidney tissue. d ELISA for KIM-1, NGAL, and TIMP-1 measured in the urine. Each group has n = 5 mice. Significant difference a p < 0.05: relative to untreated control; b p < 0.05: relative to AKI; c p < 0.05: relative to AKI-EV; d p < 0.05: relative to AKI-EV-pFUS
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
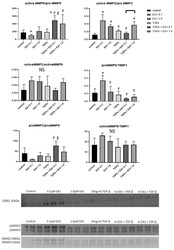
- Experimental details
- Figure 7 Expression Changes in MMP-2, MMP-9, TIMP1, DDR2 and (p)SMAD2/3/total SMAD2/3 in B6 and EphA2-R-M Mouse Hearts. MMP-9, MMP-2 and TIMP-1 expression in ephrinA1-Fc-treated +/-TGF-beta primary cardiac fibroblasts are modulated in a dose-dependent manner ( n = 3/group; significance: * vs. control, + vs. 0.1 uM EA1, ++ vs. TGF-beta). Western blots of DDR2 and (p)SMAD2/3/totalSMAD2/3 show dose-dependent changes in response to ephrinA1-Fc-treatment +/-TGF-beta (NS).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
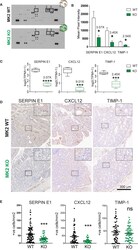
- Figure 2 MK2 deficiency halts the production and secretion of pro-angiogenic factors. (A) Representative images of Mouse Angiogenesis Protein Array (R&D) membranes. Squares highlight the spots corresponding to Timp-1 (1), Serpin-E1 (2) and Cxcl-12 (3). (B) Protein quantification of Timp-1, Serpin-E1, and Cxcl-12 in macrophage culture supernatants from M2-polarized MK2-WT or KO macrophages. Spot intensity was quantified in ImageJ. Data corresponds to three membranes per genotype, where culture supernatants from three independent biological replicates were tested. (C) RNA quantification of Timp-1, Serpin-E1, and Cxcl-12 from M2-polarized MK2-WT or KO macrophages from the experiments shown Figure 1 . Box and whisker plots display median (line), 25 th to 75 th percentiles (boxes) and min and maximum (whiskers) RNA expression levels from 6 biological replicates each of MK2-WT and KO macrophages. (D) Representative pictures of angiogenic factors detected by immunohistochemistry in MK2 WT (upper panel, 10 mice) and MK2 KO (lower panel, 5 mice) colon tumors. (E) Stained slides from serial sections of Swiss-rolled entire colons were scanned and quantified in an automated manner using the positive nuclei algorithm in ImageScope and normalized by area analyzed in mm 2 . Each data point corresponds to a single tumor area, and all tumors from all mice with the same genotype [MK2 WT (10 mice) and MK2 KO (5 mice)] are shown. Scale bar 300 um. In panels (B, C, E) statistical significance was
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
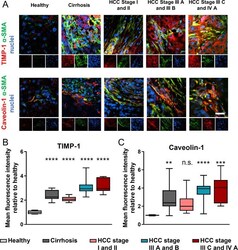
- Caveolin-1 and TIMP-1 are upregulated in cirrhosis and HCC. (A) Representative epifluorescent images of human tissue caveolin-1 and TIMP-1 immunostaining of healthy, cirrhotic, and HCC (hepatocellular carcinoma) liver; protein of interest (red), alpha smooth muscle actin (green) and nuclei (blue); scale bar is 20 mum. Quantification of TIMP-1 (B), and caveolin-1 (C) immunofluorescence staining for panel (A) ( n = number of patients: 5 healthy, 21 cirrhosis, 19 HCC stage I and II, 26 HCC stage III A and B, 7 HCC stage III C and IV A). Whiskers on box and whiskers graphs represent minimum and maximum. Markers denote significant difference from healthy condition by one-way ANOVA with Dunnett's post hoc test, n.s. : not significant, ** 0.001 < p < 0.01, *** 0.0001 < p < 0.001, **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
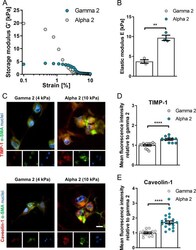
- Figure 6 3D self-assembling peptide gels of high fibrosis-mimicking stiffness trigger upregulation of caveolin-1 and TIMP-1. (A) Average storage modulus ( G ') across 0.1-10% strain sweep and (B) elastic modulus of PeptiGels calculated as E = 2 x G ' (1 + upsilon) where, upsilon = Poisson's ratio of 0.5. Histogram bars represent mean +- s.e.m; dots represent individual data points. (C) Representative epifluorescent images of hepatic stellate cells caveolin-1 and TIMP-1 immunostaining of formalin fixed, paraffin embedded soft (4 kPa, gamma 2), and stiff (10 kPa, alpha 2) 3D MBG PeptiGels; protein of interest (red), alpha smooth muscle actin (green) and nuclei (blue), scale bar is 20 mum. TIMP-1 (D) and caveolin-1 (E) immunofluorescence staining for panel (C). TIMP-1 n = 13 and 11, caveolin-1 n = 14 and 18 for gamma 2 and alpha 2 respectively. Mean +- s.e.m. Markers denote significant difference between gamma 2 and alpha 2 by t test, ** 0.001 < p < 0.01, **** p < 0.0001.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot