Antibody data
- Antibody Data
- Antigen structure
- References [23]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [2]
- Immunoprecipitation [1]
- Other assay [14]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12902 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- MYOD Monoclonal Antibody (5.8A)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- MA5-12902 targets MyoD1 in IF, IM, and IP applications and shows reactivity with Chicken, Human, mouse, and Rat samples. The MA5-12902 immunogen is recombinant mouse MyoD1 protein.
- Reactivity
- Human, Mouse, Rat, Chicken/Avian
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 5.8A
- Vial size
- 500 μL
- Concentration
- 0.2 mg/mL
- Storage
- 4°C
Submitted references MSTN Regulatory Network in Mongolian Horse Muscle Satellite Cells Revealed with miRNA Interference Technologies.
Simple and effective serum-free medium for sustained expansion of bovine satellite cells for cell cultured meat.
Integrative molecular roadmap for direct conversion of fibroblasts into myocytes and myogenic progenitor cells.
FOXP1 Interacts with MyoD to Repress its Transcription and Myoblast Conversion.
Upregulation of miR181a/miR212 Improves Myogenic Commitment in Murine Fusion-Negative Rhabdomyosarcoma.
Myo/Nog cells are nonprofessional phagocytes.
Brain-specific angiogenesis inhibitor 1 is expressed in the Myo/Nog cell lineage.
Myo/Nog cells expressing muscle proteins are present in preretinal membranes from patients with proliferative vitreoretinopathy.
Rhabdomyosarcoma and Wilms tumors contain a subpopulation of noggin producing, myogenic cells immunoreactive for lens beaded filament proteins.
Isolation, cultivation and immunostaining of single myofibers: An improved approach to study the behavior of satellite cells.
Inflammation-associated miR-155 activates differentiation of muscular satellite cells.
PAX3-FOXO1 transgenic zebrafish models identify HES3 as a mediator of rhabdomyosarcoma tumorigenesis.
The RNA Surveillance Factor UPF1 Represses Myogenesis via Its E3 Ubiquitin Ligase Activity.
Genetic disruption of Smad7 impairs skeletal muscle growth and regeneration.
Molecular characterization of an EWSR1-POU5F1 fusion associated with a t(6;22) in an undifferentiated soft tissue sarcoma.
MyoD regulates apoptosis of myoblasts through microRNA-mediated down-regulation of Pax3.
CD90-positive cells, an additional cell population, produce laminin alpha2 upon transplantation to dy(3k)/dy(3k) mice.
Molecular signature of quiescent satellite cells in adult skeletal muscle.
ADP-ribosylation of integrin alpha7 modulates the binding of integrin alpha7beta1 to laminin.
Role of metalloprotease disintegrin ADAM12 in determination of quiescent reserve cells during myogenic differentiation in vitro.
MyoD and myogenin protein expression in skeletal muscles of senile rats.
Roles of peroxisome proliferator-activated receptors delta and gamma in myoblast transdifferentiation.
Roles of peroxisome proliferator-activated receptors delta and gamma in myoblast transdifferentiation.
Budsuren U, Ulaangerel T, Shen Y, Liu G, Davshilt T, Yi M, Bold D, Zhang X, Bai D, Dorjgotov D, Davaakhuu G, Jambal T, Li B, Du M, Dugarjav M, Bou G
Genes 2022 Oct 11;13(10)
Genes 2022 Oct 11;13(10)
Simple and effective serum-free medium for sustained expansion of bovine satellite cells for cell cultured meat.
Stout AJ, Mirliani AB, Rittenberg ML, Shub M, White EC, Yuen JSK Jr, Kaplan DL
Communications biology 2022 Jun 2;5(1):466
Communications biology 2022 Jun 2;5(1):466
Integrative molecular roadmap for direct conversion of fibroblasts into myocytes and myogenic progenitor cells.
Kim I, Ghosh A, Bundschuh N, Hinte L, Petrosyan E, von Meyenn F, Bar-Nur O
Science advances 2022 Apr 8;8(14):eabj4928
Science advances 2022 Apr 8;8(14):eabj4928
FOXP1 Interacts with MyoD to Repress its Transcription and Myoblast Conversion.
Wright WE, Li C, Zheng CX, Tucker HO
Journal of cellular signaling 2021;2(1):9-26
Journal of cellular signaling 2021;2(1):9-26
Upregulation of miR181a/miR212 Improves Myogenic Commitment in Murine Fusion-Negative Rhabdomyosarcoma.
Pozzo E, Giarratana N, Sassi G, Elmastas M, Killian T, Wang CC, Marini V, Ronzoni F, Yustein J, Uyttebroeck A, Sampaolesi M
Frontiers in physiology 2021;12:701354
Frontiers in physiology 2021;12:701354
Myo/Nog cells are nonprofessional phagocytes.
Gerhart J, Gugerty L, Lecker P, Abdalla F, Martin M, Gerhart O, Gerhart C, Johal K, Bernstein J, Spikes J, Mathers K, Bravo-Nuevo A, George-Weinstein M
PloS one 2020;15(8):e0235898
PloS one 2020;15(8):e0235898
Brain-specific angiogenesis inhibitor 1 is expressed in the Myo/Nog cell lineage.
Gerhart J, Bowers J, Gugerty L, Gerhart C, Martin M, Abdalla F, Bravo-Nuevo A, Sullivan JT, Rimkunas R, Albertus A, Casta L, Getts L, Getts R, George-Weinstein M
PloS one 2020;15(7):e0234792
PloS one 2020;15(7):e0234792
Myo/Nog cells expressing muscle proteins are present in preretinal membranes from patients with proliferative vitreoretinopathy.
Gerhart J, Morrison N, Gugerty L, Telander D, Bravo-Nuevo A, George-Weinstein M
Experimental eye research 2020 Aug;197:108080
Experimental eye research 2020 Aug;197:108080
Rhabdomyosarcoma and Wilms tumors contain a subpopulation of noggin producing, myogenic cells immunoreactive for lens beaded filament proteins.
Gerhart J, Behling K, Paessler M, Milton L, Bramblett G, Garcia D, Pitts M, Hurtt R, Crawford M, Lackman R, Nguyen D, Infanti J, FitzGerald P, George-Weinstein M
PloS one 2019;14(4):e0214758
PloS one 2019;14(4):e0214758
Isolation, cultivation and immunostaining of single myofibers: An improved approach to study the behavior of satellite cells.
Lim CL, Ling KH, Cheah PS
Journal of biological methods 2018;5(1):e87
Journal of biological methods 2018;5(1):e87
Inflammation-associated miR-155 activates differentiation of muscular satellite cells.
Onodera Y, Teramura T, Takehara T, Itokazu M, Mori T, Fukuda K
PloS one 2018;13(10):e0204860
PloS one 2018;13(10):e0204860
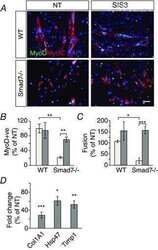
PAX3-FOXO1 transgenic zebrafish models identify HES3 as a mediator of rhabdomyosarcoma tumorigenesis.
Kendall GC, Watson S, Xu L, LaVigne CA, Murchison W, Rakheja D, Skapek SX, Tirode F, Delattre O, Amatruda JF
eLife 2018 Jun 5;7
eLife 2018 Jun 5;7
The RNA Surveillance Factor UPF1 Represses Myogenesis via Its E3 Ubiquitin Ligase Activity.
Feng Q, Jagannathan S, Bradley RK
Molecular cell 2017 Jul 20;67(2):239-251.e6
Molecular cell 2017 Jul 20;67(2):239-251.e6
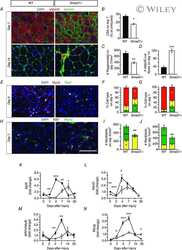
Genetic disruption of Smad7 impairs skeletal muscle growth and regeneration.
Cohen TV, Kollias HD, Liu N, Ward CW, Wagner KR
The Journal of physiology 2015 Jun 1;593(11):2479-97
The Journal of physiology 2015 Jun 1;593(11):2479-97
Molecular characterization of an EWSR1-POU5F1 fusion associated with a t(6;22) in an undifferentiated soft tissue sarcoma.
Deng FM, Galvan K, de la Roza G, Zhang S, Souid AK, Stein CK
Cancer genetics 2011 Aug;204(8):423-9
Cancer genetics 2011 Aug;204(8):423-9
MyoD regulates apoptosis of myoblasts through microRNA-mediated down-regulation of Pax3.
Hirai H, Verma M, Watanabe S, Tastad C, Asakura Y, Asakura A
The Journal of cell biology 2010 Oct 18;191(2):347-65
The Journal of cell biology 2010 Oct 18;191(2):347-65
CD90-positive cells, an additional cell population, produce laminin alpha2 upon transplantation to dy(3k)/dy(3k) mice.
Fukada S, Yamamoto Y, Segawa M, Sakamoto K, Nakajima M, Sato M, Morikawa D, Uezumi A, Miyagoe-Suzuki Y, Takeda S, Tsujikawa K, Yamamoto H
Experimental cell research 2008 Jan 1;314(1):193-203
Experimental cell research 2008 Jan 1;314(1):193-203
Molecular signature of quiescent satellite cells in adult skeletal muscle.
Fukada S, Uezumi A, Ikemoto M, Masuda S, Segawa M, Tanimura N, Yamamoto H, Miyagoe-Suzuki Y, Takeda S
Stem cells (Dayton, Ohio) 2007 Oct;25(10):2448-59
Stem cells (Dayton, Ohio) 2007 Oct;25(10):2448-59
ADP-ribosylation of integrin alpha7 modulates the binding of integrin alpha7beta1 to laminin.
Zhao Z, Gruszczynska-Biegala J, Zolkiewska A
The Biochemical journal 2005 Jan 1;385(Pt 1):309-17
The Biochemical journal 2005 Jan 1;385(Pt 1):309-17
Role of metalloprotease disintegrin ADAM12 in determination of quiescent reserve cells during myogenic differentiation in vitro.
Cao Y, Zhao Z, Gruszczynska-Biegala J, Zolkiewska A
Molecular and cellular biology 2003 Oct;23(19):6725-38
Molecular and cellular biology 2003 Oct;23(19):6725-38
MyoD and myogenin protein expression in skeletal muscles of senile rats.
Dedkov EI, Kostrominova TY, Borisov AB, Carlson BM
Cell and tissue research 2003 Mar;311(3):401-16
Cell and tissue research 2003 Mar;311(3):401-16
Roles of peroxisome proliferator-activated receptors delta and gamma in myoblast transdifferentiation.
Holst D, Luquet S, Kristiansen K, Grimaldi PA
Experimental cell research 2003 Aug 1;288(1):168-76
Experimental cell research 2003 Aug 1;288(1):168-76
Roles of peroxisome proliferator-activated receptors delta and gamma in myoblast transdifferentiation.
Holst D, Luquet S, Kristiansen K, Grimaldi PA
Experimental cell research 2003 Aug 1;288(1):168-76
Experimental cell research 2003 Aug 1;288(1):168-76
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
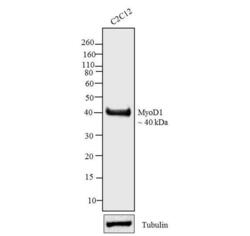
- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of C2C12 (Lane 1). The blots were probed with Anti-MyoD1 Mouse Monoclonal Antibody (Product # MA5-12902, 2.5 µg/mL) and detected by chemiluminescence Goat anti-Mouse IgG (H+L) Secondary Antibody, HRP conjugate (Product # 62-6520, 1:4000 dilution). A 40 kDa band corresponding to MyoD1 was observed across cell lines tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 12 % Bis-Tris gel (Product # NP0342BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane by iBlot® 2 Dry Blotting System (Product # IB21001).The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
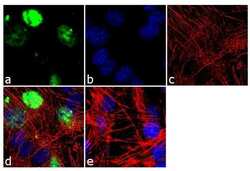
- Experimental details
- Immunofluorescence analysis of MyoD1 was done on 70% confluent log phase C2C12 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with MyoD1 (5.8A) Mouse Monoclonal Antibody (Product # MA5-12902) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing nuclear localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
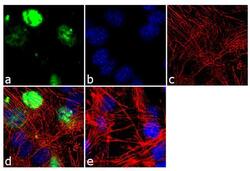
- Experimental details
- Immunofluorescence analysis of MyoD1 was done on 70% confluent log phase C2C12 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with MyoD1 (5.8A) Mouse Monoclonal Antibody (Product # MA5-12902) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing nuclear localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of MyoD1 using MyoD1 Monoclonal Antibody (Product # MA5-12902) on Native Human SW80 Cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
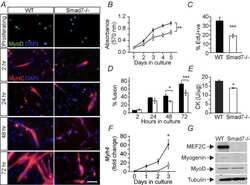
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of MyoD1 using MyoD1 Monoclonal Antibody (Product # MA5-12902) on Native Human SW80 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
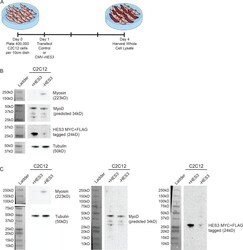
- Figure 5--figure supplement 1. HES3 overexpression inhibits the myogenic differentiation kinetic in mouse muscle myoblasts. ( A ) C2C12 cells were transfected with control or CMV- HES3 , allowed to become confluent and begin fusion, and then whole cell lysate harvested four days post plating. ( B ) Western blots to detect Myosin, MyoD, MYC-HES3, and Tubulin protein expression levels. ( C ) Entire membranes for the cropped images shown in panel B.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
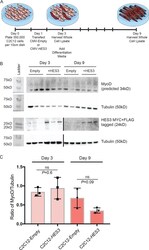
- Figure 5--figure supplement 2. HES3 does not alter MyoD expression during fusion initiation but inhibits MyoD during terminal differentiation. ( A ) C2C12 cells were transfected with CMV-Empty or CMV-HES3, allowed to become confluent, and then whole cell lysate harvested 4 days post plating. Remaining C2C12 conditions were changed to differentiation media, fused, and whole cell lysate was harvested at 9 days post plating. ( B ) The kinetic of MyoD protein expression was evaluated between the beginning and end of myogenic differentiation. Shown are representative Western blots for MyoD, MYC-HES3, and tubulin for three biological replicates from day 3 and day 9 of fusion. HES3 is presented on two independent blots (demarcated by a black line) between days 3 and 9. ( C ) Quantification of MyoD expression levels by densitometry. MyoD was normalized to the tubulin loading control. Plotted is the ratio of MyoD/Tubulin for each sample with n = 3 biological replicates per group +- SEM. Significance was assessed with a two-tailed Student's t-test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
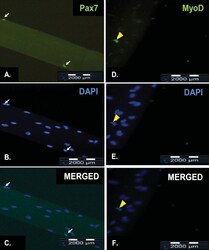
- Figure 4. Representative results of immunofluorescence staining of satellite cells on a freshly isolated myofiber and on a myofiber after cultured in floating condition for 72 h. Pax7 is expressed in the nuclei of quiescent satellite cells (A, white arrow) while MyoD is expressed in the nuclei of activated satellite cells (D, yellow arrowhead). All myonuclei present were revealed by counterstaining with DAPI (B and E). The merged fluorescence image of both Pax7-staining and DAPI-staining of a quiescent satellite cell (C, white arrow). The merged fluorescence image of both MyoD-staining and DAPI-staining of an activated satellite cell (F, yellow arrow). The scale bar represents 2000 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
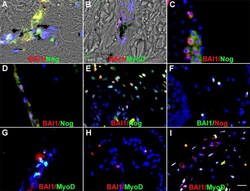
- Fig 5 Co-localization of the R&D BAI1 mAb with antibodies to Noggin and MyoD in the skin and eyes. Tissue sections of human tattooed skin (A and B), human anterior lens tissue (C) and rabbit anterior cavity (D-I) were double labeled with the R&D BAI1 mAb and antibodies to noggin or MyoD. The colors of the fluorescent secondary antibodies are indicated in each photograph. Overlap of red and green appears yellow in merged images. Nuclei were stained with Hoechst dye. Double labeled cells were present in the skin (A and B), anterior human lens tissue (C) and the rabbit lens (D and G), ciliary body (E and H) and cornea (F and I). Bar = 9 muM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
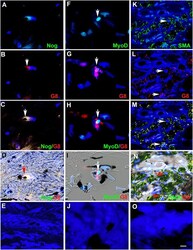
- Fig 2 Myo/Nog cells contain ink in human tattooed skin. Tissue sections of tattooed skin were double labeled with the G8 mAb and antibodies to Noggin (Nog) (A-D), MyoD (F-I) or alpha-SMA (K-N). The colors of the secondary antibodies are indicated in the photographs. Nuclei were stained with Hoechst dye (blue). Images were produced with the epifluorescence (A-E, J and O) and confocal microscopes (F-I and K-N) with 60x lenses. Overlap of green and red fluorescence appears yellow in merged images (C, D, H, I, M and N). Fluorescent photomicrographs were merged with the corresponding DIC image to visualize the ink (black in D, I and N). Some double labeled cells appeared to contain ink (arrows in D, I and N). All ink laden G8+ cells were alpha-SMA+ (N). Smooth muscle cells of blood vessels also contained alpha-SMA (K). Minimal to no background fluorescence was visible after staining with the anti-goat (E), anti-IgM and anti-IgG (I), and anti-rabbit (O) secondary antibodies only. Bar = 28 muM in E and 5.6 muM in A-D and G-O.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
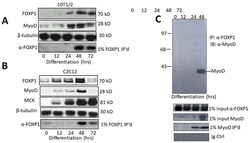
- Figure 1: FOXP1 and MyoD are coordinately expressed and interact physically in vivo . A, B. FOXP1 and MyoD expression peaks at ~3 days following serum withdrawal-mediated differentiation in C2C12 myocytes and or 5-azacytidine-mediated differentiation in 10T1/2 fibroblasts. SDS-PAGE fractionation was followed by anti-MyoD Ab or anti-FOXP1 Ab Western blotting. C. FOXP1 and MyoD heterodimerize in differentiated C2C12 myoblasts. Standard co-immunoprecipitation (Co-IP)/Western blotting was performed 3 days following serum withdrawal. Nuclear lysates were immunoprecipitated with anti-FOXPI Ab, fractionated on SDS gels and then blotted with anti-MyoD Ab. IP, immunoprecipitation; WB, Western blotting; Loading controls (CTRL) include 1% input lanes, 1% IP'd lanes, Ig-only, and beta- tubulin loading controls. Molecular weights are indicated on the right.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
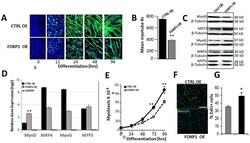
- Figure 5: FOXP1 retards MyoD-mediated myoblastic differentiation. A. C2C12 myocytes were infected with lentivirus Foxp1-EF1a-TetRGFP-Bsd ), cultured for several generations (black arrow). At 18 hr following initiation of differentiation (green arrow), Tet on FOXP1 overexpression (OE) was initiated by addition of 1 µg/mL doxycycline (dox) as detailed in Materials and Methods . OE kinetics of GFP-illuminated C2C12 cells were measured through day 6. Mock transduced control C2C12 (CTRL) myocytes converted to myoblasts by day 4, whereas FOXP1 OE myoblast conversion was significantly retarded as judged by morphology and GFP fluorescence (green). B. Quantification of the data of (A) using epifluorescence microscopy. Shown is the mean of 5 independent measurements (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
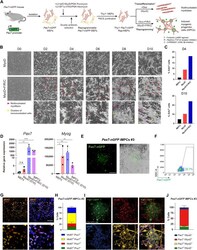
- Fig. 1. A reprogrammable system to study lineage conversion into myogenic cells. ( A ) Schematic of experimental design denoting the conversion of Thy1 + Pax7-nGFP Rep-MEFs into multinucleated myotubes solely by MyoD overexpression or iMPCs by MyoD+F/R/C treatment. ( B ) Representative bright-field images of Thy1 + Rep-MEFs subjected to either MyoD or MyoD+F/R/C treatment for the indicated days. Scale bars, 400 mum. ( C ) Bar graphs based on flow cytometry analysis showing the percentage of EdU + cells in Thy1 + Rep-MEFs subjected to either the MyoD or MyoD+F/R/C condition for the indicated days. ( D ) qRT-PCR for the indicated myogenic genes at day 10. Data are shown as means +- SD. N = 3 cell lines per group. Statistical significance was determined by a two-tailed unpaired t test (* P < 0.05; ** P < 0.01; **** P < 0.0001; n.s., nonsignificant). ( E ) Representative images of a stable Pax7-nGFP iMPC clone at passage 1. Scale bar, 100 mum. ( F ) Flow cytometry analysis of a Pax7-nGFP iMPC clone at P1. ( G ) Representative immunofluorescence images of iMPCs immunostained for Mki67 and Pax7. Nuclei were counterstained with DAPI. Scale bar, 100 mum. ( H ) Quantification of (G). Three different images were quantified from the respective iMPC clone. ( I ) Representative immunofluorescence images of Pax7-nGFP iMPCs immunostained for MyoD and MyHC. Nuclei were counterstained with DAPI. White arrowheads point to mononucleated Pax7-nGFP + cells. Scale bar, 100 mum. ( J ) Quantificatio
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
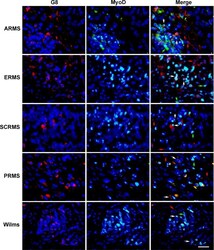
- Fig 5 Co-localization of antibodies to G8 and MyoD in human RMS and Wilms tumor tissue sections. Sections of human ARMS from soft tissue, ERMS from the retroperitoneum, spindle cell RMS (SCRMS) from the testis, pleomorphic RMS (PRMS) from striated muscle and Wilms (kidney) tumors were double labeled with antibodies to G8 (red) and MyoD (green). Nuclei were stained with Hoechst dye (blue). Overlap of red and green appears yellow in merged images. RMS and Wilms tumors sections contain subpopulations of G8+/MyoD- cells (red arrows), G8-/MyoD+ cells (green arrows) and G8+/MyoD+ cells (white arrows). Bar = 9 muM.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoelectron microscopy
Immunoelectron microscopy